овосибирск-2010
ћетодическа€ разработка составлена на основании следующих нормативных документов:
√осударственный образовательный стандарт высшего профессионального образовани€. Ќаправление подготовки дипломированного специалиста 260900.65 -У“ехнологи€ и конструирование изделий легкой промышленностиФ. валификаци€ выпускника Ц инженер. Ц ћ.: ћинистерство образовани€ –‘. 2000. Ќомер государственной регистрации 194 тех./ƒ—. ”твержден 23.03.2000.
–азработчик: ¬.Ќ. Ѕондарев
доцент, к.т.н.
–ецензент: ¬.ј. Ћогвиненко
профессор, д.х.н.
ћетодическа€ разработка рассмотрена на заседании кафедры химии,
протокол є ______ от Ђ____ї ___________ 2010 г.
«ав. кафедрой ј.‘. ‘ед€шина
ƒекан ‘«ќиЁ ј.–. —околовский
—ќƒ≈–∆јЌ»≈
| —тр. | |
| 1. ћетодические указани€ ≠≠≠≠≠≠≠≠≠≠≠≠______________________________________ | |
| 2. –аздел 1. ќсновные пон€ти€ и законы химии ____________________ | |
| 2.1 ќсновные пон€ти€ химии__________________________________ | |
| 2.2 ќсновные законы химии ________________________________ | |
| 3. –аздел 2. —троение атома. ’имическа€ св€зь ___________________ | |
| 2.1 —троение атома _________________________________________ | |
| 2.2 ’имическа€ св€зь _______________________________________ | |
| 4. –аздел 3. Ёнергетика химических процессов. “ермохими€ ________ | |
| 3.1 “ермохими€ ___________________________________________ 3.2 Ёнтропи€ химических реакций ___________________________ 3.3 Ёнерги€ √иббса ________________________________________ | |
| 5. –аздел 4. инетика химических реакций. ’имическое равновесие __ | |
| 4.1 —корость химических реакций ___________________________ 4.1.1 —корость химических реакций в гомогенной системе ________ 4.1.2 —корость химических реакций в гетерогенной системе ______ 4.2 ‘акторы, вли€ющие на скорость реакции ___________________ 4.2.1 онцентраци€ реагирующих веществ _____________________ 4.2.2 ƒавление _____________________________________________ 4.2.3 “емпература ___________________________________________ 4.3 ’имическое равновесие __________________________________ 4.3.1 ѕринцип Ће-Ўателье __________________________________ | |
| 6. –аздел 5. –астворы. онцентраци€ растворов. √идролиз солей _____ | |
| 5.1 онцентраци€ растворов _________________________________ 5.2 —войства растворов неэлектролитов ________________________ 5.2.1 ќсмотическое давление _________________________________ 5.2.2 ƒавление пара растворител€ над разбавленными растворами неэлектролитов _________________________________________ 5.2.3 “емпература кипени€ и кристаллизации разбавленных растворов неэлектролитов ______________________________________ 5.3 —войства растворов электролитов _______________________ 5.3.1 —тепень диссоциации __________________________________ 5.3.2 онстанта диссоциации ________________________________ 5.3.3 »онное произведение воды. ¬одородный показатель ________ 5.4 –еакции ионного обмена ________________________________ 5.5 √идролиз солей _________________________________________ | |
| 7. –екомендуема€ литература ___________________________________ 8. “аблица вариантов __________________________________________ |
|
|
|
ћетодические указани€
–абота студента над разделами курса Ђ’ими€ї должна начинатьс€ с самосто€тельного изучени€ материала по учебникам и учебным пособи€м. ƒл€ лучшего усвоени€ материала необходимо конспектировать основные формулировки законов, незнакомые термины, химические формулы и уравнени€ реакций, математические зависимости и т.п.
онтрольна€ работа выполн€етс€ в ученической тетради. ѕосле указани€ номера задани€ пишетс€ содержание вопроса или условие задачи так, как они даютс€ в данном пособии. «адани€ выполн€ютс€ в том пор€дке, в каком они следуют в ¬ашем варианте. ѕри решении задач необходимо производить все математические действи€, использу€ при этом стандартные обозначени€ мер и других величин. ќтветы на теоретические вопросы должны быть краткими, но обоснованными.
јккуратно оформленна€ работа, подписанна€ студентом с указанием даты выполнени€ и шифра (номера студенческого билета), представл€етс€ на рецензирование в институт. Ќезачтенна€ работа возвращаетс€ студенту на доработку. »справление необходимо выполн€ть в той же тетради в соответствии с указани€ми рецензента, но не в рецензированном тексте. –абота после исправлени€ замечаний возвращаетс€ на повторное рецензирование.
онтрольна€ работа, выполненна€ не по своему варианту, преподавателем не рецензируетс€ и не засчитываетс€.
¬ариант контрольной работы, выполн€емой студентом, определ€етс€ двум€ последними цифрами номера его студенческого билета (шифра). Ќапример, номер студенческого билета 2046, последние две цифры Ц 46, из чего следует, что студент должен выполн€ть задани€, включенные в вариант 46.
аздел 1
ќсновные пон€ти€ и законы химии
’ими€ изучает превращение одних веществ в другие. ѕри химических процессах происходит обмен атомами между различными веществами, перераспределение электронов между атомами элементов, разрушение одних соединений и возникновение других. Ќапример:
NaOH + HCl = NaCl +H2O.
1.1 ќсновные пон€ти€ химии
ќбъектом изучени€ химии €вл€ютс€ химические элементы и их соединени€. Ќапример: N, Cl2, NH3, HCl.
’имический элемент Ц вид атомов с одинаковым зар€дом €дра: Al, K, S.
јтом Ц наименьша€ электронейтральна€ частица химического элемента, сохран€юща€ все его химические свойства. —остоит из положительно зар€женного €дра и отрицательно зар€женных электронов. Ќапример,  : пор€дковый номер элемента в периодической системе Ц 30, относительна€ атомна€ масса Ц 65, число электронов в электронной оболочке (Ц30) равно числу протонов, определ€ющих зар€д €дра атома (+30). ядро атома: число протонов (+30) и число нейтронов (65 Ц 30 = 35)
: пор€дковый номер элемента в периодической системе Ц 30, относительна€ атомна€ масса Ц 65, число электронов в электронной оболочке (Ц30) равно числу протонов, определ€ющих зар€д €дра атома (+30). ядро атома: число протонов (+30) и число нейтронов (65 Ц 30 = 35)
ћолекула Ц это наименьша€ частица вещества, обладающа€ его химическими свойствами. ’имические свойства молекулы определ€ютс€ ее составом и химическим строением. Ќапример: H2O, H2SO4, KOH.
¬ещества, молекулы которых содержат атомы только одного элемента, называют простыми. Ќапример: K, H2, O2, O3, Ar, N2.
|
|
|
¬ещества, молекулы которых состо€т из атомов разных элементов, называют сложными веществами или химическими соединени€ми. Ќапример: CO, H2O, HCl, H2SO4, Na2 SO4, CH4, C2H5OH.
’имическое уравнение Ц это запись химической реакции с помощью формул реагентов и продуктов с указанием их количеств в мол€х. Ќапример:
CaCO3 + 2HCl = CaCl2 + CO2 +H2O.
¬ химическом уравнении число атомов каждого элемента в левой и правой част€х уравнени€ одинаково. „исла, сто€щие перед химическими формулами, называютс€ стехиометрическими коэффициентами.
јбсолютные массы атомов ничтожно малы. “ак, например, масса атома водорода составл€ет 1,674 Ј 10-27 кг, кислорода Ц 26,67 Ј 10-27 кг, углерода -
19,93Ј10-27 кг. ѕоэтому в химии используютс€ не абсолютные значени€ атомных масс, а относительные. «а единицу атомной массы прин€та 1/12 часть массы изотопа углерода 12—.
ќтносительна€ атомна€ масса элемента (Ar) Ц это величина, равна€ отношению абсолютной массы атома к 1/12 части абсолютной массы изотопа углерода 12—. Ќапример:
Ar(H) = m(H) / 1/12 m 12C = 1,674 Ј 10-27/ 1/12 × 19,93Ј10-27кг = 1,0079.
Ar(O) = 
ќтносительна€ молекул€рна€ масса (Mr) Ц это величина, равна€ отношению абсолютной средней массы молекулы естественного изотопического состава вещества к 1/12 массы атома углерода 12—. ќна численно равна сумме относительных атомных масс всех атомов, вход€щих в состав молекулы вещества. Ќапример: Mr(H2O):
2Ar(H) = 2 Ј 1,00797 = 2,01594
Ar(O) = 1 Ј 15,9994 = 15,9994
 Mr(H2O) = 18,01534 или Mr(H2O) = 18.
Mr(H2O) = 18,01534 или Mr(H2O) = 18.
ѕолученное число показывает, во сколько раз масса молекулы H2O больше 1/12 массы 12—.
оличество вещества определ€етс€ числом структурных единиц (атомов, молекул, ионов и др.) этого вещества. ≈диницей количества вещества €вл€етс€ моль.
ћоль Ц это количество вещества, содержащее столько структурных единиц (молекул, атомов, ионов и др.), сколько атомов содержитс€ в 0,012 кг изотопа углерода 12—.
«на€ массу одного атома углерода (19,93 Ј10-27 кг), можно вычислить число атомов Nј в 0,012 кг углерода:
Nј = 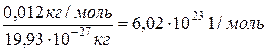
Ёто число называетс€ посто€нной јвогадро, показывает число структурных единиц в моле любого вещества.
ћол€рна€ масса вещества (ћ) Ц это масса одного мол€, равна€ отношению массы к количеству вещества.
ћ = 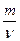 ,
,
где m Ц масса вещества, г;
 - количество вещества, моль.
- количество вещества, моль.
ћол€рна€ масса имеет размерность г/моль или кг/моль.
ћол€рную массу вещества легко вычислить, зна€ массу молекулы.
Ќапример, масса молекулы воды равна 2,99Ј10-26 кг, то мол€рна€ масса ћ(H2O) = 2,99Ј10-26 кг Ј 6,02Ј1023 г/моль = 0,018 кг/моль.
„исленное значение мол€рной массы равно относительной молекул€рной или атомной массе.
Ќапример: M(H2O) = 18 г/моль; Mr(H2O) =18
M(H2SO4) = 98 г/моль; Mr(H2SO4) = 98.
ќтсюда, зна€ мол€рную массу вещества, легко определить массу атома или молекулы этого вещества, разделив мол€рную массу на число јвогадро (NA). Ќапример: m(O) = 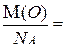

