Ћабораторна€ работа є 1.
Ёлектролиз растворов.
÷ель работы: »зучить электродные процессы при электролизе растворов.
ќбщие сведени€.
ќкислительно-восстановительна€ реакци€, протекающа€ в растворе или расплаве электролита у электродов при пропускании посто€нного электрического тока, называетс€ электролизом.
“аблица є1.
атодные процессы в водных растворах
| Ёлектрический р€д напр€жений металлов | ||
| Li, K, Ca, Na, Mg, Al | Mn, Zn, Fe | Ni, Sn, Pb Ќ2 Cu, Ag, Pt, Au |
| 2Ќ2O+ 2e→ Ќ2+2OHֿ ћеn++nќЌ-= =ће(ќЌ)n-католит | ¬ыдел€етс€ Ќ2 или ће | Men++ne-=Me0 |
“аблица є2.
јнодные процессы в водных растворах
| јнод | ислотный остаток јсm- | |
| Ѕескислородный | ислородосодержащий | |
| –астворимый | окисление металла анода Me0 - ne→Men+ анод раствор | |
| Ќерастворимый | ќкисление анионов кроме фторидов јс m-- me →јс0 | ¬ щелочной среде: 4ќЌ--4е→ќ2+2Ќ2ќ ¬ кислой и нейтральной: 2Ќ2ќ-4е→ќ2 + 4Ќ + |
јнионы по их способности окисл€тьс€ располагаютс€ в следующем пор€дке
  J-, Br-, S2-, —l-, ќЌ-, SO42-, NO3-, F- J-, Br-, S2-, —l-, ќЌ-, SO42-, NO3-, F-
|
1.
’од работы:
HHm
2. 









3. 

 јнод.
јнод.
4. 

 атод.
атод.
 | ||||||||||||||||
 | ||||||||||||||||
 |  |  | ||||||||||||||
 | ||||||||||||||||
 | ||||||||||||||||
 | ||||||||||||||||
Ёлектролизный стакан.
1. –аствор электролита
2. јнод
3. атод
”равнение электролизного раствора CuSO4 с медным анодом.
A(+): Cu 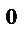 ‒ 2e→Cu
‒ 2e→Cu 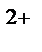
K(‒): Cu 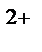 + 2e→Cu
+ 2e→Cu 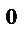 ↓ (масса катода увеличиваетс€)
↓ (масса катода увеличиваетс€)
«акон ‘араде€
m 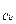
 =
= 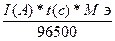
m = 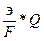
F - константа ‘араде€= 96500 л.
Q Ц количество электричества прошедшего через систему.
Q = I(A) * t(c) = 1,1(A) * 1200(c) = 1320
m = 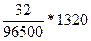 = 0,43
= 0,43
η = 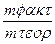 *100 = 90,9
*100 = 90,9
M 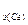 =
= 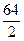 =32 г/моль.
=32 г/моль.
—хема процесса электролиза.

 |  | ||




|
|



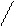
|











|
| J(A) | τ (c) | ћасса катода (Cu) | mт (теор.) | mτ- mo (факт.) | η(—u)% | |
| mo | mт | |||||
| 1,1 | 48,83 | 48,83 | 0,44 | 0,40 | 90,9 |
¬ывод: я изучил электродные процессы при электролизе растворов.
онтрольные вопросы.
1. ¬ каких случа€х образуютс€ вторичные продукты при электролизе? ќтвет: ¬торичные продукты при электролизе образуютс€ во всех случа€х.
2. ¬ какой последовательности и выдел€ютс€ на катоде металлы при электролизе раствора, содержащего ионы Ni2+, Cr2+,Fe3+?
ќтвет: Cr2+, Fe3+, Ni2+
3. акой силы должен быть ток, чтобы при пропускании его через расплав MgSO4 выделить на катоде 6 г. магни€ за 10 часов?
|
|
|
m 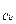
 =
= 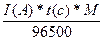
ћ 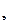

 =
= 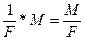
m = 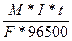 ; I=
; I= 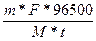
t = 3600с * 10 = 36000с
F = 2
M(Mg) = 24,305
m(Mg) = 6г.
I= 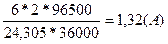
4. „ерез последовательно соединенные электролитические ванны с платиновыми электродами пропустили ток. ¬ первой ванне раствор сульфата натри€, во второй - раствор нитрата серебра, а в третьей - раствор сол€ной кислоты. акие продукты выделились на электродах во всех трех ваннах и сколько их получилось по массе, если в первой ванне выделитс€ на катоде 1 мг. водорода?
K1: 2H2O + 2e = H2 + 2OH-
A1: 2H2O - 4e = O2 + 4H+
m(H 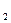 ) = 1мг.
) = 1мг.
m = 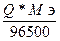
 ; Q =
; Q = 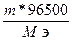
ћэ(Ќ 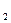 ) =
) =  = 1
= 1
Q = 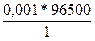 = 96,5 л
= 96,5 л
K2: Ag+ + e = Ag0
A2: 2H2O - 4e = O2 + 4H+
m = 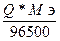
ћэ(Ag) = 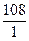 = 108
= 108
m(Ag) = 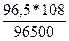 = 0,108г.
= 0,108г.
K3: 2H+ + 2e = H20
A3: 2Cl- - 2e = Cl20
Mэ(Cl) = 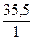 = 35,5
= 35,5
m(Cl) = 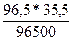 = 0,0355г.
= 0,0355г.
5. „ерез водный раствор NaCl (электроды угольные) пропускали ток сначала при перемешивании раствора, а затем без перемешивани€, отделив в последнем вторичные продукты. Ќаписать уравнени€ электродных процессов дл€ двух случаев.
–ешение дл€ 1 случа€.
: 2H2O + 2e = H2 + 2OH
ј: 2Cl - 2e = Cl2
—уммарное ионное ур-ие:
2H2O + 2Cl = H2 + Cl2 + 2OH
—уммарное молекул€рное ур-ие:
2H2O + 2NaCl = H2 + Cl2 + 2NaOH
–ешение дл€ 2 случа€.
K1: 2H2O + 2e = H2 + 2OH-
A1: 2H2O - 4e = O2 + 4H+
K2: Ag+ + 1e = Ag0
A2: 2H2O - 4e = O2 + 4H+
K3: 2H+ + 2e = H20
A3: 2Cl- - 2e = Cl20






