f Ё = ЧЧ (3) n∙z
где n - число функциональных групп в молекуле,
z - абсолютна€ величина зар€да функциональной группы.
‘ункциональными группами в кислотах €вл€ютс€ ионы водорода, в основани€х - ионы гидроксила, в сол€х - ионы металла. онечно, в кислых сол€х также ионы Ќ +, а в основных - ќЌ -, в зависимости от реакции. “аким образом:
дл€ Ќ 3 –ќ 4 а) f Ё = 1/3, б) Ё = 1/3{Ќ 3 –ќ 4 },
в) ћ Ё = f Ё ∙ћ(Ќ 3 –ќ 4) = 1/3(3,0 + 31,0 + 4∙16,0) = 98,0/3 = 32,7 г/моль
дл€ —а(ќЌ) 2 а) f Ё = 1/2, б) Ё = 1/2{—а(ќЌ) 2 },
в) ћ Ё = f Ё ∙ћ(—а(ќЌ) 2) = 1/2(40,1 + 2∙17,0) = 74,1/2 = 37,0 г/моль.
дл€ јl 2 (Sќ 4) 3 а) f Ё = 1/(2∙3) = 1/6, б) Ё = 1/6{јl 2 (Sќ 4) 3 },
в) ћ Ё = f Ё ∙ћ(јl 2 (Sќ 4) 3) = 1/6(2∙27,0 + 3∙96,0) = 342/6 = 57,0 г/моль
ќксиды дел€тс€ на солеобразующие (кислотные, амфотерные, основные) и несолеобразующие. ƒл€ несолеобразующих (безразличных) оксидов —ќ, N 2 ќ, Nќ характерны окислительно-восстановительные реакции, (ќ¬–). ¬ ќ¬– эквивалент всегда рассчитываетс€ по изменению степени окислени€.
ƒл€ солеобразующих оксидов в реакци€х не ќ¬– фактор эквивалентности определ€етс€ по формуле (3) дл€ кислот (оснований), ангидридом которых €вл€етс€ данный оксид.
ѕример 2. ќпределить в реакци€х присоединени€/разложени€, не €вл€ющихс€ ќ¬–, а) фактор эквивалентности, f Ё; б) химическую формулу эквивалента, Ё; в) мол€рную массу эквивалентов, ћ Ё, а дл€ газов и г)объем мол€ эквивалентов, V Ё, при максимальном содержании эквивалентов в молекуле дл€ следующих веществ из класса оксидов. —ќ 2 - газ, —аќ, – 2 ќ 5.
–ешение: —ќ 2 - кислотный оксид, €вл€етс€ ангидридом двухосновной угольной кислоты Ќ 2 —ќ 3, в соответствии с чем его фактор эквивалентности составл€ет 1/2.
а) f Ё = 1/2; б) Ё = 1/2 {—ќ 2 }, в) ћ Ё = f Ё ∙ћ—ќ 2 = 1/2 (12,0 + 2∙16,0) = 44,0/2 = 22,0 г/моль. “ак как —ќ 2 - газ, определ€ем еще объем мол€ эквивалентов (эквивалентный объем): г) V Ё = f Ё ∙22,4 = 11,2 л.
—аќ - основной оксид, €вл€ющийс€ ангидридом двухкис-лотного основани€ —а(ќЌ) 2, в соответствии с чем его фактор эквивалентности составл€ет 1/2.
а) f Ё = 1/2, б) Ё = 1/2{—аќ}, в) ћ Ё = f Ё ∙ћ—аќ = 1/2(40,1 + 16,0) = 56,1/2 = 28,0 г/моль.
– 2 ќ 5 - кислотный оксид, дающий при взаимодействии с водой две молекулы трехосновной фосфорной кислоты Ќ 3 –ќ 4. по реакции:
–2ќ5 + 3Ќ2ќ = 2Ќ3–ќ4
—ледовательно, одна молекула – 2 ќ 5 эквивалентна 6 ионам водорода, в соответствии с чем, его фактор эквивалентности находитс€ из формулы f Ё = 1/(2∙3) и составл€ет 1/6.