m(H2O) = 


ѕример 1. ќпределите количество вещества брома Br2, содержащегос€ в молекул€рном броме массой 12,8 г.
–ешение.
ћол€рна€ масса Br2 составл€ет:
ћ(Br2) = 2ћ(Br) = 2Ј80 = 160 г/моль
ќпредел€ем количество вещества Br2

ѕример 2. —колько молекул содержитс€ в молекул€рном йоде массой 50,8 г?
–ешение.
ћол€рна€ масса йода составл€ет
ћ(J2) = 2ћ(J) = 2Ј127 = 254 г/моль
ќпредел€ем количество вещества J2

»спользу€ число јвогадро (NA), находим число (n) молекул J2.
N(J2) = n(J2)×NA = 0,2×6,02×1023 = 1,2×1023 молекул.
Ёквивалент Ц это реальна€ или условна€ частица вещества, котора€ соответствует одному иону (атому) водорода в ионноЦобменных реакци€х, или одному электрону в окислительноЦвосстановительных реакци€х.
ћол€рна€ масса эквивалента Ц это масса одного мол€ эквивалента.
ќбозначаетс€ ћ(1/z B) Ц г/моль.,
где 1/z - фактор эквивалентности;
z - число эквивалентности;
¬ Ц вещество.
n(1/z B) Ц количество эквивалентов вещества, (моль).
|
|
|
ћ(1/z ¬) Ц мол€рна€ масса эквивалента вещества ¬, (г/моль).
Ќапример:
 ;
;
 .
.
1.2 ќсновные законы химии
«акон сохранени€ массы вещества. ћасса веществ, вступающих в реакцию, равна массе веществ, образующихс€ в результате реакции.
Ќапример, Ќ2 + —l2 = 2HCl
“ак как атомы имеют посто€нную массу, то не мен€етс€ масса веществ до и после реакции.
«акон посто€нства состава
аждое химически чистое вещество имеет один и тот же качественный и количественный состав независимо от способа его получени€.
Ќапример: 2H2 + O2 = 2H2O
NaOH + HCl = NaCl + H2O
CH4 + 2O2 = CO2 + 2H2O
¬о всех реакци€х состав H2O: (ω(H)=1,11%; ω(ќ)=88,89%).
«акон эквивалентов
¬ещества взаимодействуют друг с другом в количествах, пропорциональных их эквивалентам, т.е. моль эквивалентов одного вещества реагирует с одним молем эквивалентов другого вещества.
Ќапример, в реакции 1/3Al + HCl = 1/3AlCl3 + 1/2H2 один моль эквивалентов алюмини€ (1/3 моль Al)реагирует с одним молем эквивалентов кислоты (1 моль HCl) с образованием одного мол€ эквивалентов хлорида алюмини€ (1/3 моль AlCl3) и одного мол€ эквивалентов водорода (1/2 моль Ќ2).
ћатематическое выражение закона эквивалентов:
 ,
,
где m(A) и m(B) Ц массы взаимодействующих веществ ј и ¬, г;
ћ(1/z ј) и ћ(1/z ¬) Ц мол€рные массы эквивалентов этих веществ, г/моль.
≈сли одно из веществ €вл€етс€ газом, то закон эквивалентов принимает вид:
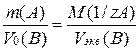 ,
,
где Vo(¬) Ц объем газа (при н.у.), л;
Vэкв(¬) Ц объем мол€ эквивалентов газа, л.
ќпредел€етс€ как 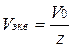 , где Vo Ц мол€рный объем газа, л.
, где Vo Ц мол€рный объем газа, л.
ѕример 3.ќпределите мол€рную массу эквивалента металла, если при взаимодействии металла массой 7,20г с хлором была получена соль массой 28,20г. ћол€рна€ масса эквивалента хлора Ц 35,45 г/моль.
–ешение. Ќаходим массу хлора.
m(Cl) = m(соли) Ц m(Me) = 28,20 Ц 7,20 = 21,00 г
ѕо закону эквивалентов определ€ем мол€рную массу эквивалента металла.


