1. ƒругий закон термодинам≥ки (перший закон термодинам≥ки, ентроп≥€ ≥ њњ властивост≥, в≥льна енерг≥€ √≥ббса тощо).
2. ѕри утворенн≥ 8,10 г купрум(≤≤) хлориду ≥з простих речовин (Cu i Cl2) вид≥л€Їтьс€ 13,4 кƒж тепла. ќбчислити ентальп≥ю утворенн€ CuCl2.
(¬ар≥анти: а) ¬изначити зм≥ну ентроп≥њ 5,85г NaCl при нагр≥ванн≥ в≥д 25 º— до 820 º—, €кщо температура його плавленн€ (tпл) 800 º—, мол€рна теплоЇмн≥сть до “пл становить 50,66 кƒж/мольЈ , а вище “пл - —р = 66,46 кƒж/мольЈ . “еплота плавленн€ 30,18 кƒж/моль; б) ≈нтальп≥€ згор€нн€ метанолу становить - 722 кƒж/моль. ¬изнач≥ть ентальп≥ю його утворенн€, €кщо 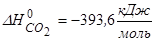 ,
,  )
)
3. ƒл€ реакц≥њ: CaCO3(к) = CaO(к) + CO2(г), користуючись даними дов≥дника:
- обчисл≥ть ∆Ќº ≥ ∆Sº х≥м≥чноњ реакц≥њ;
- зроб≥ть висновок про термодинам≥чну можлив≥сть чи неможлив≥сть ц≥Їњ реакц≥њ за стандартних умов ≥ при 2000 ;
- визнач≥ть р≥вноважну температуру цього процесу.
«ј¬ƒјЌЌя ƒЋя —јћќ—“≤…Ќќѓ –ќЅќ“»
«авданн€ ≤
ƒл€ наведених р≥вн€нь реакц≥й визнач≥ть двома способами стандартну енерг≥ю √≥ббса реакц≥њ. ÷€ реакц≥€ екзо- чи ендотерм≥чна? „и може в≥дбуватись ц€ реакц≥€ при стандартних умовах?
| ¬ | –≥вн€нн€ реакц≥њ |
| CO2(г) + H2 (г) = CO(г) + H2O(г) | |
| CO2(г) + 4 H2 (г) = CH4 (г) + 2 H2O (p) | |
| 4 NH3(г) + 3 O2(г) = 2N2(г) + 6H2O(p) | |
| 4 NH3(г) + 5 O2(г) = 4NO(г) + 6H2O | |
| 2SO2(г) + O2(г) = 2SO3(г) | |
| 2H2S(г) + 3 O2(г) = 2SO2(г) + 2H2O(г) | |
| CH4(г) + H2O(г) = CO(г) + 3H2(г) | |
| C2H5OH(p) = C2H4(г) + H2O(г) | |
| 2CO2(г) + 2 H2O(г) = C2H4(г) + 3O2(г) | |
| Ca(OH)2(k) + CO2(г) = CaCO3(k) + H2O(p) | |
| 2KCl(k) + 3O2(г) = 2KClO3(k) | |
| NH4NO3(k) = N2O(г) + 2H2O(г) | |
| 2SnO2(k) = 2SnO(k) + O2(г) | |
| 4FeO(k) + O2(г) = 2Fe2O3(k) | |
| Fe3O4(k) + H2(г) = 3FeO(k) + H2O(г) |
«авданн€ ≤≤
¬-1. ѕ≥д час в≥дновленн€ 19,88 г купрум(II) оксиду воднем до купрум(≤) оксиду вид≥л€Їтьс€ 12,63 кƒж.
¬изначте ентальп≥ю утворенн€ —u2ќ(к), €кщо:
∆Ќ∞утв. (—uO) = -156 кƒж/моль,
∆Ќ∞ утв. (Ќ2ќ) = -286 кƒж/моль.
¬-2. ¬изнач≥ть ступ≥нь розкладу —а—ќ3, €кщо при терм≥чному розклад≥ його наважки масою 20 г затрачаЇтьс€ 30 кƒж тепла.
∆Ќ0утв. (—аO) = -635 кƒж/моль;
∆Ќ0утв. (—а—O3) = -1206 кƒж/моль;
∆Ќ0утв. (—O2) = -393,5кƒж/моль.
¬-3. яка к≥льк≥сть тепла вид≥литьс€ внасл≥док згор€нн€
336 дм3 вод€ного газу (сум≥ш однакових об'Їм≥в Ќ2 ≥ —ќ), вз€того за нормальних умов?
(∆Ќ0утв. (Ќ2ќ) = -286 кƒж/моль;
∆Ќ0утв. (—ќ) = -110,6 кƒж/моль;
∆Ќ0утв. (—ќ2) = -393,8 кƒж/моль).
¬-4. ¬изначте ∆G∞298 реакц≥њ:
3 Niќ(к) + 2ј1(к) = 3 N≥(к) + ј12ќ3(к)
∆Gº298 (NiO) = -211,6 кƒж/моль,
∆Gº298 (ј12ќ3) = -1583,3 кƒж/моль.