а) f Ё = 1/6, б) Ё = 1/6 {– 2 ќ 5 },
в) ћ Ё = f Ё ∙ћ– 2 ќ 5 = 1/6(2∙31,0 + 5∙16,0) = 142/6 = 23,67 г/моль
≈сли дана конкретна€ реакци€, то состав эквивалента следует определ€ть из сопоставлени€ начальных и конечных продуктов реакции.
¬ первую очередь следует определить, с каким типом реакции мы имеем дело: с окислительно-восстановительной реакцией (ќ¬–) или с не-ќ¬–. последним относ€тс€ реакции, в которых не мен€ютс€ степени окислени€ элементов, например, ионнообменные реакции и часть реакций разложени€. »з определени€ эквивалента следует, что в зависимости от типа реакции, по разному определ€етс€ состав эквивалента вещества. ¬ ионообменных реакци€х (не-ќ¬–) надо рассматривать, сколько ионов водорода или эквивалентных ему частиц (Nа +, +, ќЌ -, —l - и т.д.) взаимодействует с рассматриваемым веществом. Ќапоминаем, что окислительно-восстановительными реакци€ми (ќ¬–), €вл€ютс€ такие, в которых измен€ютс€ степени окислени€ (—.ќ.) элементов.
|
|
|
¬ ќ¬– дл€ того, чтобы определить фактор эквивалентности и правильно записать химическую формулу эквивалента, надо определить —.ќ. окисл€ющегос€ или восстанавливающегос€ элемента в данном веществе до и после реакции и определить число электронов, перемещаемых в оболочке этого элемента. —огласно определению, эквивалент составит такую часть молекулы, котора€ приходитс€ на 1 электрон. Ќикакого учета коэффициентов в реакци€х при этом не требуетс€.
ѕример 3. ќпределить: фактор эквивалентности, f Ё, химическую формулу эквивалента, Ё, мол€рную массу эквивалентов, ћ Ё, и (дл€ газов) мол€рный объем эквивалентов, V Ё, реагирующих веществ в следующих реакци€х:
1. јl(ќЌ)3 + 2Ќ—l = јlќЌ—l2 + 2Ќ2ќ
2. 2Ќ2S(√) + 3ќ2(√) = 2Sќ2 + 2Ќ2ќ
–ешение. –еакци€ 1 €вл€етс€ ионообменной ¬ ней јl(ќЌ) 3 превращаетс€ в јlќЌ—l 2, т.е. в молекуле гидроксида алюмини€ замещаютс€ два иона ќЌ -, каждый из которых эквивалентен одному иону водорода, на ионы —l -. —ледовательно, ее эквивалент в данной конкретной реакции составл€ет 1/2 молекулы јl(ќЌ) 3. f Ё =1/2; Ё= 1/2{јl(ќЌ) 3 }; ћ Ё = f Ё ∙ћјl(ќЌ)3 = 1/2(27,0 + 3∙17,0) = 39 г/моль.
ћолекула Ќ—l в любой ионообменной реакции может отдавать только 1 ион водорода Ќ +, следовательно, содержит 1 эквивалент. fЁ=1, Ё = {Ќ—l}. ћ Ё = fЁ ∙ ћ Ќ—l = 1∙(1,0 + 35,5) = 36,5 г/моль.
–еакци€ 2 окислительно-восстановительна€. ¬ ней сера мен€ет свою —.ќ. от -2 (в Ќ 2 S) до +4 (в Sќ 2). ѕеремещаютс€ 6 электронов. —ледовательно, в данной конкретной реакции молекула сероводорода содержит 6 эквивалентов. f Ё = 1/6, Ё = 1/6 {Ќ 2 S} ћ Ё = f Ё ∙ ћЌ 2 S = 1/6(2,0 + 32,1) = 5,7 г/моль. —ероводород - газ. V Ё = fЁ ∙22,4 = 3,73 л.