ѕример 4. Ќа восстановление оксида металла массой 1,8 г израсходован водород объемом (н.у.) 833 см3. ¬ычислите мол€рные массы эквивалентов оксида и металла.
–ешение. ѕо закону эквивалентов определ€ем мол€рную массу эквивалента оксида, зна€ что Vэкв(Ќ2) = 11,2 л /моль


M(1/zMe) = M(1/z окс) Ц M(1/z O) =24,2 Ц 8,0 = 16,2 г/моль.
—осто€ние газа характеризуетс€ его температурой, давлением и объемом. ≈сли температура газа равна 273 (0о—), а давление 101,3 кѕа (760 мм.рт.ст.), то услови€, при которых находитс€ газ, называютс€ нормальными. ќбъем, занимаемый газом при этих услови€х, прин€то обозначать через Vo, а давление через –о.
ѕри нормальных услови€х 1 моль различных газов занимает объем, равный 22,4 л Јмоль-1.
Ётот объем называют мольным объемом газа (Vm). ћольный объем газа (Vm) Ц это отношение объема газа к его количеству:
 ,
,
где Vm Ц мольный объем газа (л/моль);
Vo Цобъем газа (л);
n - количество газа (моль).
¬ равных объемах газов при одинаковых услови€х содержитс€ одинаковое число молекул, а следовательно, и молей газов. ќтношение масс данных объемов газов равно отношению их мол€рных масс:
m1: m2 = M1: M2,
где m1 и m2 Ц массы газов, г;
M1 и M2 Ц их мол€рные массы, г/моль.
ќтношение массы определенного объема одного газа к массе такого же объема другого газа (вз€тых при одинаковых услови€х) называют плотностью первого газа по второму (обозначаетс€ буквой ƒ).
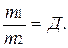 ќтсюда ƒ =
ќтсюда ƒ =  и M1 = ƒћ2
и M1 = ƒћ2
ќбычно плотность газа определ€ют по водороду (обозначаетс€  ), мол€рна€ масса которого 2 г/моль, или по воздуху (обозначают ƒ(¬)), средн€€ мол€рна€ масса которого равна 29 г/моль. ќтсюда, мол€рна€ масса вещества в газообразном состо€нии равна: ћ = 2
), мол€рна€ масса которого 2 г/моль, или по воздуху (обозначают ƒ(¬)), средн€€ мол€рна€ масса которого равна 29 г/моль. ќтсюда, мол€рна€ масса вещества в газообразном состо€нии равна: ћ = 2  или ћ = 29ƒ(¬).
или ћ = 29ƒ(¬).
|
|
|
ѕример 5. ќтносительна€ плотность галогеноводорода по воздуху равна 2,8. ќпределите плотность этого газа по водороду и назовите его.
–ешение. ќпредел€ем мол€рную массу галогеноводорода(Ќ√).
ћ(Ќ√) = 29ƒ(¬) = 29 × 2,8 = 81 г/моль.
ƒалее находим мол€рную массу галогена.
ћ(√) = ћ(Ќ√) Ц ћ(Ќ) = 81 Ц 1 = 80 г/моль.
—ледовательно, галоген-бром, а газ-бромоводород.
¬ычисл€ем плотность HBr по водороду:

«ависимость между объемом газа, давлением и температурой выражаетс€ уравнением лапейрона:

 ,
,
где P,V Ц давление и объем при данной температуре, “;
Pо, Vо, “о Ц давление, объем и температура при нормальных услови€х.
ѕример 6. ѕри температуре 25о— и давлении 99,3 кѕа некоторое количество газа занимает объем 152 см3. ќпределите, какой объем будет занимать этот газ при нормальных услови€х.
–ешение. »спользу€ уравнение лапейрона получаем:
Vо = 
 .
.
ѕример 7. акой объем (л) займет при температуре 20о— и давлении 250 кѕа аммиак массой 51 г?
–ешение. ќпредел€ем количество вещества аммиака:
n(NH3) = 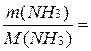

