‘изические свойства
ѕо физическим свойствам алкины напоминают алкены и алканы. “емпературы их плавлени€ и кипени€ увеличиваютс€ с ростом молекул€рной массы. ¬ обычных услови€х алкины —2Ц—3 Ц газы, —4Ц—16 Ц жидкости, высшие алкины Ц твердые вещества. Ќаличие тройной св€зи в цепи приводит к повышению температуры кипени€, плотности и растворимости их в воде по сравнению с олефинами и парафинами. ‘изические свойства некоторых алкинов сведены в таблице.
“аблица. ‘изические свойства некоторых алкинов
| Ќазвание | ‘ормула | t∞пл., ∞C | t∞кип., ∞C | d420 |
| јцетилен | HCЇCH | -80,8 | -83,6 | 0,565 1 |
| ћетилацетилен | CH3ЦCЇCH | -102,7 | -23,3 | 0,670 1 |
| Ѕутин-1 | C2H5ЦCЇCH | -122,5 | 8,5 | 0,678 2 |
| Ѕутин-2 | CH3ЦCЇCЦCH3 | -32,3 | 27,0 | 0,691 |
| ѕентин-1 | CH3ЦCH2ЦCH2ЦCЇCH | -98,0 | 39,7 | 0,691 |
| ѕентин-2 | CH3ЦCH2ЦCЇCЦCH3 | -101,0 | 56,1 | 0,710 |
| 3-ћетилбутин-1 | CH3ЦCHЦCЇCH I CH3 | Ц | 28,0 | 0,665 |
| 1 ѕри температуре кипени€. 2 ѕри 0∞C. |
’имические свойства
”глеродные атомы в молекуле ацетилена наход€тс€ в состо€нии sp-гибридизации. Ёто означает, что каждый атом углерода обладает двум€ гибридными sp- орбитал€ми, оси которых расположены на одной линии под углом 180∞ друг к другу, а две p- орбитали остаютс€ негибридными.

sp- √ибридные орбитали двух атомов углерода в состо€нии,
предшествующем образованию тройной св€зи и св€зей CЦH
ѕо одной из двух гибридных орбиталей каждого атома углерода взаимно перекрываютс€, привод€ к образованию s- св€зи между атомами углерода. ажда€ оставша€с€ гибридна€ орбиталь перекрываетс€ с s- орбиталью атома водорода, образу€ s- св€зь —ЦЌ.
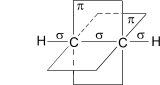
—хематическое изображение строени€ молекулы ацетилена (€дра атомов углерода и водорода на одной пр€мой,
две p- св€зи между атомами углерода наход€тс€ в двух взаимно перпендикул€рных плоскост€х)
ƒве негибридные p- орбитали каждого атома углерода, расположенные перпендикул€рно друг другу и перпендикул€рно направлению s- св€зей, взаимно перекрываютс€ и образуют две p- св€зи. “аким образом, тройна€ св€зь характеризуетс€ сочетанием одной s- и двух p- св€зей.
ƒл€ алкинов характерны все реакции присоединени€, свойственные алкенам, однако у них после присоединени€ первой молекулы реагента остаетс€ еще одна p- св€зь (алкин превращаетс€ в алкен), котора€ вновь может вступать в реакцию присоединени€ со второй молекулой реагента. роме того, "незамещенные" алкины про€вл€ют кислотные свойства, св€занные с отщеплением протона от атома углерода, составл€ющего тройную св€зь (Ї—ЦЌ).
I. –еакции присоединени€
1) √идрирование. √идрирование алкинов осуществл€етс€ при нагревании с теми же металлическими катализаторами (Ni, Pd или Pt), что и в случае алкенов, но с меньшей скоростью.
CH3ЦCЇCH(пропин) ЦЦt∞,Pd;H2ХЃ CH3ЦCH=CH2(пропен) ЦЦt∞,Pd;H2Ѓ CH3ЦCH2ЦCH3(пропан)
2) √алогенирование. јлкины обесцвечивают бромную воду (качественна€ реакци€ на тройную св€зь). –еакци€ галогенировани€ алкинов протекает медленнее, чем алкенов.
|
|
|
HCЇCH ЦЦBr2Ѓ CHBr=CHBr(1,2- дибромэтен) ЦЦBr2Ѓ CHBr2ЦCHBr2(1,1,2,2- тетрабромэтан)
3) √идрогалогенирование. ќбразующиес€ продукты определ€ютс€ правилом ћарковникова.
