–еакции, в ходе которых элементы, вход€щие в состав реагирующих веществ, измен€ют степень окислени€, называютс€ окислительно Ц восстановительными (ќ¬–).
—тепень окислени€. ƒл€ характеристики состо€ни€ элементов в соединени€х введено пон€тие степени окислени€. —тепень окислени€ (с.о.) Ц это условный зар€д, который приписываетс€ атому в предположении, что все св€зи в молекуле или ионе предельно пол€ризованы. —тепень окислени€ элемента в составе молекулы вещества или иона определ€етс€ как число электронов, смещенных от атома данного элемента (положительна€ степень окислени€) или к атому данного элемента (отрицательна€ степень окислени€). ƒл€ вычислени€ степени окислени€ элемента в соединении следует исходить из следующих положений (правил):
1. —тепень окислени€ элементов в простых веществах, в металлах в элементном состо€нии, в соединени€х с непол€рными св€з€ми равны нулю.ѕримерами таких соединений €вл€ютс€ N20, Ќ20, —l20, I20, ћg0, Fe0 и т.д.††
2. ¬ сложных веществах отрицательную степень окислени€ имеют элементы с большей электроотрицательностью.
ѕоскольку при образовании химической св€зи электроны смещаютс€ к атомам более электроотрицательных элементов, то последние имеют в соединени€х отрицательную степень окислени€.
+1 †+7 †-2
HClO4†††††††††††††† †††††††††††††
 ††††† ќ-2†††
††††† ќ-2†††
ќ-2† Cl  ќ-2 †††††††††Ќ+†† Ёлемент †††††††††Ёќ†
ќ-2 †††††††††Ќ+†† Ёлемент †††††††††Ёќ†
†††††† ќ-2††††††††††††††††††††††††††††††††††† Ќ†††††††† 2,1
††††††††††††††††††††††††††††††††††††††††† † Cl††††††† 3,0††††††††
††††††††††††††††††††††††††††††††††††††††† †† ќ†† 3,5
¬ некоторых случа€х степень окислени€ элемента численно совпадает с валентностью (¬) элемента в данном соединении, как, например, в ЌClќ4.
ѕриведенные ниже примеры показывают, что степень окислени€ и валентность элемента могут численно различатьс€:
N ≡ N†††††††††††††††††††††† ¬ (N)=3; с.о.(N)=0
†††  †Ќ+†††††††††††††††††††† ††††††
†Ќ+†††††††††††††††††††† ††††††
Ќ+† C-2†† ќ-2†† †Ќ+
†††† ††Ќ+
Ёќ (C) = 2,5†††††††† ¬(—) = 4††††††††††† с.о.(—) = -2
Ёќ (ќ) = 3,5† ¬(ќ) = 2††††††††††††† с.о.(ќ) = -2
Ёќ (Ќ) = 2,1† ¬(Ќ) = 1††††††††††††† с.о.(Ќ) = +1
3. –азличают высшую, низшую и промежуточные степени окислени€.
¬ысша€ степень окислени€ Ц это ее наибольшее положительное значение. ¬ысша€ степень окислени€, как правило, равна номеру группы (N) периодической системы, в которой элемент находитс€. Ќапример, дл€ элементов III периода она равна: Na+2, Mg+2, AI+3, Si+4, P+5, S+6, CI+7. »сключение составл€ют фтор, кислород, гелий, неон, аргон, а также элементы подгруппы кобальта и никел€: их высша€ степень окислени€ выражаетс€ числом, значение которого ниже, чем номер группы, к которой они относ€тс€. ” элементов подгруппы меди, наоборот, высша€ степень окислени€ больше единицы, хот€ они и относ€тс€ к I группе.
Ќизша€ степень окислени€ определ€етс€ количеством электронов, не достающих до устойчивого состо€ни€ атома ns2nр6. Ќизша€ степень окислени€ дл€ неметаллов равна (N-8), где N Ц номер группы периодической системы, в которой элемент находитс€. Ќапример, дл€ неметаллов III периода она равна: Si-4, P-3, S-2,CI ˉ. Ќизша€ степень окислени€ дл€ металлов Ц это наименьшее ее положительное значение из возможных. Ќапример, марганец имеет следующие степени окислени€: Mn+2, Mn+4, Mn+6, Mn+7; с.о.=+2 Ц это низша€ степень окислени€ дл€ марганца.
|
|
|
¬се остальные встречающиес€ степени окислени€ элемента называют промежуточными. Ќапример, дл€ серы степень окислени€, равна€ +4, €вл€етс€ промежуточной.