ќбъем газа при нормальных услови€х составит:
V0(NH3) = Vm × ν(NH3) = 22,4 × 3 = 67,2 (л).
ƒалее по уравнению лапейрона определ€ем объем аммиака при данных услови€х задачи:
V(NH3) = 
 .
.
ƒл€ любого газа количеством 1 моль величина  посто€нна, одинакова и ее называют универсальной газовой посто€нной R. ќна имеет различные значени€. “ак, если давление измерено в ѕа, а объем в м3, то R = 8,31ƒж×моль-1× -1 (1ƒж=ѕа×м3). ≈сли давление в мм.рт.ст., а объем в (л), то R = 62,36 мм.рт.ст.×л×моль-1× -1. ”равнение лапейрона принимает вид PV=RT (дл€ ν=1 моль).
посто€нна, одинакова и ее называют универсальной газовой посто€нной R. ќна имеет различные значени€. “ак, если давление измерено в ѕа, а объем в м3, то R = 8,31ƒж×моль-1× -1 (1ƒж=ѕа×м3). ≈сли давление в мм.рт.ст., а объем в (л), то R = 62,36 мм.рт.ст.×л×моль-1× -1. ”равнение лапейрона принимает вид PV=RT (дл€ ν=1 моль).
ƒл€ n-молей газа:
PV=nRT или PV=  .
.
ƒанное уравнение получило название уравнени€ ћенделеева- лапейрона (уравнение состо€ни€ идеального газа.) ќно св€зывает массу газа (кг), температуру ( ), давление (ѕа) и объем (м3) с его мол€рной массой (кг/моль). ѕри этом R=8,31 ƒж/моль× .
ѕример 8. ¬ычислите мол€рную массу бензола, если масса 600 см3 его паров при температуре 870— и давлении 83,2 кѕа равна 1,3 ×10-3 кг.
–ешение. »спользу€ уравнение ћенделеева- лапейрона, находим:
ћ=  .
.
ѕример 9. √азометр объемом 20 л наполнен газом. ѕлотность этого газа по воздуху 0,4, давление 103,3 кѕа, температура 170—. ќпределите массу газа.
–ешение. Ќаходим мол€рную массу газа.
ћ(газа) = 29 × ƒ(¬) = 29×0,4 = 11,6 г/моль
ƒалее по уравнению ћенделеева- лапейрона определ€ем массу газа.
m(газа) = 
√азы часто представл€ют собой смеси индивидуальных газообразных веществ. “ак, воздух €вл€етс€ смесью азота, кислорода, аргона и др.
ѕарциальным давлением газа в газовой смеси называетс€ давление, которое производил бы газ, занима€ при тех же услови€х объем газовой смеси.
«акон парциальных давлений ( ƒальтон).
ќбщее давление смеси газов, не вступающих друг с другом в химическое взаимодействие, равно сумме парциальных давлений газов, составл€ющих смесь.
– = р1 + р2 +Е+рn, где
– Ц общее давление газовой смеси
р1,р2,рn Ц парциальное давление газов.
ѕарциальное давление газа в газовой смеси можно рассчитать по уравнению ћенделеева- лапейрона.
ѕример 10. √азова€ смесь приготовлена из 2 л водорода (–нач. =93,3 кѕа) и 5 л метана (–ннач. = 112 кѕа). ќбъем смеси равен 7 л. –ассчитайте парциальные давлени€ газов и общее давление смеси.
–ешение. «на€ объемы газов и смеси, находим парциальные давлени€ газов:



ќбщее давление газовой смеси равно сумме парциальных давлений газов.
– = –(H2) + P(CH4) = 26,66 + 80 = 106,66 кѕа.
ѕример 11. ѕри температуре 0о— в сосуде объемом 14,0×10-3 м3 содержитс€ водород массой 0,8 ×10-3 кг и азот массой 6,3×10-3 кг. ќпределите парциальное давление газов и общее давление газовой смеси.
–ешение. ѕо уравнению ћенделеева- лапейрона находим парциальное давление газов.
–(H2) = 
–(N2) = 
–общ. = –(Ќ2) + P(N2) = 64,80 + 36,46 = 101,26 кѕа
≈сли газ собран над жидкостью, то при расчетах следует иметь в виду, что его давление €вл€етс€ парциальным и равно разности общего давлени€ газовой смеси и парциального давлени€ пара жидкости. Ќапример, газ собран над водой при температуре 20о— и давлении 100 кѕа, а давление насыщенного пара воды при 20о— равно 2,3 кѕа. “огда, парциальное давление газа будет равно 100 Ц 2,3 = 97,7 кѕа. Ётой величиной пользуютс€ в дальнейших расчетах.
ѕример 12. ¬одород объемом 250 см3 собран над водой при температуре 26о— и давлении 98,7 кѕа. ƒавление насыщенного пара воды при 26о— составл€ет 3,4 кѕа. ¬ычислите объем водорода при нормальных услови€х и его массу.
|
|
|
–ешение. Ќаходим парциальное давление водорода.
–(Ќ2) = – - –(Ќ2ќ) = 98,7 Ц 3,4 = 95,3 кѕа.
ѕо уравнению лапейрона определ€ем объем водорода (н.у.)
V0(H2) =  см3
см3
Ќаходим количество водорода этого объема.
n(Ќ2) = 
ќтсюда масса водорода:
m(H2) = nM = 0,0096×2=0,019 г.
ѕример 13. ќпределите молекул€рную формулу соединени€, содержащего 30,43% азота и 69,57% кислорода (по массе). ѕлотность этого вещества по водороду равна 46.
–ешение. Ќаходим мол€рную массу этого вещества.
M(NxOy) = 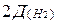 = 2×46 =92 г/моль.
= 2×46 =92 г/моль.
ќпредел€ем массу азота и кислорода в моле этого соединени€.
m(N) = 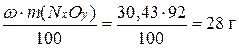
m(O) = 
ќтсюда 14 х: 16 у = 28: 64
х: y =  x = 2; y = 4
x = 2; y = 4
‘ормула Ц N2O4.
«адачи к разделу 1:
Ђќсновные пон€ти€ и законы химииї
1 Ќа осаждение хлора, содержащегос€ в 0,666×10-3 кг соли, израсходовано 1,088×10-3 кг AgNO3. ¬ычислите мол€рную массу эквивалента соли.
2 ¬ закрытом сосуде объемом 5 л находитс€ при температуре 15о— смесь, состо€ща€ из 4,4г —ќ2, 6,4 г ќ2 и 2,4г —Ќ4. ¬ычислите общее давление газовой смеси, парциальные давлени€ газов и процентный состав смеси по объему.
3 ћеталл массой 0,70 г вытеснил из кислоты 418 см3 водорода, собранного над водой при температуре 150— и давлении 94,3 кѕа. ƒавление вод€ного пара при этой температуре равно 1,7 кѕа. ќпределите мол€рную массу эквивалента металла.
4 ¬ычислите среднюю плотность по воздуху газовой смеси, имеющей объемный состав: 52% —Ќ4 и 48% —2Ќ4.
5 ѕри нагревании 0,612г некоторого вещества выделилось 0,168л кислорода (н.у.) и образовалось 0,372г хлорида кали€. ќпределите формулу этого вещества.
6 ¬ газометре над водой при температуре 200— и давлении 98500 ѕа находитс€ 8×10-3 м3 кислорода. ƒавление вод€ного пара при 200— равно 2335 ѕа. акой объем (н.у.) займет кислород, наход€щийс€ в газометре.
7 ћасса газа объемом 0,327 л при температуре 130— и давлении 1,040×105 ѕа равна 0,838 г. ¬ычислите молекул€рную массу газа.
8 ќбъем резиновой камеры автомобильной шины равен 0,025 м3, давление в ней 5,066×105 ѕа. ќпределите массу воздуха в камере при температуре 200—.
9 ¬ычислите, какое давление надо приложить, чтобы хлор массой 28,4г занимал объем 2,0 л при температуре 200—?
10 ¬ сосуде объемом 0,050 м3 при температуре 250— содержитс€ смесь из 0,020 м3 этилена под давлением 83950 ѕа и 0,015 м3 метана под давлением 95940 ѕа. Ќайдите парциальные давлени€ газов и общее давление газов в сосуде.
11 √азова€ смесь состоит из 5 л азота, наход€щегос€ под давлением 95940 ѕа и 3 л кислорода. ќбъем смеси 8 л. ќбщее давление газовой смеси 104200 ѕа. ѕод каким давлением был вз€т кислород?
12 ¬ закрытом сосуде объем 0,6 м3 при температуре 00— находитс€ смесь, состо€ща€ из 0,20 кг —ќ2, 0,40 кг ќ2 и 0,15 кг —Ќ4. ¬ычислите: а) парциальные давлени€ газов; б) общее давление газовой смеси.
13 ¬ газонаполненных лампах содержитс€ смесь газов, имеюща€ объемный состав: 86% Ar и 14% N2. –ассчитайте парциальные давлени€ газов, если общее давление равно 39990 ѕа.
14 —ухой воздух имеет примерно следующий объемный состав: 78,09 % N2, 20,95 % ќ2, 0,93% Ar и 0,03% —ќ2. ¬ычислите парциальные давлени€ этих газов, если общее давление газовой смеси 101325 ѕа.
15 ¬ закрытом сосуде объемом 6 л находитс€ при температуре 100— смесь, состо€ща€ из 8,8 г —ќ2, 3,2 г ќ2 и 1,2 г —Ќ4. ¬ычислите общее давление газовой смеси, парциальные давлени€ газов и процентный состав смеси по объему.