ислород в реакции 2 мен€ет свою —.ќ. от 0 до -2. ѕри этом у каждого атома кислорода перемещаютс€ 2 электрона. ¬ молекуле кислорода ќ 2 неразрывно св€заны 2 атома. —ледовательно, молекула кислорода содержит 4 эквивалента. f Ё = 1/4, Ё = ¼{ ќ 2 } ћ Ё = fЁ ∙ћќ2 = 1/4(2∙16) = 8 г/моль. ислород - газ. V Ё = fЁ ∙22,4 = 5,6 л.
»так, обобща€ вышеизложенный материал, расчет фактора эквивалентности дл€ некоторых классов химических соединений можно представить в виде таблицы 1.
“аблица 1 - –асчет фактора эквивалентности
| „астица | ‘актор эквивалентности | ѕримеры |
| ѕростое вещество |  ,где n (Ё) Ц число атомов элемента (индекс в химической формуле), ¬ (Ё) Ц валентность элемента ,где n (Ё) Ц число атомов элемента (индекс в химической формуле), ¬ (Ё) Ц валентность элемента
| f Ё(H2) = 1/(2×1) = 1/2; f Ё(O2) = 1/(2×2) = 1/4; f Ё(Cl2)= 1/(2×1) = 1/2; f Ё(O3) = 1/(3×2) = 1/6 |
| ќксид |  ,где n (Ё) Ц число атомов элемента (индекс в химической формуле оксида), ¬ (Ё) Ц валентность элемента ,где n (Ё) Ц число атомов элемента (индекс в химической формуле оксида), ¬ (Ё) Ц валентность элемента
| f Ё(Cr2O3)=1/(2×3)= 1/6; f Ё(CrO) = 1/(1×2) = 1/2; f Ё(H2O) = 1/(2×1) = 1/2; f Ё(P2O5)=1/(2×5) = 1/10 |
| ислота |  ,где n (H+) Ц число ионов водорода (основность кислоты) ,где n (H+) Ц число ионов водорода (основность кислоты)
| f Ё (HCl) = 1/1 = 1 f Ё (H2SO4) = 1/2 f Ё (H3PO4) = 1/3 |
| ќснование |  ,где n (ќHЦ) Ц число гидроксид-ионов (кислотность основани€) ,где n (ќHЦ) Ц число гидроксид-ионов (кислотность основани€)
| f Ё (KOH) = 1 f Ё (Cu(OH)2) = 1/2 |
| —оль |  ,где n (ће) Ц число атомов металла (индекс в химической формуле соли), ¬ (ће) Ц валентность металла; n (ј) Ц число кислотных остатков, ¬ (ј) Ц валентность кислотного остатка ,где n (ће) Ц число атомов металла (индекс в химической формуле соли), ¬ (ће) Ц валентность металла; n (ј) Ц число кислотных остатков, ¬ (ј) Ц валентность кислотного остатка
| f Ё(Cr2(SO4)3) = 1/(2 × 3) = 1/6 (расчет по металлу) или f Ё(Cr2(SO4)3) = 1/(3 × 2) = 1/6 (расчет по кислотному остатку)f Ё (ZnCl2) = 1/(1×2) =1/2 (расчет по металлу) f Ё (NaCl) = 1/(1 × 1) =1/2 (расчет по металлу) |
| „астица в окислительно-восстано≠вительных реакци€х | 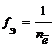 ,где ,где  Ц число электронов, участвующих в процессе окислени€ или восстановлени€ Ц число электронов, участвующих в процессе окислени€ или восстановлени€
| Fe2+ + 2  → Fe0 f Ё(Fe2+) =1/2;MnO4Ц+8H++ 5 =Mn2+ + 4H2O f Ё(MnO4Ц) = 1/5 → Fe0 f Ё(Fe2+) =1/2;MnO4Ц+8H++ 5 =Mn2+ + 4H2O f Ё(MnO4Ц) = 1/5
|
| »он |  ,где z Ц зар€д иона ,где z Ц зар€д иона
| f Ё(SO42Ц) = 1/2 |
|
|
|
«акон эквивалентов
«акон эквивалентов был сформулирован в 1800 г так: '' все вещества реагируют в эквивалентных отношени€х''. —овременное определение закона эквивалентов гласит:






