ѕри составлении уравнений ќ¬– нужно учесть, что число электронов, отданных восстановителем, равно числу электронов, прин€тых окислителем.
¬ химии электрон обозначаетс€ 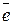
 его условный зар€д прин€т за Ђ-1ї
его условный зар€д прин€т за Ђ-1ї  ƒл€ подбора стехиометрических коэффициентов можно использовать несколько методов. наиболее распространенным относ€тс€ метод электронного баланса и метод электронно-ионных уравнений (метод полуреакций).
ƒл€ подбора стехиометрических коэффициентов можно использовать несколько методов. наиболее распространенным относ€тс€ метод электронного баланса и метод электронно-ионных уравнений (метод полуреакций).
ћетод электронного баланса €вл€етс€ наиболее универсальным методом и применим дл€ любых окислительно-восстановительных процессов, протекающих в любых системах (растворы, расплавы, газы). ¬ основе метода лежит принцип сравнени€ степеней окислени€ атомов в исходных веществах и в продуктах реакции с последующим составлением схемы электронного баланса.
ѕример 2. –ассмотрим метод электронного баланса дл€ уравнивани€ ќ¬-реакции:
 +
+  +
+ 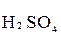 →
→  +
+  +
+  +
+ 
ƒл€ расстановки коэффициентов выполн€ем следующие действи€.
1. ќпредел€ем элементы, атомы которых измен€ют степень окислени€:
 +
+  +
+ 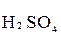 →
→  +
+  +
+  +
+ 
2. Ќаходим окислитель и восстановитель в данной ќ¬–, составл€ем схему перехода электронов от восстановител€ к окислителю и пишем отдельно электронные уравнени€ процессов окислени€ и восстановлени€ с учетом того, что количество атомов, вход€щих в соединение, должно сохран€тьс€. Ќапример, в  имеетс€ два атома Cr, следовательно, в уравнении они должны присутствовать:
имеетс€ два атома Cr, следовательно, в уравнении они должны присутствовать:
+6 ē


 -2 ē
-2 ē
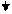


 +
+  +
+ 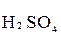 →
→  +
+  +
+  +
+ 
окислитель восстановитель
2Cr+6 + 6 ē = 2Cr+3 (а) - восстановление
S-2 - 2 ē  = S0 (б) Ц окисление
= S0 (б) Ц окисление
3. ”равниваем число электронов в процессе окислени€ и восстановлени€ (составл€ем электронный баланс). ¬ приведенной схеме необходимо уравнение (б) умножить на 3, тогда будет прин€то и отдано по 6 электронов. ѕосле умножени€ уравнени€ складываютс€ как обычные алгебраические:

 2Cr+6 + 6 ē = 2Cr+3 1
2Cr+6 + 6 ē = 2Cr+3 1
+ S-2 - 2 ē  = S0 3
= S0 3
 |
2Cr+6 +3S-2 = 2Cr+3 + 3 S0
4. ѕолученные коэффициенты называют основными. ќни перенос€тс€ в молекул€рную схему реакции и став€тс€ перед соответствующими веществами. “ак как в молекулах K2Cr2O7 и Cr2(SO4)3 содержитс€ по два атома хрома, двойки перед этими веществами опускаютс€.
K2Cr2O7 + 3H2S + H2SO4 = Cr2(SO4)3 + 3S + K2SO4 + H2O
5. ќкончательно уравниваем число атомов каждого элемента в обеих част€х молекул€рного уравнени€. ѕродукты реакции (Cr2(SO4)3, K2SO4), имеющие коэффициенты по единице, содержат 4 моль сульфат-ионов (SO42-), которые содержатс€ в серной кислоте, следовательно, перед ней ставитс€ коэффициент 4. „тобы количество атомов водорода было одинаково в левой и правой част€х уравнени€, перед водой ставитс€ коэффициент 7:
K2Cr2O7 + 3H2S + 4H2SO4 = Cr2(SO4)3 + 3S + K2SO4 + 7H2O
ѕроверка количества остальных атомов показывает, что все коэффициенты подобраны.
ћетод электронно-ионных уравнений (метод полуреакций). Ётот метод основан на составлении уравнений процессов окислени€ и восстановлени€ с помощью ионов и молекул, реально существующих в растворе. —тепени окислени€ атомов не используют, а учитывают зар€ды ионов и характер среды (рЌ), в которой протекает ќ¬-реакци€. ¬ качестве частиц среды в водных растворах могут принимать участие следующие частицы: Ќ+, ќЌ- и Ќ2ќ.
|
|
|
ƒл€ написани€ уравнени€ в ионно-молекул€рной форме следует знать:
1. —ильные кислоты (например, HCl, H2SO4, HNO3) распадаютс€ на ионы H+ и кислотный остаток, например, H2SO4 Ѓ2H+ +SO42-
—лабые кислоты, дл€ которых константа диссоциации д<10-3 (например, H2S, HCN), практически не диссоциируют на ионы и записываютс€ в молекул€рном виде.
2. —ильные основани€ (например, LiOH, NaOH, KOH) распадаютс€ на катион металла и гидроксид-ион OH-, например, ќЌ Ѓ + +OH-
—лабые основани€, дл€ которых д<10-3 (например, NH4OH), практически не диссоциируют на ионы и записываютс€ в молекул€рном виде.
3. —оли распадаютс€ на катионы металла и кислотный остаток. Ќапример, KBr ЃK+ +Br-1:
K Brќ3 Ѓ K+ + Brќ3-1
K2Cr2O7Ѓ 2 K+ + Cr2O72-
4. ќксиды (например, MnO2, FeO, Fe2O3, CO2, SO2, NO) не распадаютс€ на ионы.
5. ¬ода Ќ2ќ ( д=1,8.10-16), пероксид водорода Ќ2ќ2 ( д=1.10-25) как слабые электролиты, не распадаютс€ на ионы.
—хема метода ионно-электронных уравнений:
1. Ќаписать уравнение в ионно-молекул€рной форме.
2. ќпределить кислотно-щелочность среды.
3. ќпределить частицы, изменившие свой зар€д или состав, и записать реакции их превращени€.
4. —оставить материальный баланс дл€ этих превращений, то есть количество атомов каждого из присутствующих элементов в левой и правой част€х уравнени€ должно быть одинаково.
≈сли исходные вещества содержат большее число атомов кислорода, чем полученные продукты, то в кислой среде каждый атом кислорода можно св€зать двум€ ионами водорода в воду, а в нейтральной и щелочной средах молекулой воды в гидроксид-ионы.
≈сли исходные вещества не содержат кислорода или содержат меньшее
число атомов кислорода, чем полученные продукты, то недостающее число атомов кислорода можно восполнить в нейтральной и кислой средах за счет молекул воды, а в щелочной Ц за счет двух ионов ќЌ-.
5. ”равн€ть полученные реакции по зар€дам с участием электронов.
6. —оставить электронный баланс между полуреакци€ми, учитыва€, что количество прин€тых электронов должно равн€тьс€ количеству отданных. ƒалее просуммировать полуреакции: сложить отдельно левые части и отдельно правые части уравнений. ≈сли в суммарном уравнении имеютс€ одинаковые частицы в левой и правой част€х уравнени€, то их сократить.
7. ѕолученные коэффициенты перенести в молекул€рное уравнение и проверить материальный баланс.
ѕример 3. –ассмотрим метод электронно-ионных уравнений дл€ уравнивани€ ќ¬-реакций между KMnO4 и KNO2 в кислой, нейтральной и щелочной средах.