16 ћеталл массой 0,35 г вытеснил из кислоты 209 см3 водорода, собранного над водой при температуре 200— и давлении 104,3 кѕа. ƒавление вод€ного пара при этой температуре равно 2,3 кѕа. ќпределите мол€рную массу эквивалента металла.
17 ѕри нагревании 1,225г некоторого вещества выделилось 0,336л кислорода (н.у.) и образовалось 0,745г хлорида кали€. ќпределите формулу этого вещества.
18 ќпределите молекул€рную формулу оксида хлора, если при разложении оксида объемом 0,10л получились кислород и хлор объемом 0,10л и 0,05л соответственно (н.у.) ќтносительна€ плотность этого оксида по воздуху равна 2,34.
19 ќпределите формулу вещества, состо€щего из углерода, водорода и кислорода, зна€, что при сжигании 0,145г его получено 0,330г —ќ2 и 0,135г Ќ2ќ. ќтносительна€ плотность пара этого вещества по водороду равна 29.
20 акова простейша€ и молекул€рна€ формулы газообразного углеводорода, если в нем содержитс€ 81,82% углерода и 18,18% водорода, а 1л этого углеводорода (н.у.) имеют массу 2,6 г?
аздел 2
—троение атома. ’имическа€ св€зь
2.1 —троение атома
ѕор€дковый номер химического элемента в периодической системе ƒ.».ћенделеева соответствует числу протонов в €дре атома элемента и числу электронов в электронной оболочке атома. ’имические свойства соединений завис€т в основном от строени€ внешних электронных уровней атомов элементов.
ќписать строение электронных оболочек атомов можно с помощью принципа ѕаули, наименьших энергий и правила √унда, использу€ дл€ этого представлени€ о квантовых числах.
√лавное квантовое число ФnФ в многоэлектронном атоме обозначает номер электронного уровн€. ќно принимает целочисленные значени€ от 1 до ∞: n = 1, 2, 3, Е и характеризует энергию электрона, размер электронного облака, число энергетических подуровней внутри каждого уровн€.
ќрбитальное квантовое число ФℓФ характеризует электронные подуровни. ќно принимает дл€ данного ФnФ все целочисленные значени€ от 0 до (n - 1): ℓ = 0, 1, 2Е(n - 1). ѕодуровни, характеризующиес€ значени€ми ℓ, равными 0, 1, 2, 3, называют соответственно s-, p-, d-, f-подуровн€ми, а наход€щиес€ на них электроны - s-, p-, d-, f - электронами. ¬се орбитали одного подуровн€ имеют одинаковую форму электронных облаков.
ћагнитное квантовое число ФmℓФ принимает все целочисленные значени€ от Цℓ до + ℓ, включа€ Ф0Ф: mℓ = -3, -2, -1, 0, +1, +2, +3. „исло значений ФmℓФ соответствует числу орбиталей в подуровне и их ориентации в пространстве.
ѕример 1. —колько орбиталей включает р-подуровень?
–ешение. ƒл€ р-подуровн€ ℓ = 1. „исло значений mℓ равно трем (+1, 0, -1), следовательно р-подуровень включает три р-орбитали (px, py, pz) с их ориентацией по координатам x, y, z.
—пиновое квантовое число ФmsФ принимает два значени€ +1/2 и Ц1/2 и характеризует магнитное поле электрона. ≈сли атом имеет два электрона, и оба они характеризуютс€ ms = +1/2, то суммарный спин равен 1; если один электрон атома характеризуетс€ ms = +1/2, а другой - ms = -1/2, то суммарный спин равен нулю (правило √унда). Ёлектрон с соответствующим квантовым числом обозначаетс€ в электронно-графических формулах стрелочкой, например, ↓, что соответствует ms = -1/2 или ↑ - ms = +1/2.
—истема четырех квантовых чисел позвол€ет установить, как распредел€ютс€ по уровн€м, подуровн€м и орбитал€м электроны атома. ќднако в атоме не может быть двух электронов, характеризующихс€ одинаковыми наборами четырех квантовых чисел (принцип ѕаули). ќн определ€ет общее число электронов в атоме.
ѕример 2. —колько электронов может содержать первый энергетический уровень?
–ешение. ƒл€ первого уровн€ n = 1. —ледовательно, ℓ = 0; mℓ = 0; ms может быть +1/2 и Ц1/2. ратко комбинации квантовых чисел можно выразить такой последовательностью: n, ℓ, mℓ, ms. ƒл€ первого уровн€ можно составить следующие комбинации квантовых чисел 1, 0, 0, +1/2 и 1, 0, 0, -1/2. ќтсюда следует, что первый уровень может содержать два электрона, а первый период таблицы ƒ.».ћенделеева должен содержать два элемента.
ћаксимальное число электронов на энергетическом уровне определ€ют по формуле z = 2n2, где n Ц номер энергетического уровн€, совпадающий со значением главного квантового числа (n = 1, 2, 3 и тд.).
ћаксимальное число электронов на энергетическом подуровне определ€ют по формуле z=2ּ(2ℓ+1), где ℓ Ц номер энергетического подуровн€, совпадающий со значением орбитального квантового числа (ℓ = 0, z = 2; ℓ = 1, z = 6; ℓ = 2, z = 10; ℓ = 3, z = 14).
«аполнение электронами атомных орбиталей (јќ) в многоэлектронных атомах основано на принципе наименьшей энергии. аждый электрон занимает ту из доступных дл€ него орбиталей, на которой его энерги€ будет наименьшей.
ѕоследовательность заполнени€ электронами подуровней многоэлектронных атомов основана на этом же принципе и согласно правила лечковского зависит от суммы (n+ℓ).


