’»ћ»„≈— јя “≈–ћќƒ»Ќјћ» ј
’имическа€ термодинамика €вл€етс€ наукой об энергетических эффектах химических реакций, их направлении и равновесии. »зучает применение законов термодинамики к химическим и физико-химическим процессам.
“ермодинамический метод полезен тем, что позвол€ет:
- установить взаимосв€зь между параметрами равновесной системы, что дает возможность вычислить неизвестный параметр системы по другим известным параметрам;
- оценить величину изменени€ параметров системы при осуществлении какого-либо процесса в ней;
- определить параметры системы после перехода ее из одного состо€ни€ в другое термодинамическое состо€ние или из одного технологического режима в другой режим проведени€ процесса;
- определить принципиальную возможность протекани€ процесса в нужном направлении;
- оценить глубину протекани€ процесса в зависимости от условий.
ѕри определении параметров простейших термодинамических процессов идеальных газов используйте формулы, приведенные в таблицах 1.1 и 1.2. Ќеобходимо обратить особое внимание на правильное применение соответствующих размерностей параметров. »спользуйте следующие значени€ универсальной газовой посто€нной: R = 8,31 ƒж/(мольЈ ) = 1,98 кал/(мольЈ ) = 0,082(лЈатм)/(мольЈ ).
“епловой эффект реакций при стандартных услови€х определ€етс€ по формуле:

 (1.1)
(1.1)
—оответствующие значени€  компонентов приведены в таблице 1.4.
компонентов приведены в таблице 1.4.
ѕри расчете тепловых эффектов при других температурах необходимо опиратьс€ на закон ирхгофа и на вытекающее из него уравнение:
 (интегральна€ форма), (1.2)
(интегральна€ форма), (1.2)
в котором Δс - разность суммы теплоемкостей продуктов и исходных реагентов:
 , (1.3)
, (1.3)
где индекс i Ц стехиометрические коэффициенты соответствующих исходных реагентов; j Ц стехиометрические коэффициенты соответствующих продуктов реакции.
ѕри этом можно сделать следующие допущени€:
1. Δс = 0. “огда ΔHT = ΔH0298.
2. Δc = const. “огда Δc определ€етс€ по значени€м теплоемкости компонентов реакционной системы при стандартных услови€х (таблица 1.4) и значение ΔHT вычисл€ют по формуле:
 . (1.4)
. (1.4)
3. 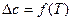 . ѕри этом допущении необходимо учитывать температурную зависимость теплоемкости компонентов реакционной системы
. ѕри этом допущении необходимо учитывать температурную зависимость теплоемкости компонентов реакционной системы
 , (1.5)
, (1.5)
из которой следует
 , (1.6)
, (1.6)
где  . ѕараметры
. ѕараметры  рассчитываютс€ аналогичным образом. «начени€ а,b, c, c/ компонентов реакции приведены в таблице 1.4.
рассчитываютс€ аналогичным образом. «начени€ а,b, c, c/ компонентов реакции приведены в таблице 1.4.
»зменение энтропии реакции при стандартных услови€х определ€ютс€ исход€ из значений S0 (таблица 1.4) по формуле:
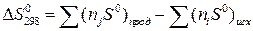 . (1.7)
. (1.7)
ѕри определении  необходимо учитывать температурную зависимость теплоемкости компонентов от температуры:
необходимо учитывать температурную зависимость теплоемкости компонентов от температуры:
 . (1.8)
. (1.8)
¬ычисление  провод€т по формуле:
провод€т по формуле:
 . (1.9)
. (1.9)
«начение  определ€ют по аналогичной формуле
определ€ют по аналогичной формуле
 . (1.10)
. (1.10)
ѕо знаку величин  и
и  определ€ют возможное направление протекани€ реакции при стандартных услови€х и при температуре “.
определ€ют возможное направление протекани€ реакции при стандартных услови€х и при температуре “.
≈сли: DG < 0 Ц самопроизвольное протекание химической реакции возможно;
|
|
|
DG > 0 Ц самопроизвольное протекание химической реакции невозможно;
DG = 0 Ц система находитс€ в равновесии.
“аблица 1.1 —в€зь между основными параметрами состо€ни€ в изопроцессах и определение их функции перехода дл€ n молей вещества ( )
)
| ѕроцесс | ”равнение процесса | —в€зь между параметрами состо€ни€ | –абота в процессе | оличество теплоты, сообщенное в процессе |
| »зобарный | 
| 
| 
| 
|
| »зотерми- ческий | 
| 
| 
| 
|
| »зохорный | 
| 
| 
| 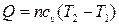
|
| јдиабатный | 
| 


| 

| 
|
“аблица 1.2 »зменение функций состо€ни€ системы в изопроцессах (n - количество вещества,  )
)
| ѕроцесс | »зменение энтальпии | »зменение внутренней энергии | »зменение энтропии | ћол€рна€ теплоемкость |
| »зобарный | Δ Ќ= Δ U+W=Q | Δ U=ncv(T2 Ц T1) | 
| 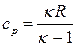
|
| »зотерми-ческий | Δ Ќ = 0 | Δ U = 0 | 
| 
|
| »зохорный | Δ Ќ= nсp(T2 ЦT1) | Δ U= ncv(T2 Ц T1) | 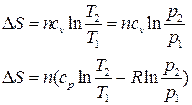
| 
|
| јдиабатный | 
| 
| 
| сад = 0 |
Ћитература: [1], с. 12 Ц 69; [2], с. 8 Ц 49.
ѕримеры решени€ задач
ѕример 1.1 –ассчитайте изменение внутренней энергии гели€ (одноатомный идеальный газ) при изобарном расширении от 5 до 10 л под давлением 196 кѕа.
–ешение: »з таблицы 1.2 дл€ изобарного процесса находим ΔU = ncv(T2 Ц T1). »зменение внутренней энергии идеального газа определ€етс€ только начальной и конечной температурой: T1 = p1V1 /nR, T2 = p2V2 /nR. ƒл€ одноатомного идеального газа Cv = 3/2ЈnR.