CH3ЦCЇCH ЦЦHBrЃ CH3ЦCBr=CH2(2- бромпропен -1) ЦЦHBrЃ CH3ЦCBr2ЦCH3(2,2- дибромпропан)
4) √идратаци€ (реакци€ учерова). ѕрисоединение воды осуществл€етс€ в присутствии сульфата ртути. Ёту реакцию открыл и исследовал в 1881 году ћ.√. учеров.
ѕрисоединение воды идет по правилу ћарковникова, образующийс€ при этом неустойчивый спирт с гидроксильной группой при двойной св€зи (так называемый, енол) изомеризуетс€ в более стабильное карбонильное соединение - кетон.
| C2H5ЦCЇCH + H2O ЦЦHgSO4Ѓ [C2H5Ц | C=CH2](енол) Ѓ C2H5Ц I ø OH | CЦCH3(метилэтилкетон) II O |
¬ случае гидратации собственно ацетилена конечным продуктом €вл€етс€ альдегид.
| O II | ||
HCЇCH + H2O ЦЦHgSO4Ѓ [CH2= 
| CH](енол) Ѓ CH3Ц I | C(уксусный альдегид) I |
| H | O | H |
5) ѕолимеризаци€. јцетиленовые углеводороды ввиду наличи€ тройной св€зи склонны к реакци€м полимеризации, которые могут протекать в нескольких направлени€х:
a) ѕод воздействием комплексных солей меди происходит димеризаци€ и линейна€ тримеризаци€ ацетилена.
HCЇCH ЦЦkat.HCЇCHЃ CH2=CHЦCЇCH ЦЦkat.HCЇCHЃ CH2=CHЦCЇCЦCH=CH2
b) ѕри нагревании ацетилена в присутствии активированного угл€ (реакци€ «елинского) осуществл€етс€ циклическа€ тримеризаци€ с образованием бензола.
| + | 
| ЦЦ600∞C,CЃ | 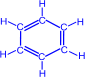
|
II. ислотные свойства
¬одородные атомы ацетилена способны замещатьс€ металлами с образованием ацетиленидов. “ак, при действии на ацетилен металлического натри€ или амида натри€ образуетс€ ацетиленид натри€.
HCЇCH ЦЦNaNH2Ѓ HCЇCNa + NH3
јцетилениды серебра и меди получают взаимодействием с аммиачными растворами соответственно оксида серебра и хлорида меди.
HCЇCH + 2[Ag(NH3)2]OH Ѓ AgCЇCAg + 4NH3 + 2H2O
јцетилениды серебра, меди обладают исключительной взрывчатостью. ќни легко разлагаютс€ при действии сол€ной кислоты.
AgCЇCAg + 2HCl Ѓ HCЇCH≠ + 2AgClѓ
ƒанным свойством ацетиленидов пользуютс€ при выделении ацетиленовых углеводородов из смесей с другими газами.
ѕрименение
ѕри горении ацетилена в кислороде температура пламени достигает 3150∞C, поэтому ацетилен используют дл€ резки и сварки металлов. роме того, ацетилен широко используетс€ в органическом синтезе разнообразных веществ - например, уксусной кислоты, 1,1,2,2- тетрахлорэтана и др. ќн €вл€етс€ одним из исходных веществ при производстве синтетических каучуков, поливинилхлорида и других полимеров.
јлкины Ч алифатические непредельные углеводороды, в молекулах которых между углеродными атомами имеетс€ одна тройна€ св€зь.
”глеводороды р€да ацетилена €вл€ютс€ еще более непредельными соединени€ми, чем соответствующие им алкены (с тем же числом углеродных атомов). Ёто видно из сравнени€ числа атомов водорода в р€ду:
—2Ќ6 C2H4 —2H2
<div style="height: 14px;overflow:hidden"><div style="margin-top: 14px;"><a href="http://chemistry.narod.ru/">ћир химии.</a></div></div>
Ётан этилен ацетилен
(этен) (этин)
|
|
|
јлкины образуют свой гомологический р€д с общей формулой, как и у диеновых углеводородов —nH2n-2
—“–ќ≈Ќ»≈ јЋ »Ќќ¬
ѕервым и основным представителем гомологического р€да алкинов €вл€етс€ ацетилен (этин) —2Ќ2. —троение его молекулы выражаетс€ формулами:
ЌЧ—Ї—ЧЌ или Ќ:—:::—:Ќ
—труктурна€ электронна€
‘ормула формула
ѕо названию первого представител€ этого р€да Ч ацетилена Ч эти непредельные углеводороды называют ацетиленовыми.