 ¬ пределах каждого значени€ суммы (n+ℓ) пор€док заполнени€ подуровней соответствует возрастанию главного квантового числа.
¬ пределах каждого значени€ суммы (n+ℓ) пор€док заполнени€ подуровней соответствует возрастанию главного квантового числа.
Ќапример,
ѕодуровни
а) 3s 3p; б) 3d 4s; в) 3d 4p
n 3 3 3 4 3 4
ℓ 0 1 2 0 2 1
n+ℓ 3 4 5 4 5 5
3s < 3p 4s < 3d 3d ≈ 4p
≈3s < ≈3р ≈4s< ≈3d ≈3d ≈ E4p
¬ случае в) при одинаковой сумме квантовых чисел предпочтение в распределении электронов по орбитал€м следует отдать орбитали 3d, как имеющей меньшее значение n = 3.
ѕример 3. акова последовательность заполнени€ электронами таких подуровней, дл€ которых сумма (n + ℓ) равна 5?
–ешение. —оставим таблицу возможных значений n и ℓ.
n + ℓ 5
n 3 4 5
ℓ 2 1 0
подуровни 3 d 4 p 5 s
ѕример 4. Ќапишите электронную формулу атома элемента с пор€дковым номером 12. ќпределите число электронов на внешнем уровне, к какому семейству элементов данный атом принадлежит, в каком периоде, группе он находитс€.
–ешение. ¬ соответствии с правилом лечковского подуровни заполн€ютс€ в такой последовательности: 1s 2s 2p 3s 3p. »з принципа ѕаули следует, что максимальное число электронов на s Ц подуровне равно 2, на p Ц подуровне 6. —уммарное число электронов составило 12: 1 s2 2 s2 2 p6 3 s2. Ёто элемент второй группы, так как на внешнем уровне два электрона на s-подуровне и поэтому он относитс€ к семейству s-элементов. Ёлемент находитс€ в третьем периоде, так как максимальное значение главного квантового числа равно трем.
—троение атома выражают электронной и электронно-графической формулами. Ќапример, строение атома фтора с пор€дковым номером дев€ть выражают электронной формулой: 1s2 2s2 2p5 и электронно-графической формулой:
9F
| ↑↓ | ↑↓ | ↑ | ||
| ↑↓ | ||||
| ↑↓ |
»ли 1 s 2 s 2 p