DU = ncv (T2 Ц T1) = 3/2nR (T2 Ц T1) = 3/2(p2V2 Ц p1V1) = 3/2(196×103)×(10 - 5)×10-3 = =1470 ƒж.
ќтвет: DU = 1470 ƒж.
ѕример 1.2 –ассчитайте энтальпию сгорани€ метана при 1000 , если даны энтальпии образовани€ при 298 : Df H∞ (CH4) = -17,9 ккал/моль, Df H∞ (CO2) = -94,1 ккал/моль, Df H∞ (H2O(г)) = -57,8 ккал/моль. “еплоемкости газов (в калЈмоль-1Ј -1) в интервале от 298 до 1000 равны:
сp (CH4) = 3,422 + 0,0178 × T; сp (O2) = 6,095+ 0,0033 × T;
сp (CO2) = 6,396 + 0,0102 × T; сp (H2O(г)) = 7,188 + 0,0024 × T.
–ешение: «апишем уравнение процесса:
CH4(г) + 2O2(г) Ѓ CO2(г) + 2H2O(г)
Ёнтальпи€ реакции при температуре “ рассчитываетс€ по закону ирхгоффа  .
.
„тобы им воспользоватьс€, необходимо рассчитать стандартную энтальпию реакции DH0298 и Dc Ц разность суммы теплоемкостей продуктов и исходных реагентов
 .
.
—тандартный тепловой эффект реакции определ€етс€ по формуле
 .
.
ƒл€ данной химической реакции:
DH∞298 = Df H∞ (CO2) + 2∙ Df H∞ (H2O(г)) Ц Df H∞ (CH4) = -94,1+ 2×(-57,8) Ц (-17,9) = = -191,8 ккалЈмоль-1.
Dсp = сp (CO2) + 2∙ сp (H2O(г)) Ц сp (CH4) Ц 2∙ сp (O2) = 6,396 + 0,0102× “ + 2×(7,188 + +0,0024× “) Ц 3,422 Ц 0,0178× “ Ц 2×(6,095 + 0,0033× “) = 5,16 Ц 0,0094× “ (калЈмоль-1Ј -1).


ќтвет: DЌ1000 = -192,5 ккалЈмоль-1.
онтрольные задани€
1 “ермодинамические системы и внешн€€ среда. “ермодинамические параметры состо€ни€ системы.
2 “ермодинамические процессы. –авновесные (квазистатические) и неравновесные процессы.
3 ѕон€тие о внутренней энергии, теплоте и работе. ѕервое начало термодинамики, его математическое выражение.
4 ѕервое начало термодинамики дл€ изохорного и изобарного процессов. ѕон€тие об энтальпии.
5 “ермохими€. «акон √есса и следстви€ из него.
|
|
|
6 “епловой эффект химической реакции, теплота образовани€ химического вещества, теплота сгорани€ химического вещества.
7 «ависимость теплового эффекта реакции от температуры. ”равнение ирхгофа.
8 “еплоемкость. «ависимость теплоемкости от температуры и расчет тепловых эффектов реакций.
9 Ёнерги€ химических св€зей. ќценки теплот химических реакций по энерги€м св€зей.
10 ¬торое начало термодинамики. Ёнтропи€ и термодинамическа€ веро€тность.
11 –асчет изменени€ энтропии дл€ различных процессов.
12 “ретье начало термодинамики. –асчет абсолютного значени€ энтропии.
13 “ермодинамические потенциалы. »спользование свободной энергии √иббса и свободной энергии √ельмгольца дл€ оценки возможности самопроизвольного протекани€ процессов.
14 ’имические потенциалы, их определение, вычисление и свойства.
15 √аз, расшир€€сь от 10 л до 16 л при посто€нном давлении 101,3 кѕа, поглощает 126 ƒж теплоты. ќпределите изменение внутренней энергии газа.
16 ќпределите изменение внутренней энергии, количество теплоты и работу, совершаемую при обратимом изотермическом расширении азота от 0,5 до 4 м3 (начальные услови€: температура 26,8 ∞—, давление 93,2 кѕа).
17 ќдин моль идеального газа, вз€того при 25 ∞— и 100 атм, расшир€етс€ обратимо и изотермически до 5 атм. –ассчитайте работу, поглощенную теплоту, изменение внутренней энергии и энтальпии.
18 акое количество теплоты необходимо дл€ повышени€ температуры 16 г кислорода от 300 до 500 при давлении 1 атм? ак при этом изменитс€ внутренн€€ энерги€?
19 „айник, содержащий 1 кг кип€щей воды, нагревают до полного испарени€ при нормальном давлении. ќпределите работу, количество теплоты, изменение внутренней энергии и энтальпии дл€ этого процесса. ћольна€ теплота испарени€ воды 40,6 кƒж/моль.
20 ќпределите конечную температуру и работу, необходимую дл€ адиабатического сжати€ азота от 10л до 1 л, если начальные температура и давление равны 26,8∞— и 101,3 кѕа соответственно.
21 –ассчитайте количество теплоты, необходимое дл€ нагревани€ воздуха в квартире общим объемом 600 м3 от 20 до 25 ∞—. ѕримите, что воздух Ц идеальный двухатомный газ, а давление при исходной температуре нормальное. Ќайдите изменение внутренней энергии и энтальпии дл€ процесса нагревани€ воздуха.
22 ¬ычислите стандартную энтальпию реакции S(т) + O2(г) → SO2(г), использу€ данные: 2S(т) + 3O2(г) → 2SO3(г), Dr H∞ = - 790 кƒж,
2SO2(г) + O2(г) → SO3(г), Dr H∞ = - 196 кƒж.
23 ¬ычислите тепловой эффект перехода C(алмаз) → C(графит), использу€ данные:
C(графит) + O2(г) → CO2(г), Dr H∞ = - 396,3 кƒж,
C(алмаз) + O2(г) → CO2(г), Dr H∞ = - 398,2 кƒж.