¬-5. –еакц≥€ м≥ж хлором ≥ йодоводнем в≥дбуваЇтьс€ за р≥вн€нн€м:
—12 + 2Ќ≤ = ≤2 + 2Ќ—1
якщо в реакц≥њ використано 1 л —12 (за н.у.), то вид≥л€Їтьс€ 10,47 кƒж тепла. ќбчисл≥ть тепловий ефект реакц≥њ.
|
|
|
¬-6. ¬изначте зм≥ну ентроп≥њ внасл≥док плавленн€ 31,77 г м≥д≥. “емпература плавленн€ м≥д≥ дор≥внюЇ 1083 ∞—, теплота плавленн€ - 10 кƒж/моль.
¬-7. ¬изначте ∆Gº298 реакц≥њ:
2 —1ќ3(к) = 2 —1(к) + 3 ќ2(г)
∆Ќ∞утв.( —lO3)= -391,2 кƒж/моль; Sº( —lO3)=143,0 ƒж/мольЈ ; ∆Ќ∞утв. (KCl)= -435,9 кƒж/моль; Sº(KC1)=82,6 ƒж/мольЈ ;
Sº(O2) = 205,0 ƒж/мольЈ .
¬-8. –еакц≥€ гор≥нн€ метану в≥дбуваЇтьс€ за р≥вн€нн€м:
—Ќ4 + 2ќ2 = —ќ2 + 2 Ќ2ќ; ∆Ќ = -891,3 кƒж.
—к≥льки тепла вид≥литьс€ при згор€нн≥ 100 л метану за н.у.?
¬-9. ¬изначте ∆G∞298 реакц≥њ ≥ покаж≥ть, в €кому напр€мку можливий њхн≥й переб≥г за стандартних умов.
—а—ќ3(к) = —аќ(к) + —ќ2(г),
∆Ќºутв.(CaCO3)= -1207,7кƒж/моль; Sº(CaCO3)=91,6ƒж/мольЈ ;
∆Ќºутв.(—аќ)= -635,0 кƒж/моль; Sº(CaO) = 39,7 ƒж/мольЈ ; ∆Ќ∞утв.(—ќ2)= -393,8 кƒж/моль; Sº(CO2) = 213,8 ƒж/мольЈ .
¬-10. ѕитома теплота плавленн€ зал≥за р≥вна 269,2 ƒж/г. “емпература плавленн€ його р≥вна 1539∞ —. ¬изначити зм≥ну ентроп≥њ при плавленн≥ 2 мол≥в зал≥за.
¬-11. ¬изначити ступ≥нь розкладу ћg—ќ3, €кщо при терм≥чному розклад≥ наважки масою 8,4 г затрачено 8,8 кƒж тепла.
∆Ќ∞утв.(MgO) = -601,2 кƒж/моль;
∆Ќ∞утв.(MgCO3) = -1096 кƒж/моль;
∆Ќ∞утв.(CO2) = -393,5 кƒж/моль.
¬-12. яка реакц≥€ термодинам≥чно можлива за стандартних умов:
а) 2 —1(к) + ¬r2(г) = 2 ¬r(к) + —l2(г),
б) 2 ¬r(к) + —12(г) = 2 —1(к) + ¬г2(г).
∆G0298 ( —1) = - 408,0 кƒж/моль,
∆G0298 ( ¬г) = - 379,2 кƒж/моль.
¬-13. ¬изначте ∆G∞298 реакц≥њ ≥ покаж≥ть, в €кому напр€мку можливий њхн≥й переб≥г за стандартних умов:
—uќ(к) + —(гр) = —u(к) + —O(г)
€кщо ∆G0298 (CuO) = - 129,9кƒж/моль,
∆G0298 (CO) = -137,2 кƒж/моль.
¬-14. ѕри взаЇмод≥њ 13,49 г ј1 з надлишком с≥рки вид≥лилось 127,2 кƒж тепла. ¬изначте, ск≥льки тепла вид≥литьс€ при взаЇмод≥њ трьох моль алюм≥н≥ю з надлишком с≥рки.
¬-15. яку масу етену треба спалити, щоб нагр≥ти 200 г води в≥д 20 ∞— до 80 ∞—? (мольна теплоЇмн≥сть води
75,3 ƒж/мольЈ )
19 ≈Ћ≈ “–ќ’≤ћ≤„Ќ»… «ј’»—“ ћ≈“јЋ≤¬
¬≤ƒ ќ–ќ«≤ѓ
(лабораторна робота)
ѕрограмн≥ питанн€
1. ѕон€тт€ про електродний потенц≥ал. ѕодв≥йний електричний шар.
2. –≥вн€нн€ Ќернста.
3. —тандартний водневий електрод. –€д стандартних електродних потенц≥ал≥в.
4. √альван≥чний елемент. Ќапруга гальван≥чного елемента.
5. ороз≥€ метал≥в. ћехан≥зм електрох≥м≥чноњ короз≥њ.
6. «ахист метал≥в в≥д короз≥њ: метал≥чн≥ ≥ неметал≥чн≥ покритт€, електрох≥м≥чний захист, зм≥на властивостей короз≥йного середовища, легуванн€ метал≥в.
Ћ≥тература
[1], [5], [20], [22]






