ќпыт є1а ѕолучение слабой кислоты.
д>1 д>1 д<1 д>1
CH3COONa + HCl = CH3COOH + NaCl
ацетат натри€ сол€на€ кислота уксусна€ кислота хлорид натри€
ѕри проведении реакции отметим выделение запаха уксуса, что означает по€вление уксусной кислоты в продуктах реакции.
CH3COO- + Na + + H+ + Cl - = CH3COOH + Na + + Cl -
CH3COO- + H+ = CH3COOH (уксусна€ кисллота)
д(CH3COOH) <1 Ц слаба€ кислота = 1*107
д(HCl)>1 - сильна€ кислота = 1,85*10-5
¬ывод: ѕри взаимодействии соли с кислотой можно получить слабую соль. —лабый электролит имеет д<1; д(CH3COOH) <1.
ќпыт є2б —мещение равновеси€ диссоциации слабого основани€.
1. NH4OH + ф-ф (ќкраска раствора стала малиновой (есть гидроксид аммони€), что говорит о наличие щелочной среды)
2. NH4OH + ф-ф + NH4Cl (ќкраска стала розовой)
I) NH4OH↔ NH4+ + OH- II) NH4Cl↔ NH4+ + Cl-
д = 1,7*10-5 < 1
¬ывод: ѕри добавлении к слабому основанию (NH4OH) хлорида аммони€ произошло смещение равновеси€ диссоциации, так как цвет раствора стал более светлым.
д(NH4OH) < 1 д(NH4OH) = [NH4+][ OH-]/[NH4OH]
ќпыт є3 –еакции нейтрализации.
1. NaOH + ф-ф + —Ќ3—ќќЌ→—Ќ3—ќќNa + H2O
гидроксид натри€ уксусна€ кислота ацетат натри€ вода
2. NaOH + ф-ф + HCl ↔ NaCl + H2O
сол€на€ кислота хлорид натри€
ѕри добавлении к NaOH индикатора ф-ф пронаблюдали малиновую окраску раствора (так как щелочна€ среда)
ќба раствора обесцвечены, так как среда стала нейтральной.
д>1 д<1 д>1 д<1
1. NaOH + —Ќ3—ќќЌ→—Ќ3—ќќNa + H2O
Na + + OH- + —Ќ3—ќќЌ → —Ќ3—ќќ- + Na + + H2O
OH- + —Ќ3—ќќЌ → —Ќ3—ќќ- + H2O
д>1 д>1 д>1 д<1
2. NaOH + HCl ↔ NaCl + H2O
Na + + OH- + H+ + Cl - ↔ Na + + Cl - + H2O
OH- + H+ ↔ H2O
¬ывод: –еакции нейтрализации проход€т при участии сильного основани€ и кислоты. ¬ зависимости от добавленного электролита используетс€ разное кол-во кислоты, участвующей в реакции.
ќпыт є4 ”слови€ выпадени€ осадка.
1. Pb(NO3)2 + 2KCl = PbCl2 + 2KNO3
нитрат свинца хлорид кали€ хлорид свинца нитрат кали€
Pb2+ + 2NO3- + 2K+ + 2Cl- = Pb2+ + 2Cl- + 2K+ + 2NO3-
(–еакци€ не прошла, осадка не образовалось).
ѕ [Pb2+][2Cl-]=6,25*10-8 < ѕ–PbCl2 = 2,4*10-4, значит осадок не выпадает.
2. Pb(NO3)2 + 2KI = PbI2↓ + 2KNO3
Pb2+ + 2NO3 - + 2K + + 2I- = PbI2↓ + 2K + + 2NO3 -
Pb2+ + 2I- = PbI2↓
(ќбразовалс€ блест€щий осадок €рко-желтого цвета)
ѕ [Pb2+][2I-]=6,25*10-6 > ѕ–PbI2 = 8,7*10-9 , значит осадок выпадает.
¬ывод: ”слови€ выпадени€ осадка €вл€етс€ разница между ѕ (произведение концентрации) и ѕ–(произведение растворимости). ≈сли ѕ > ѕ–, то осадок выпадает, если ѕ < ѕ–, то осадка не образуетс€.
ќпыт є5 ¬ли€ние величины ѕ– электролита на его способность к химическому взаимодействию.
1. FeSO4 + Na2S → FeS↓ + Na2SO4
сульфат железа II сульфид натри€ сульфид железа II сульфат натри€
ѕолучилс€ осадок черного цвета.
|
|
|
Fe2+ + SO4 -2 + 2Nа + + S2- → FeS↓ + 2Na + + SO4 2-
Fe2+ + S2- → FeS↓
FeS↓ + 2HCl = FeCl2 + H2S
ќсадок (FeS) растворилс€, так как раствор изменил цвет и стал светло-серым, а примерно через несколько минут стал голубоватым (мутным).
FeS↓ + 2H+ + 2Cl - = Fe2+ + 2Cl - + H2S
FeS↓ + 2H+ = Fe2+ + H2S
ѕ–FeS = 5*10-18 
д = дI * дII = 5,7*10-8*1,2*10-15 = 6,84*10-23
–еакци€ протекает в пр€мом направлении
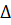 G0 =
G0 = 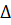 G0298(H2S) +
G0298(H2S) + 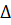 G0298(Fe2+) -
G0298(Fe2+) - 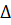 G0298(FeS) - 2
G0298(FeS) - 2 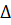 G0298(H+) = -27,97 Ц 92,26 + +100,88 Ц 0 = -19,35 кƒж
G0298(H+) = -27,97 Ц 92,26 + +100,88 Ц 0 = -19,35 кƒж
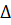 G0 < 0 Ц реакци€ протекает самопроизвольно только в пр€мом направлении.
G0 < 0 Ц реакци€ протекает самопроизвольно только в пр€мом направлении.
2. CuSO4 + Na2S = CuS↓ + Na2SO4
сульфат меди сульфид натри€ сульфид меди сульфат натри€
ѕолучитс€ осадок графитного цвета
Cu2+ + SO4 2- + 2Na + + S2- = CuS↓ + 2Na + + SO4 2-
Cu2+ + S2- = CuS↓
CuS↓ + 2HCl = CuCl2 + H2S
÷вет раствора не изменитс€, никаких признаков протекани€ реакции нет, реакци€ не прошла, осадок не образуетс€.
CuS↓ + 2H+ + 2Cl - = Cu2+ + 2Cl - + H2S
CuS↓ + 2H+ = Cu2+ + H2S
ѕ–CuS = 3,2*10-38 
дH2S = 6,84*10-23
–еакци€ протекает в обратном направлении
 G0298 =
G0298 = 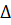 G0298(H2S) +
G0298(H2S) + 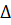 G0298(Cu2+) -
G0298(Cu2+) - 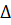 G0298(CuS) - 2
G0298(CuS) - 2 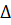 G0298(H+) = -27,97 + 64,98 +
G0298(H+) = -27,97 + 64,98 +
+ 53,76 Ц 2*0 = 90,77 кƒж
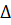 G0 > 0 Ц реакци€ протекает самопроизвольно только в обратном направлении (осадок не образуетс€).
G0 > 0 Ц реакци€ протекает самопроизвольно только в обратном направлении (осадок не образуетс€).
¬ывод: ¬ ходе опыта определили вли€ние ѕ– на реакционную способность электролита, чем больше ѕ–, тем способность электролита к химическому взаимодействию лучше, чем меньше величина ѕ–, тем хуже реакционна€ способность.