24 ¬ычислите тепловой эффект перехода S(моноклинна€) → S(ромбическа€), использу€ данные:
S(моноклинна€) + O2(г) → SO2(г), Dr H∞ = - 296,96 кƒж,
S(ромбическа€) + O2(г) → SO2(г), Dr H∞ = - 396,65 кƒж.
25 ќпределите стандартную энтальпию образовани€ этилена C2H4 по следующим данным:
C2H4(г) +3O2(г) → 2CO2(г) + 2H2O(г), Dr H∞ = - 1323 кƒж
C(графит) + O2(г) → CO2(г), Dr H∞ = - 393,5 кƒж,
H2(г) + 1/2O2(г) → H2O(г), Dr H∞ = - 241,8 кƒж.
26 ¬ычислите тепловой эффект реакции CH3OCH3(г) → CH3CH2OH(г), если
CH3OCH3(г) + 3O2(г) → 2CO2(г) + 3H2O(г), Dr H∞ = - 1454 кƒж,
CH3CH2OH(г) + 3O2(г) → 2CO2(г) + 3H2O(г), Dr H∞ = - 1402 кƒж.
27 ¬ычислите тепловой эффект реакции Na2CO3(т) → Na2O(т) + CO2(г), если известно
Na2CO3(т) + SiO2(т) → Na2SiO3(т) + CO2(г), Dr H∞ = 81,04 кƒж,
Na2O(т) + SiO2(т) → Na2SiO3(т), Dr H∞ = - 243,17 кƒж.
28 –ассчитайте мольную энтропию неона при 500 , если при 298 и том же объеме энтропи€ неона равна 146,2 ƒж/моль× .
29 –ассчитайте изменение энтропии при нагревании 11,2 л азота от 0 ∞— до 50 ∞— и одновременном уменьшении давлени€ от 1 атм до 0,01 атм.
30 ќдин моль гели€ при 100∞ — и 1 атм смешивают с 0,5 моль неона при 0 ∞— и 1 атм. ќпределите изменение энтропии, если конечное давление равно 1 атм.
|
|
|
31 “ри мол€ идеального одноатомного газа (сv = 3 кал/моль× ), наход€щегос€ при 350 и давлении 5 атм, обратимо и адиабатически расшир€ютс€ до давлени€ 1 атм. –ассчитайте конечные температуру, объем, совершенную работу, изменение внутренней энергии, энтальпии и энтропии в этом процессе.
32 –ассчитайте изменение энтропии при нагревании 0,4 моль NaCl от 20∞ до 850 ∞—. ћольна€ теплоемкость равна: сp (NaCl(тв)) = 45,94 + 16,32×10-3×T (ƒж/моль× ),
сp (NaCl(ж)) = 66,53 ƒж/моль× . “емпература плавлени€ хлорида натри€ 800 ∞—, теплота плавлени€ 31 кƒж/моль.
33 –ассчитайте изменение энтропии при смешении 5 кг воды при 80 ∞— с 10 кг воды при 20 ∞—. ”дельную теплоемкость воды прин€ть равной 4,184 ƒж/г× .
34 –ассчитайте изменение энтропии при добавлении 200 г льда, наход€щегос€ при температуре 0 ∞—, к 200 г воды (90 ∞—) в изолированном сосуде. “еплота плавлени€ льда равна 6 кƒж/моль. ”дельную теплоемкость воды прин€ть равной 4,184 ƒж/г× .
35 Ц 80 ћноговариантна€ задача. ƒл€ реакции, приведенной в таблице 1.3:
1 рассчитайте стандартный тепловой эффект реакции по известным величинам стандартных теплот образовани€ исходных веществ и продуктов реакции;
2 определите DЌ“ из предположени€:
а) D с = 0;
б) D с = const;
в) D с = f (“).
3 определите 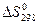 и DS“ дл€ реакции;
и DS“ дл€ реакции;
4 вычислите значение  и DG“ и сделайте вывод о направлении реакции в стандартных услови€х и при температуре “.
и DG“ и сделайте вывод о направлении реакции в стандартных услови€х и при температуре “.
“ермодинамические характеристики веществ приведены в таблице 1.4
“аблица 1.3 ”равнени€ реакций дл€ различных вариантов
| є п/п | –еакци€ | “, |
| 2H2(г) + CO(г) = CH3OH(г) | ||
| 4HCl(г) + O2(г) = 2H2O(г) + 2Cl2(г) | ||
| NH4Cl(т) = NH3(г) + HCl(г) | ||
| 2N2(г) + 6H2O(г) = 4NH3(г) + 3O2(г) | ||
| 4NO(г) + 6H2O(г) = 4NH3(г) + 5O2(г) | ||
| 2NO2(г) = 2NO(г) + O2(г) | ||
| N2O4(г) = 2NO2(г) | ||
| Mg(OH)2(т) = MgO(т) + H2O(г) | ||
| CaCO3(т) = CaO(т) + CO2(г) | ||
| Ca(OH)2(т) = CaO(т) + H2O(г) | ||
 (г) + 2Ќ2ќ(г) = SO2(г) + 2H2(г) (г) + 2Ќ2ќ(г) = SO2(г) + 2H2(г)
| ||
 (г) + 2—ќ2(г) = SO2(г) + 2CO(г) (г) + 2—ќ2(г) = SO2(г) + 2CO(г)
| ||
| 2SO2(г) + O2(г) = 2SO3(г) | ||
| SO2(г) + Cl2(г) = SO2Cl2(г) | ||
| CO(г) + 3H2(г) = CH4(г) + H2O(г) | ||
2CO(г) + SO2(г) =  (г) + 2—ќ2(г) (г) + 2—ќ2(г)
| ||
| CO(г) + Cl2(г) = COCl2(г) | ||
| CO2(г) + H2(г) = CO(г) + H2O(г) | ||
| CH4 + CO2 = 2CO + 2H2 | ||
| CO + H2O = CO2 + H2 | ||