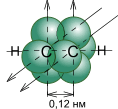
¬ алкинах атомы углерода наход€тс€ в третьем валентном состо€нии (sp -гибридизаци€). ¬ этом случае между углеродными атомами возникает тройна€ св€зь, состо€ща€ из одной s- и двух p-св€зей. ƒлина тройной св€зи равна 0,12 нм, а энерги€ ее образовани€ составл€ет 830 кƒж/моль. ћодели пространственного строени€ ацетилена представлены на рис. 1.
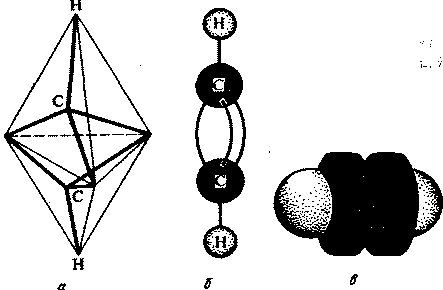
–ис.1. ћодели пространственного строени€ молекулы ацетилена:
а - тетраэдрическа€; б - шаростержнева€; в -по Ѕриглебу
Ќќћ≈Ќ Ћј“”–ј » »«ќћ≈–»я
Ќоменклатура. ѕо систематической номенклатуре ацетиленовые углеводороды называют, замен€€ в алканах суффикс -ан на суффикс -ин. ¬ состав главной цепи об€зательно включают тройную св€зь, котора€ определ€ет начало нумерации. ≈сли молекула содержит одновременно и двойную, и тройную св€зи, то предпочтение в нумерации отдают двойной св€зи:
—Ќ3
|
ЌЧ—Ї—Ч—Ќ2Ч—Ќ3 Ќ3—Ч—Ї—Ч—Ќ3 Ќ2—=—Ч—Ќ2Ч—Ї—Ќ
бутин-1 бутин-2 2-метилпентен-1-ин-4
(этилацетилен) (диметилацетилен)
ѕо рациональной номенклатуре алкиновые соединени€ называют, как производные ацетилена.
Ќепредельные (алкиновые) радикалы имеют тривиальные или систематические названи€:
ЌЧ—Ї—Ч - этинил;
Ќ—Ї—Ч—Ќ2Ч -пропаргил
»зомери€. »зомери€ алкиновых углеводородов (как и алкеновых) определ€етс€ строением цепи и положением в ней кратной (тройной) св€зи:
ЌЧ—Ї—Ч—ЌЧ—Ќ3 ЌЧ—Ї—Ч—Ќ2Ч—Ќ2Ч—Ќ3 Ќ3—Ч—=—Ч—Ќ2Ч—Ќ3
|
—Ќ3
3-метилбутин-1 пентин-1 пентин-2
4.3. ѕќЋ”„≈Ќ»≈ јЋ »Ќќ¬
јцетилен в промышленности и в лаборатории можно получать следующими способами:
1. ¬ысокотемпературным разложением (крекинг) природного газа Ч метана:
2—Ќ4 1500∞C Ѓ Ќ—Ї—Ќ + 3Ќ2
или этана:
—2Ќ6 1200∞C Ѓ Ќ—Ї—Ќ + 2Ќ2
2. –азложением водой карбида кальци€ —а—2, который получают спеканием негашеной извести —аќ с коксом:
—аќ + 3C 2500∞C Ѓ CaC2 + CO
—а—2 + 2Ќ2O Ѓ Ќ—Ї—Ќ + —а(ќЌ)2
3. ¬ лаборатории производные ацитилена можно синтезировать из дигалогенопроизводных, содержащих два атома галогена при одном или соседних углеродных атомах, действием спиртового раствора щелочи:
¬r
|
Ќ3—Ч—ЌЧ—ЌЧ—Ќ3 + 2 ќЌ Ѓ Ќ3—Ч—Ї—Ч—Ќ3 + 2KBr + 2Ќ2ќ
|
Br
2,3-дибромбутан бутин-2
(диметилацетилен)