4. –€д элементов про€вл€ют в сложных соединени€х посто€нную степень окислени€:
а) щелочные металлы Ц (+1);
б) металлы второй группы обеих подгрупп (за исключением Ќg) Ц (+2); ртуть может про€вл€ть степени окислени€ (+1) и (+2);
в) металлы третьей группы, главной подгруппы Ц (+3), за исключением Tl, который может про€вл€ть степени окислени€ (+1) и (+3);
г) F-
†††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††† ††††††††††††††††††††††††††††+1 -1 ††††+2 -1
д) H+, кроме гидридов металлов (NaH, CaH2 и т.д.), где его степень окислени€ равна (-1);
†††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††† ††††††††††††††††††††††††††††††††††+1 -1 †††††††+2 -1
е) ќ-2, за исключением пероксидов элементов (Ќ2ќ2, —аќ2 и т.д.), где степень окислени€ кислорода равна (-1), надпероксидов элементов
††+1 -1/2 ††††††††+1 -1/2
( ќ2, NaO2 и т.д.), в которых его степень окислени€ равна Ц ½, фторида
††††††††††††††††††††††††††††† ††††††††+2 -1
кислорода ќF2.
5. Ѕольшинство элементов могут про€вл€ть разную степень окислени€ в соединени€х. ѕри определении их степени окислени€ пользуютс€ правилом, согласно которому сумма степеней окислени€ элементов в электронейтральных молекулах равна нулю, а в сложных ионах Ц зар€ду этих ионов.
¬ качестве примера вычислим степень окислени€ фосфора в ортофосфорной кислоте Ќ3–ќ4. —умма всех степеней окислени€ в соединении должна быть равна нулю, поэтому обозначим степень окислени€ фосфора через ’ и, умножив известные степени окислени€ водорода (+1) и кислорода (-2) на число их атомов в соединении, составим уравнение: (+1)*3+’+(-2)*4 = 0, из которого ’ = +5.
¬ычислим степень окислени€ хрома в дихромат Ц ионе (Cr2ќ7)2-.
—умма всех степеней окислени€ в сложном ионе должна быть равна (-2), поэтому обозначим степень окислени€ хрома через ’, составим уравнение 2’ +(-2)*7 = -2, из которого ’ = +6.
ѕон€тие степени окислени€ дл€ большинства соединений имеет условный характер, т.к. не отражает реальный эффективный зар€д атома. ¬ простых ионных соединени€х степень окислени€ вход€щих в них элементов равна электрическому зар€ду, поскольку при образовании этих соединений происходит практически полный переход электронов от одного
†††††††††††††††††††††††††††††† ††††††+1 -1 †††+2 -1† ††+3 -1
†атома к другому: NaI,MgCI2, AIF3. ƒл€ соединени€ с пол€рной ковалентной св€зью фактический эффективный зар€д меньше степени окислени€, однако это пон€тие весьма широко используетс€ в химии.
ќсновные положени€ теории ќ¬–:
1. ќкислением называют процесс отдачи электронов атомом, молекулой или ионом. „астицы, отдающие электроны, называют восстановител€ми; во врем€ реакции они окисл€ютс€, образу€ продукт окислени€. ѕри этом элементы, участвующие в окислении, повышают свою степень окислени€. Ќапример:
AI Ц 3e- Ѓ AI3+
|
|
|
H2 Ц 2e- Ѓ 2H+
Fe 2+ - e- Ѓ Fe3+
2. ¬осстановлением называют процесс присоединени€ электронов атомом, молекулой или ионом. „астицы, присоедин€ющие электроны, называют окислител€ми; во врем€ реакции они восстанавливаютс€, образу€ продукт восстановлени€. ѕри этом элементы, участвующие в восстановлении, понижают свою степень окислени€. Ќапример:
S + 2e- Ѓ S2-
CI2 + 2e- Ѓ 2 CI ˉ
Fe3+ + e- Ѓ Fe 2+
3.¬ещества, содержащие частицы восстановители или окислители, соответственно называют восстановител€ми или окислител€ми. Ќапример, FeCI2 €вл€етс€ восстановителем за счет Fe2+, а FeCI3 - †окислителем за счет Fe3+.
4. ќкисление всегда сопровождаетс€ восстановлением и, наоборот, восстановление всегда св€зано с окислением. “аким образом ќ¬– представл€ют собой единство двух противоположенных процессов Ц окислени€ и восстановлен舆††
5. „исло электронов, отданных восстановителем, равно числу электронов, прин€тых окислителем.