| CH3OH + NH3 = CH3NH2 + H2O | ||
| H2 + O2 = 2H2O | ||
| C6H6 + 3H2 = C6H12 | ||
| C2H6 = C2H4 + H2 | ||
| C2H5OH = CH3COH + H2 | ||
| C2H4(г) + H2O(г) = C2H5OH(г) | ||
| C2H5OH = C2H4 + H2O | ||
| C4H8 = C2H4 | ||
| CH3OH + CO = CH3COOH | ||
| 2NO = N2 + O2 | ||
| C4H10 = C4H8 + H2 бутан бутен | ||
| 2H2S = 2H2 + S2(г) | ||
| FeO(кр) + H2 = Fe(кр) + H2O(г) | ||
| Fe3O4(кр) + H2 = 3FeO(кр) + H2O(г) | ||
| N2 +C2H2 = 2HCN | ||
| CH4 + H2O(г) = CO + 3H2 | ||
| CCl4(г) + 2H2O(г) = CO2 +3H2 | ||
| C(гр) + CO2 = 2CO | ||
| HCOOH(г) = CO2 +H2 | ||
| Fe2O3(кр) +—O = 2FeO(кр) + CO2 | ||
| CO + H2 = C(кр) + H2O(г) | ||
| 2NO + O2 = 2NO2 | ||
| 2CO + O2 = 2CO2 | ||
| 2H2S + 3O2 = 2H2O(г) +2SO2 | ||
| 2ZnS(кр) + 3O2 = 2ZnO(кр) + 2 SO2 | ||
| 4NH3(г) + 3O2 = 2N2 + 6H2O(г) |
|
|
|
“аблица 1.4 “ермодинамические характеристики веществ
| ¬ещество | DЌ0обр

| S0,

| ср = а + bT +  + с“2, + с“2, 
|  , ,

| |||
| а | b ∙ 10-3 | c ∙ 10-6 | c' ∙ 105 | ||||
| H2(г) | 130,7 | 27,3 | 3,27 | - | 0,5 | 28,85 | |
| O2 (г) | 205,2 | 31,46 | 3,39 | - | -3,77 | 29,37 | |
| N2 (г) | 191,5 | 27,87 | 4,27 | - | - | 29,14 | |
| Cl2 (г) | 223,1 | 36,9 | 1,05 | - | -2,52 | 33,96 | |
| H2O (г) | -242,0 | 188,9 | 30,02 | 10,72 | - | 0,33 | 33,6 |
| CO(г) | -110,6 | 197,7 | 28,43 | 4,1 | - | -0,46 | 29,13 |
| CO2 (г) | -393,51 | 213,8 | 44,17 | 9,04 | - | -8,54 | 37,14 |
| NO (г) | 90,3 | 210,7 | 29,58 | 3,85 | - | -0,58 | 29,89 |
| NO2 (г) | 33,89 | 240,3 | 42,16 | 8,54 | - | -6,74 | 37,11 |
| N2O4 (г) | 9,37 | 304,0 | 83,89 | 39,75 | - | -14,90 | 78,99 |
| NH3 (г) | -46,19 | 192,5 | 29,80 | 25,48 | - | -1,67 | 35,65 |
| HCl (г) | -92,4 | 186,9 | 26,54 | 4,61 | - | 1,09 | 29,15 |
| CH4 (г) | -74,9 | 186,4 | 17,46 | 60,5 | 1,118 | - | 35,63 |
| —2Ќ2(г) | 226,75 | 200,82 | 26,44 | 66,65 | -26,48 | - | 43,93 |
| —2H4 (г) | 52,28 | 219,4 | 4,19 | 154,59 | -81,09 | - | 43,63 |
| C2H6 (г) | -84,67 | 229,7 | 4,494 | 182,26 | -74,86 | - | 52,71 |
| —4Ќ8(г) 1-бутен | -0,13 | 305,60 | 21,47 | 258,40 | -80,84 | - | 85,65 |
| —4Ќ10(г) бутан | -126,15 | 310,12 | 18,23 | 303,56 | -92,65 | - | 97,45 |
| C6H6 (г) | 82,93 | 269,2 | -33,90 | 471,87 | -298,34 | - | 81,67 |
| C6H12 (г) | -123,2 | 298,4 | -51,75 | 598,87 | -230,0 | - | 106,34 |
| CH3OH (г) | -202,1 | 239,9 | 15,29 | 105,27 | -31,07 | - | 44,00 |
| CH3CHO (г) | -166,0 | 264,2 | 13,02 | 153,51 | -53,72 | - | 54,64 |
| —Ќ3—ќ—Ќ3(г) | -217,57 | 294,93 | 14,82 | 196,70 | -77,70 | - | 74,90 |
| C2H5OH (г) | -235,3 | 282,0 | 19,09 | 212,86 | -108,69 | - | 73,6 |
| CCl4(г) | -100,42 | 310,12 | 59,36 | 97,00 | -49,57 | - | 83,76 |
| NH4Cl (т) | -315,4 | 94,56 | 49,37 | 133,89 | - | - | 84,20 |
| Mg(OH)2(т) | -924,66 | 63,2 | 54,56 | 66,15 | - | - | 76,99 |
| MgO (т) | -601,24 | 26,94 | 42,59 | 7,28 | - | -6,20 | 37,41 |
| CaCO3 (т) | -1207,9 | 92,9 | 104,5 | 21,92 | - | -25,94 | 81,85 |
| CaO (т) | -635,5 | 39,8 | 49,65 | 4,52 | - | -6,95 | 42,83 |
| Ca(OH)2 (т) | -986,8 | 83,4 | 105,27 | 11,953 | - | -18,979 | 87,55 |
| S2 (г) | 129,1 | 228,2 | 36,11 | 1,09 | - | -3,52 | 32,49 |
| SO2 (г) | -296,9 | 248,2 | 42,55 | 12,55 | - | -5,65 | 39,90 |
| SO3 (г) | -395,2 | 256,23 | 57,36 | 26,88 | - | -13,06 | 50,70 |
| SO2Cl2 (г) | -358,7 | 311,3 | 53,72 | 79,50 | - | - | 77,4 |
| COCl2 (г) | -223,0 | 289,2 | 67,16 | 12,11 | - | -9,03 | 60,67 |
’»ћ»„≈— ќ≈ –ј¬Ќќ¬≈—»≈
Ѕольша€ часть химических реакций обратима. ќбратимые реакции Ц реакции, которые при определенных услови€х протекают одновременно в двух направлени€х: пр€мом и обратном. “ермодинамически химическое равновесие определ€етс€ как такое соотношение концентраций исходных веществ и продуктов реакции, при котором энтропи€ имеет максимальное, а свободна€ энерги€ Ц минимальное значени€.
онстанта равновеси€ химической реакции зависит от природы и температуры и не зависит от концентрации реагирующих веществ. ƒл€ реакции:
mA + nB ↔ pC +qD
константа равновеси€ определ€етс€ из следующего уравнени€:
 (2.1)
(2.1)
”равнение (2.1) выражает закон действующих масс: в состо€нии равновеси€ отношение произведений концентраций продуктов реакции к произведению концентраций исходных веществ есть величина посто€нна€. ѕри этом степени концентраций равны коэффициентам перед формулами данных веществ или элементов.
онцентрации газообразных веществ можно заменить соответствующими парциальными давлени€ми, при этом константа равновеси€ обозначаетс€ Kp.
онстанты Kc и Kp дл€ одной и той же реакции численно равны между собой, но между ними существует определенна€ зависимость:
 , (2.2.)
, (2.2.)
где Δn Ц приращение числа молей газообразных веществ.
ƒл€ определени€ направлени€ реакции при изменении одного или нескольких факторов равновеси€ необходимо руководствоватьс€ принципом Ће-Ўателье: если на систему, наход€щуюс€ в равновесии, оказать внешнее воздействие (изменить температуру, давление или концентрацию), то в ней произойдут изменени€, противодействующие этому воздействию и стрем€щиес€ восстановить первоначальные услови€.
онстанта равновеси€ св€зана с Δr G0 химической реакции:
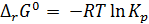 . (2.3)
. (2.3)
»зменение ΔrG или ΔrF в химической реакции при заданных (не об€зательно равновесных) парциальных давлени€х pi или ci концентраци€х реагирующих веществ можно рассчитать по уравнению изотермы химической реакции (изотермы ¬ант-√оффа):
 . (2.4)
. (2.4)
¬еличину KТp, иногда, называют Ђконстантой неравновеси€ї по аналогии с константой равновеси€ Kp, так как она тоже выражаетс€ через парциальные давлени€, однако, неравновесные.
|
|
|
«ависимость константы равновеси€ от температуры описываетс€ уравнением изобары химической реакции (изобары ¬ант-√оффа)
 (2.5)
(2.5)
и изохоры химической реакции (изохоры ¬ант-√оффа)
 (2.6)
(2.6)
–аздел€€ переменные в уравнени€х (2.5) и (2.6) и интегриру€ при ΔH = const (или ΔU = const), получаем:


где ¬ и ¬Т Ц некие константы.
»нтегриру€ в пределах K1, K2 и T1, T2, получим:
 , (2.7)
, (2.7)
 .
.
ѕо этому уравнению, зна€ константы равновеси€ при двух разных температурах, можно рассчитать ΔrH реакции. —оответственно, зна€ ΔrH реакции и константу равновеси€ при одной температуре, можно рассчитать константу равновеси€ при другой температуре.
Ћитература: [1], с. 79 Ц 87; [2], с. 92 Ц 103.
ѕримеры решени€ задач
ѕример 2.1 ќбратима€ химическа€ реакци€ протекает по уравнению:
A +2B ↔ C.
ѕри установившемс€ равновесии концентрации участвующих в реакции веществ составили: [A] = 0,6 моль/л; [B] = 1,2 моль/л; [C] = 2,16 моль/л. ќпределите исходные концентрации веществ A и B.
–ешение: онстанту равновеси€ определ€ем по уравнению (2.1)

—огласно уравнению реакции, из 1 моль ј и 2 моль ¬ образуетс€ 1 моль —. »з сказанного следует, что на образование каждых 2,16 моль — потребуетс€ 2,16 моль ј и 2,16∙2 = 4,32 моль ¬.
“аким образом, исходные концентрации веществ ј и ¬, т.е. число молей веществ ј и ¬ на каждый литр смеси до начала реакции, составл€ли
[A] = 0,6 ∙ 2,16 = 2,76 моль/л,
[B] = 1,2 ∙4,32 = 5,52 моль/л.
ќтвет: [A] =2,76 моль/л, [B] = 5,52 моль/л.
ѕример 2.2 ƒл€ реакции
N2(г) + 3H2(г) = 2NH3(г)
при 298 Kp = 6,0 ∙ 105, а  fH0 (NH3) = -46,1 кƒж/моль. ќцените значение константы равновеси€ при 500 .
fH0 (NH3) = -46,1 кƒж/моль. ќцените значение константы равновеси€ при 500 .
–ешение: —тандартна€ мольна€ энтальпи€ реакции равна
 rH0 =
rH0 =  fH0 (NH3) = -92,2 кƒж/моль.
fH0 (NH3) = -92,2 кƒж/моль.
—огласно уравнению (2.7),

откуда 2 = 0,18.
ќтвет: 2 = 0,18.
онтрольные задани€
”казание: во всех задачах считать газы идеальными.
81 «акон действи€ масс. онстанты равновеси€ и св€зь между ними. «ависимость констант равновеси€ от температуры.
82 —мещение химического равновеси€. ¬ли€ние температуры, давлени€ и концентрации на химическое равновесие.
83 »зотерма ¬ант-√оффа. »зменение энергии √иббса и энергии √ельмгольца при химической реакции.
84 ”равнени€ изобары и изохоры реакции и их термодинамический вывод.
85 —в€зь константы химического равновеси€ с максимальной работой реакции.
86 √етерогенные химические равновеси€ и особенности их термодинамического описани€.
87 –еакци€ между веществами ј и ¬ выражаетс€ уравнением
ј + 2¬ = —.
Ќачальна€ концентраци€ вещества ј равна 0.3 моль/л, а вещества ¬ Ц 0,5 моль/л. онстанта скорости реакции равна 0,4. ќпределить начальную скорость реакции и скорость реакции по истечении некоторого времени, когда концентраци€ вещества ј уменьшитс€ на 1,0 моль.
88 ѕри нагревании смеси углекислого газа и водорода в закрытом сосуде устанавливаетс€ равновесие. онстанта равновеси€ реакции
CO2 + H2 ↔ CO + H2O
при температуре 850∞— равна единице. —колько процентов углекислого газа превратитс€ в окись углерода, если смешать 1 моль углекислого газа и 4 моль водорода?
89 ѕри температуре 3000∞— 40% двуокиси углерода диссоциирует по уравнению
2CO2 ↔ 2CO + O2.
ќпределить Kp при этой температуре.
90 ќпределить константу равновеси€ реакции
CO + H2O ↔ CO2 + H2
при температуре 1000 на основании следующих данных:
| H2 | H2O | CO | CO2 | |
| ΔЌ∞ (кал/моль) | ||||
| ΔS∞ (кал/град∙моль) | 31,211 | 45,106 | 47,301 | 51,06 |
Δ—p = 1,72 кал/моль.
91 ѕри 693 константа равновеси€ реакции H2 + I2 = 2HI с = 50,25. ¬ычислите массу образующегос€ иодида водорода, если в реактор введено 0,846 г I2 и 0,0212 г Ќ2.
92 ѕри 693 константа равновеси€ реакции H2 + I2 = 2HI — = 50,25. ¬ каком направлении будет протекать данный процесс, если исходные концентрации Ќ2, I2 и HI соответственно равны: а) 2,0; 5,0; 10,0 моль/л; б) 1,5; 0,25; 5,0 моль/л; в) 1,0; 1,99; 10,0 моль/л.
93 ќпределите константу равновеси€ реакции SO2 + 1/2O2 = SO3 при 700 , если при 500 р = 588,9 ѕа-1/2, а тепловой эффект реакции в этом диапазоне температур равен Ц99,48 кƒж/моль.
94 ѕри 2000 ∞— и общем давлении 1 атм 2 % воды диссоциировано на водород и кислород. –ассчитайте константу равновеси€ реакции Ќ2ќ(г) = Ќ2(г) + 1/2ќ2(г) при этих услови€х.
95 онстанта равновеси€ реакции —ќ(г) + Ќ2ќ(г) = —ќ2(г) + Ќ2(г) при 500 ∞— равна р = 5,5. —месь, состо€щую из 1 моль —ќ и 5 моль Ќ2ќ, нагрели до этой температуры. –ассчитайте мольную долю Ќ2ќ в равновесной смеси.
96 онстанта равновеси€ реакции N2O4(г) = 2NO2(г) при 25 ∞— равна р = 0,143. –ассчитайте давление, которое установитс€ в сосуде объемом 1 л, содержащем 1 г N2O4 при этой температуре.
97 —осуд объемом 3 л, содержащий 1,79∙10-2 моль I2, нагрели до 973 . ƒавление в сосуде при равновесии оказалось равно 0,49 атм. —чита€ газы идеальными, рассчитайте константу равновеси€ при 973 дл€ реакции I2(г) = 2I(г).
98 —осуд объемом 1 л, содержащий 0,341 моль –Cl5 и 0,233 моль N2, нагрели до 250 ∞—. ќбщее давление в сосуде при равновесии оказалось равно 29,33 атм. —чита€ все газы идеальными, рассчитайте константу равновеси€ при 250 ∞— дл€ протекающей в сосуде реакции: –—l5(г) = –—l3(г) + —l2(г).
99ƒл€ реакции 2HI(г) = H2(г) + I2(г) константа равновеси€ р = 1,83∙10-2 при 698,6 . —колько граммов HI образуетс€ при нагревании до этой температуры 10 г I2 и 0,2 г H2 в трехлитровом сосуде? „ему равны парциальные давлени€ H2, I2 и HI?
100 онстанта равновеси€ реакции H2(г) + I2(г) = 2HI(г) при 717 равна 46,7. ќпределите количество разложившегос€ HI при нагревании 1 моль HI до этой температуры.
101 онстанта равновеси€ реакции CO(г) + H2O(г) = CO2(г) + H2(г) при 800 равна 4,12. —месь, содержаща€ 20 % —O и 80 % Ќ2ќ нагрета до 800 . ќпределите состав смеси при достижении равновеси€, если вз€т 1 кг вод€ного пара.
102ѕри 1273 и общем давлении 30 атм в равновесной смеси —O2 (г) + —(тв) = = 2—ќ(г) содержитс€ 17 % (по объему) —ќ2. —колько процентов —ќ2 будет содержатьс€ в газе при общем давлении 20 атм?
103 ѕри смешении 1 моль уксусной кислоты и 1 моль этилового спирта реакци€ протекает по уравнению CH3COOH(г) + C2H5OH(г) = CH3COOC2H5(г) + H2O(г). ѕри достижении равновеси€ в реакционной смеси находитс€ 1/3 моль кислоты, 1/3 моль спирта, 2/3 моль эфира и 2/3 моль воды. ¬ычислите количество моль эфира, которое будет в реакционной смеси по достижению равновеси€ при следующих услови€х: 1 моль кислоты, 1 моль спирта и 1 моль воды.
104-123 ћноговариантна€ задача. ¬ычислите величину константы равновеси€ при заданной температуре “. ѕримите, что тепловой эффект реакции в этом температурном интервале не зависит от температуры. ѕримечание: если агрегатные состо€ни€ не указаны, вещества наход€тс€ в газовой фазе.
“аблица 2.1 ”равнени€ реакций дл€ различных вариантов
| є п/п | –еакци€ | “, | “1 | “2 |  H 0,
кƒж/ моль H 0,
кƒж/ моль
|
| 2—ќ + 4Ќ2= —2Ќ5ќЌ + Ќ2ќ | 7,0∙10-8 (“ 1 = 400 ) | 8,4∙10-8 (“ 2 = 500 ) | - | ||
| 2HgO(тв) = 2Hg + ќ2 | 1,37∙1014 (“ 1 = 693 ) | - | |||
| Ag2CO3(тв) =Ag2O(тв) + CO2 | 3,98∙10-4 (“ 1 = 350 ) | 1,86∙10-4 (“ 2 = 450 ) | - | ||
| 2—Ќ3ќЌ = —2Ќ4 + 2Ќ2ќ | 1,9 (“ 1 = 700 ) | 5,57 (“ 2 = 1000 ) | - | ||
| 2C3H6 = C2H4 + C4H8 | 5,03∙10-2 (“ 1 = 300 ) | 8,77∙10-2 (“ 2 = 600 ) | - | ||
| Hr(aq) + O2 = HrO2(aq) (Hr Ц гемеритрин, кислородперенос€щий пигмент) | (“ 1 = 273 ) | (“ 2 = 298 ) | - | ||
| —Ќ3ќЌ = Ќ—Ќќ + Ќ2 | 6,1∙10-4 (“ 1 = 700 ) | 5,6∙10-4 (“ 2 = 500 ) | - | ||
| 4HCl + O2 = 2H2O + Cl2 | 6,72∙10-4 (“ 1 = 700 ) | 3,96∙10-6 (“ 2 = 900 ) | - | ||
| —3Ќ6 + 3/2ќ2 = —Ќ3—Ќќ + Ќ—ќќЌ | 9,1∙1015 (“ 1 = 600 ) | - | 168,15 | ||
| NH4Cl(тв) = NH3 + HCl | 3,80∙107 (“ 1 = 400 ) | 1,81∙1012 (“ 2 = 500 ) | - | ||
| —ќ + 2Ќ2 = —Ќ3ќЌ | 3,88∙10-11 (“ 1 = 400 ) | - | 113,13 | ||
| Mg(OH)2 (тв) = MgO(тв) + H2O | 7,26∙105 (“ 1 = 350 ) | 5,59∙109 (“ 2 = 500 ) | - | ||
| 2C5H11 + 2H2 = 2C4H10 +C2H6 | 1,46 (“ 1 = 373 ) | - | 3,18 | ||
| S2 + 4CO2 = 2SO2 + 4CO | 1,58∙1011 (“ 1 = 850 ) | 3,39∙1011 (“ 2 = 950 ) | - | ||
| SO2 + Cl2 = SO2Cl2 | 8,51∙10-6 (“ 1 = 350 ) | 2,27∙10-7 (“ 2 = 450 ) | - | ||
| C6H6 + 3H2 = C6H12 | 9,54∙10-6 (“ 1 = 350 ) | 2,30∙10-6 (“ 2 = 450 ) | - | ||
| H2 + Cl2 = 2HCl | 3,25∙1011 (“ 1 = 900 ) | 2,67∙1010 (“ 2 = 1000 ) | - | ||
| H2 + I2 = 2HI | 61,6 (“ 1 = 633 ) | 41,7 (“ 2 = 718 ) | - | ||
| C6H5C2H5 + 3H2 = C6H11C2H5 | 5,98∙10-14 (“ 1 = 485 ) | 9,30∙10-17 (“ 2 = 565 ) | - | ||
| I2 = 2I | 1,15∙102 (“ 1 = 950 ) | 7,41∙102 (“ 2 = 1050 ) | - |
‘ј«ќ¬џ≈ –ј¬Ќќ¬≈—»я
ќбщие закономерности, которым подчин€ютс€ равновесные системы, содержащие любое число фаз и компонентов, устанавливаютс€ правилом фаз √иббса, согласно которому
 , (3.1)
, (3.1)
где Ц число компонентов системы, ‘ Ц число фаз, n Ц число внешних факторов, определ€ющих существование системы (p, T, c), — Ц число степеней свободы, показывающа€ число переменных, которым можно придавать произвольные значени€, не измен€€ число фаз.
оличественно условие равновеси€ фаз в однокомпонентных системах выражаетс€ уравнением лаузиуса:
 , (3.2)
, (3.2)
где  - производна€, описывающа€ изменение давлени€ пара над жидкостью или твердым телом при испарении или возгонке;
- производна€, описывающа€ изменение давлени€ пара над жидкостью или твердым телом при испарении или возгонке;  - мол€рна€ теплота равновесного фазового перехода при температуре
- мол€рна€ теплота равновесного фазового перехода при температуре  и давлении
и давлении  ;
;  - температура фазового перехода;
- температура фазового перехода;  - изменение объема одного мол€ вещества при фазовом переходе.
- изменение объема одного мол€ вещества при фазовом переходе.
»нтегрирование уравнени€ лаузиуса из предположени€  дл€ равновеси€ газ-жидкость и газ-твердое тело приводит к уравнению лаузиуса- лапейрона:
дл€ равновеси€ газ-жидкость и газ-твердое тело приводит к уравнению лаузиуса- лапейрона:
 . (3.3)
. (3.3)
ѕоэтому дл€ системы газ-жидкость тангенс угла наклона зависимости ln р =f(1/“) дает значение  , а дл€ системы газ-твердое тело Ц значение
, а дл€ системы газ-твердое тело Ц значение  . “очка пересечени€ этих пр€мых дает координаты тройной точки.
. “очка пересечени€ этих пр€мых дает координаты тройной точки.
“емпературу кипени€ “кип вещества при нормальном давлении определ€ют по формуле, полученной из уравнени€ лаузиуса- лапейрона:
 . (3.4)
. (3.4)
ƒл€ проверки применимости правила “рутона к данной системе необходимо найти отношение  . ≈сли оно отличаетс€ от значени€ 89
. ≈сли оно отличаетс€ от значени€ 89  , то правило “рутона не применимо.
, то правило “рутона не применимо.