—оставление уравнений окислительно-восстановительных реакций. Ќа последнем правиле базируютс€ два метода составлени€ уравнений дл€ ќ¬–:
1. ћетод электронного баланса.
«десь подсчет числа присоедин€емых и тер€емых электронов производитс€ на основании значений степеней окислени€ элементов до и после реакции. ќбратимс€ к простейшему примеру:
Na0 + Cl  Ѓ Na+ Cl
Ѓ Na+ Cl 
 |  |
|
1 ††††††††† Cl2 + 2eˉ Ѓ 2 Cl  †- восстановление
†- восстановление
 ††††††††††††††††2 Na + Cl2 = 2Na+ + 2Cl
††††††††††††††††2 Na + Cl2 = 2Na+ + 2Cl 
†††††††††††††† 2 Na + Cl2 = 2NaCl
ƒанный метод используют в том случае, если реакци€ протекает не в растворе (в газовой фазе, реакции термического разложени€ и т.д.).
2. ћетод ионно-электронный (метод полуреакций).
ƒанный метод учитывает среду раствора, дает представление о характере частиц реально существующих и взаимодействующих в растворах. ќстановимс€ на нем более подробно.
јлгоритм подбора коэффициентов ионно-электронным методом:
1. —оставить молекул€рную схему реакции с указанием исходных веществ и продуктов реакции.
2. —оставить полную ионно-молекул€рную схему реакции, записыва€ слабые электролиты, малорастворимые, нерастворимые и газообразные вещества в молекул€рном виде, а сильные электролиты Ц в ионном.
3. »сключив из ионно-молекул€рной схемы ионы, не измен€ющиес€ в результате реакции (без учета их количества), переписать схему в кратком ионно-молекул€рном виде.
4. ќтметить элементы, измен€ющие в результате реакции степень окислени€; найти окислитель, восстановитель, продукты восстановлени€, окислени€.
5. —оставить схемы полуреакций окислени€ и восстановлени€, дл€ этого:
а) указать восстановитель и продукт окислени€, окислитель и продукт восстановлени€;
б) уравн€ть число атомов каждого элемента в левой и правой част€х полуреакций (выполнить баланс по элементам) в последовательности: элемент, измен€ющий степень окислени€, кислород, другие элементы; при этом следует помнить, что в водных растворах в реакци€х могут участвовать молекулы Ќ2ќ, ионы Ќ+ или ќЌ Ц в зависимости от характера среды:
| ѕроцесс | исла€ среда | Ќейтральна€ среда | ўелочна€ среда |
| —в€зывание избытка кислорода | ќ-2+2Ќ+ =Ќ2ќ | ќ-2+Ќ2ќ=2ќЌ ˉ | ќ-2+Ќ2ќ=2ќЌ ˉ |
| ¬осполнение недостатка кислорода | Ќ2ќ= ќ-2+2Ќ+ | Ќ2ќ = ќ-2+2Ќ+ | 2ќЌ ˉ=ќ-2+Ќ2ќ |
в) уравн€ть суммарное число зар€дов в обеих част€х полуреакций; дл€ этого прибавить или отн€ть в левой части полуреакций необходимое число электронов (баланс по зар€дам).
6. Ќайти наименьшее общее кратное (Ќќ ) дл€ числа отданных и полученных электронов.
7. Ќайти основные коэффициенты при каждой полуреакции. ƒл€ этого полученное в п.6 число (Ќќ ) разделить на число электронов, фигурирующих в данной полуреакции.
8. ”множить полуреакции на полученные основные коэффициенты, сложить их между собой: левую часть с левой, правую Ц с правой (получить ионно-молекул€рное уравнение реакции). ѕри необходимости Упривести подобныеФ ионы с учетом взаимодействи€ между ионами водорода и гидроксид-ионами: H++OH ˉ= H2O.
|
|
|
9. –асставить коэффициенты в молекул€рном уравнении реакции.
10. ѕровести проверку по частицам, не участвующим в ќ¬–, исключенным из полной ионно-молекул€рной схемы (п.3). ѕри необходимости коэффициенты дл€ них наход€т подбором.
11. ѕровести окончательную проверку по кислороду.
ѕримеры:
1. исла€ среда.
ћолекул€рна€ схема реакции:
KMnO4 + NaNO2 + H2SO4 Ѓ MnSO4 + NaNO3 + H2O + K2SO4
ѕолна€ ионно-молекул€рна€ схема реакции:
K++MnO 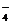 + Na++NO
+ Na++NO  +2H++ SO
+2H++ SO 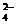 †Ѓ Mn2+ + SO
†Ѓ Mn2+ + SO 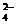 + Na+ + NO
+ Na+ + NO 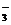 + H2O + 2K+ +SO
+ H2O + 2K+ +SO 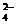 .
.
ратка€ ионно-молекул€рна€ схема реакции:
+7†††††† +3†††††††††††††††††††††††††††††† ††††+5
MnO 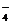 +NO
+NO  +2H+ Ѓ Mn2+ + NO
+2H+ Ѓ Mn2+ + NO 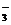 †+ H2O ††††††††††††††††††††††††††††††††††††††††††††††
†+ H2O ††††††††††††††††††††††††††††††††††††††††††††††
ок-ль†††† в-ль†††† †††продукт в-ни€ продукт ок-и€
¬ ходе реакции степень окислени€ Mn понижаетс€ от +7 до +2 (марганец восстанавливаетс€), следовательно, Mnќ 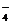 †Ц окислитель; Mn2+ - продукт восстановлени€. —тепень окислени€ азота повышаетс€ от +3 до +5 (азот окисл€етс€), следовательно, NO
†Ц окислитель; Mn2+ - продукт восстановлени€. —тепень окислени€ азота повышаетс€ от +3 до +5 (азот окисл€етс€), следовательно, NO  Ц восстановитель, NO
Ц восстановитель, NO 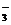 †Ц продукт окислени€.
†Ц продукт окислени€.
”равнени€ полуреакций:

 2††††††† MnO
2††††††† MnO 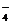 †+ 8 H + + 5e- Ѓ Mn2+ + 4 H2 O - процесс восстановлен舆††††
†+ 8 H + + 5e- Ѓ Mn2+ + 4 H2 O - процесс восстановлен舆††††
10†††††††† +7†††† +(-5) =††† +2
5††††† NO  †+ H2 O Ц 2e- Ѓ NO
†+ H2 O Ц 2e- Ѓ NO 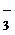 †+ 2 H + - процесс окислен舆†
†+ 2 H + - процесс окислен舆†
†††††††††††††††††† -1 -(-2)††††††††††† =†††† +1
 2MnO
2MnO 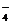 + 16H+ + 5NO
+ 16H+ + 5NO  + 5H2O = 2Mn2+ +8H2O + 5NO
+ 5H2O = 2Mn2+ +8H2O + 5NO 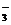 †+ 1OH+ (полное ионно-молекул€рное уравнение).
†+ 1OH+ (полное ионно-молекул€рное уравнение).
¬ суммарном уравнении исключаем число одинаковых частиц, наход€щихс€ как в левой, так и в правой част€х равенства (приводим подобные). ¬ данном случае это ионы Ќ+ и Ќ2ќ.
раткое ионно-молекул€рное уравнение будет иметь вид
2MnO 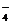 †+ 6H+ + 5NO
†+ 6H+ + 5NO  †Ѓ 2Mn2+ + 3H2O + 5NO
†Ѓ 2Mn2+ + 3H2O + 5NO 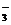 .
.
¬ молекул€рной форме уравнение имеет вид
2KMnO4 + 5 NaNO2 + 3 H2SO4 = 2MnSO4+5NaNO3 + 3H2O + K2SO4.
ѕроверим баланс по частицам, которые не участвовали в ќ¬–:
K+ (2 = 2), Na+ (5 = 5), SO 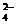 †(3 = 3). Ѕаланс по кислороду: 30 = 30.
†(3 = 3). Ѕаланс по кислороду: 30 = 30.
2. Ќейтральна€ среда.
ћолекул€рна€ схема реакции:
KMnO4 + NaNO2 + H2O Ѓ MnO2 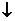 †+ NaNO3 + KOH
†+ NaNO3 + KOH
»онно-молекул€рна€ схема реакции:
K+ + MnO 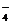 †+ Na+ + NO
†+ Na+ + NO  †+ H2O Ѓ MnO2
†+ H2O Ѓ MnO2 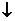 †+ Na+ + NO
†+ Na+ + NO 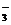 + K+ + OH
+ K+ + OH 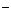
ратка€ ионно-молекул€рна€ схема:
†+7†††††††††† +3†††††††††††††††††††††††††††††††††††† +4†††††††††††† +5
MnO 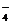 †+ NO
†+ NO  †+ H2O Ѓ† MnO2
†+ H2O Ѓ† MnO2 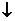 †+ NO
†+ NO 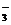 †+ OH-
†+ OH-
ок-ль ††††††††††††в-ль††††††††††††††††††††† † продукт в-ни€ продукт ок-и€

 ”равнени€ полуреакций:
”равнени€ полуреакций:
 2† MnO
2† MnO 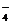 †+ 2H2O + 3eˉ †††Ѓ ††††MnO2
†+ 2H2O + 3eˉ †††Ѓ ††††MnO2 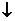 +4OH
+4OH 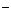 -процесс восстановлен舆†††††††††
-процесс восстановлен舆†††††††††
 6 †-1†† +(-3) =†††††††††† †††† -4
6 †-1†† +(-3) =†††††††††† †††† -4
3††††† NO  †+ H2O Ц 2eˉ †††††††Ѓ †††††††††††NO
†+ H2O Ц 2eˉ †††††††Ѓ †††††††††††NO 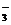 †+ 2H+ - процесс окислени€
†+ 2H+ - процесс окислени€


 ††††††††††††††††† -1††† - (-2) †=††††††††††††† +1
††††††††††††††††† -1††† - (-2) †=††††††††††††† +1
2MnO 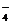 †+ 4H2O + 3NO
†+ 4H2O + 3NO  †+ 3 H2O Ѓ 2MnO2
†+ 3 H2O Ѓ 2MnO2 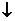
 †+ 8 OH- + 3NO
†+ 8 OH- + 3NO 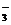 †+ 6H+.
†+ 6H+.
ѕриводим подобные, учитыва€:
8 ќЌ 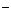
 + 6Ќ+ = 6Ќ2ќ + 2ќЌ
+ 6Ќ+ = 6Ќ2ќ + 2ќЌ 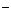 .
.
раткое ионно-молекул€рное уравнение:
2MnO 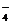 †+ 3NO
†+ 3NO  †+ H2O Ѓ 2MnO2
†+ H2O Ѓ 2MnO2 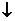 + 3 NO
+ 3 NO 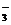 †+ 2OH
†+ 2OH 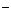 .
.
¬ молекул€рной форме уравнение имеет вид
2KMnO4 + 3NaNO2 + H2O Ѓ 2MnO2 + 3NaNO3 + 2KOH.
Ѕаланс по частицам, которые не участвовали в ќ¬–: Na+ (3 = 3); ††††K+ (2 = 2). Ѕаланс по кислороду: 15 = 15.
3. ўелочна€ среда.
ћолекул€рна€ схема реакции:
KMnO4 + NaNO2 + KOH Ѓ K2MnO4 + NaNO3 + H2O.
»онно-молекул€рна€ схема реакции:
K++MnO 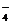 †+ Na++NO
†+ Na++NO  †+ K++ OH
†+ K++ OH 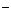 Ѓ 2K++MnO
Ѓ 2K++MnO 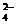 †+ Na++NO
†+ Na++NO 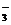 †+ H2O.
†+ H2O.
ратка€ ионно-молекул€рна€ схема реакции:
†+7†††††††††† +3†††††††††††††††††††††††††† +6†††††††††††† +5
MnO 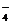 †+ NO
†+ NO  †+OH
†+OH 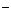 Ѓ MnO
Ѓ MnO 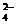 †+ NO
†+ NO 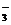 †+ H2O
†+ H2O
oк-ль††††††††††††††† в-ль†† †††† †продукт в-ни€ продукт ок-и€
”равнени€ полуреакций:

 2†††† MnO
2†††† MnO 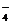 †+ 1e- Ѓ MnO
†+ 1e- Ѓ MnO 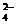 †Ц процесс восстановлен舆†††††
†Ц процесс восстановлен舆†††††

 1†††† NO
1†††† NO  †+ 2OH
†+ 2OH 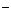 †- 2e- Ѓ ††††††NO
†- 2e- Ѓ ††††††NO 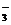 †+ H2O Ц процесс окислен舆†††
†+ H2O Ц процесс окислен舆†††
 †††††††††† -3†††††† - (-2) =††††††† -1
†††††††††† -3†††††† - (-2) =††††††† -1
|
|
|
2 MnO 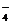 †+ NO
†+ NO  †+2OH
†+2OH 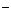 Ѓ 2MnO
Ѓ 2MnO 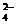 †+ NO
†+ NO 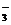 †+ H2O (краткое ионно-молекул€рное уравнение).
†+ H2O (краткое ионно-молекул€рное уравнение).
¬ молекул€рной форме уравнение имеет вид
2KMnO4 + NaNO2 + 2KOH Ѓ 2K2MnO4 + NaNO3 + H2O
Ѕаланс по частицам, которые не участвовали в ќ¬–: + (4 = 4); Na+ (1 = 1). Ѕаланс по кислороду: 12 = 12.