¬заимосв€зь параметров в равновесной системе газ-твердое тело выражаетс€ уравнением:
 . (3.5)
. (3.5)
“еплоту плавлени€ вещества  при температуре тройной точки определ€ют исход€ из теплоты возгонки и теплоты испарени€:
при температуре тройной точки определ€ют исход€ из теплоты возгонки и теплоты испарени€:
 . (3.6)
. (3.6)
ѕараметры процесса возгонки определ€ют по следующим формулам:
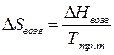 , (3.7)
, (3.7)
 , (3.8)
, (3.8)
 , (3.9)
, (3.9)
 (3.10)
(3.10)
Ћитература: [1], с. 70 Ц 78, 88-112; [2], с. 65 Ц 91.
ѕримеры решени€ задач
ѕример 3.1 ќпределить число степеней свободы системы
CaCO3 ↔ CaO + CO2
–ешение: ¬ системе существуют три фазы: две твердые Ц CaCO3 и CaO Ц и одна газообразна€ Ц CO2. “ак как из двух фаз системы CaO и CO2 может быть получена треть€ фаза Ц CaCO3, то число независимых компонентов равно 2.
Ќа основании уравнени€ правила фаз (3.1) определим число степеней свободы:
— = Ц ‘ +2 = 2-3+2 =1.
“аким образом, произвольно можно мен€ть в определенных пределах только один фактор равновеси€, например, температуру.
онтрольные задани€
124 √етерогенные системы. омпонент, фаза, число степеней свободы системы. ѕравило фаз √иббса.
125 ”равнение лаузиуса. ѕрименение данного уравнени€ дл€ описани€ процессов плавлени€, испарени€, возгонки.
126 ќднокомпонентные гетерогенные системы. ‘азова€ диаграмма воды.
127 ќднокомпонентные гетерогенные системы. ‘азова€ диаграмма серы.
128 «акон –аул€ дл€ смеси летучих жидкостей. ѕоложительные и отрицательные отклонени€ от закона –аул€.
129 –азделение неограниченно смешивающихс€ жидкостей с помощью фракционной перегонки. 1-ый закон оновалова.
130 јзеотропные смеси. 2 Цой закон оновалова. ћетоды разделени€ азеотропных смесей.
131 ƒиаграмма состо€ни€ двухкомпонентной системы с ограниченной растворимостью в жидкой фазе. ритическа€ температура растворимости. ѕравило јлексеева. ѕравило рычага.
132 Ќесмешивающиес€ жидкости. ѕерегонка с вод€ным паром.
133 ‘азова€ диаграмма состо€ни€ трехкомпонентной системы с неограниченно смешивающимис€ компонентами. »спользование метода √иббса и –озебума дл€ определени€ состава системы.
134 ћол€рна€ теплота испарени€ воды равна 43,09 кƒж/моль. ƒавление насыщенного вод€ного пара при температуре 40 ∞— равно 55,3 мм рт.ст. „ему равно давление насыщенного пара при температуре 50 ∞—?
135 ƒавление паров над жидким галлием при температуре 1029 ∞— равно 0,01 мм рт.ст., а при 1154 ∞— - 0,1 мм рт.ст. „ему равна мол€рна€ теплота испарени€ галли€?
136 ћол€рна€ энтальпи€ плавлени€ льда равна 6,01 кƒж/моль, а энтальпи€ испарени€ воды 41,09 кƒж/моль. „ему равна мол€рна€ энтальпи€ возгонки льда?
137 ƒавление пара над серной кислотой при температуре 451 равно 666ѕа, а при температуре 484,5 - 2666 ѕа. ќпределите теплоту парообразовани€ серной кислоты, счита€ ее посто€нной в указанном интервале температуры.
138 «ависимость давлени€ (ѕа) насыщенного пара над жидким TiCl4 описываетс€ уравнением ln p = 19,688 Ц 3335/“. –ассчитайте энтальпию и энтропию парообразовани€ одного мол€ тетрахлорида титана при нормальной температуре кипени€.
139 ѕод давлением 0,1013 ћѕа лед плавитс€ при 273 . ”дельный объем льда при 273 равен 991,1 см3/кг, а воды Ц 916,6 см3/кг. ћол€рна€ теплота плавлени€ льда равна 6010 ƒж/моль. ¬ычислите давление, при котором лед будет плавитьс€ при 271 .
140 ѕлотности жидкого и твердого олова при температуре плавлени€ (231,9 ∞—) равны 6,980 г/см3 и 7,184 г/см3, соответственно. Ёнтальпи€ плавлени€ олова равна 1, 690 ккал/моль. ќпределите температуру олова под давлением 500 атм. ћол€рна€ масса олова равна 118,7 г/моль.
141 ѕри замерзании бензола (5,5 ∞—) его плотность измен€етс€ от 0,879г/см3 до 0, 891 г/см3. Ёнтальпи€ плавлени€ равна 10,59 кƒж/моль. ќпределите температуру плавлени€ бензола при давлении 1000 атм.
142 ѕлотности жидкой и твердой ртути при температуре плавлени€ (-38,87 ∞—) равны 13,690 и 14,193 г/см3, соответственно. Ёнтальпи€ плавлени€ ртути равна 2,33 кал/г. ќпределите температуру плавлени€ ртути при давлении 3000 атм.
143 “емпература кипени€ жидкого метанола равна 34,7 ∞— при давлении 200 “орр и 49,9 ∞— при давлении 400 “орр. Ќайдите температуру кипени€ метанола при нормальном давлении.
144 ƒавление пара диэтилового эфира при 10 ∞— равно 286,8 “орр, а при 20 ∞— Ц 432,8 “орр. ќпределите мольную энтальпию испарени€ и нормальную температуру кипени€ эфира.
145 ƒавление пара дихлорметана при 24,1 ∞— равно 400 “орр, а его энтальпи€ испарени€ равна 28,7 кƒж/моль. –ассчитайте температуру, при которой давление пара будет равно 500 “орр.
146 ƒавление пара твердого —ќ2 равно 133 ѕа при -134,3 ∞— и 2660 ѕа п