 ↑↓ ↑↓ ↑↓ ↑↓ ↑
↑↓ ↑↓ ↑↓ ↑↓ ↑
ѕример 5. »зобразите электронно-графические формулы атомов углерода и железа.
–ешение.
а) электронна€ формула атома углерода с пор€дковым номером шесть: 6— 1s22s22p2; электронно-графическа€ формула:
6C 2p
| 2 s | ↑ | ↑ | ||
| 1 s | ↑↓ | |||
| ↑↓ |
»ли 1 s 2 s 2 p




 ↑↓ ↑↓ ↑ ↑
↑↓ ↑↓ ↑ ↑
¬нешним €вл€етс€ второй энергетический уровень: Е2s2 2p2 и соответственно
2s 2p



 ↑↓ ↑ ↑.
↑↓ ↑ ↑.
б) Ёлектронна€ формула атома железа с пор€дковым номером двадцать шесть: 1s2 2s2 2p6 3s23p64s23d6. ¬нешним €вл€етс€ Е3d64s2 и соответственно: 3d 4s





 ↑↓ ↑ ↑ ↑ ↑ ↑↓.
↑↓ ↑ ↑ ↑ ↑ ↑↓.
2.2 ’имическа€ св€зь
уществует два подхода к описанию химической св€зи: метод валентных св€зей (ћ¬—) и метод молекул€рных орбиталей (ћћќ).
1. ћетод валентных св€зей исходит из того, что химическа€ св€зь осуществл€етс€ св€зывающими электронными парами, кажда€ из которых принадлежит двум атомам. —в€зывающие электронные пары могут образовыватьс€ по обменному или донорно-акцепторному механизму.
1.1. ѕо обменному механизму св€зь возникает за счет перекрывани€ электронных орбиталей неспаренных электронов двух атомов. ¬ св€зывающей электронной паре электроны имеют антипараллельные спины.
ѕример 1. ѕокажите, какие орбитали и как участвуют в образовании химиче- ской св€зи в молекулах: а) Li2; б) LiF; в) F2. Ќапишите графические формулы этих молекул.
–ешение: Ёлектронные формулы атомов лити€ и фтора имеют вид:
3Li 1s22s12p0 9F 1s22s22p5
и электронно-графические формулы их соответственно:
2s 2p 2s 2p







 ↓ ↑↓ ↑↓ ↑↓ ↑
↓ ↑↓ ↑↓ ↑↓ ↑
Li F
Ќеспаренный электрон атома лити€ занимает 2s-орбиталь, а атома фтора 2p-орбиталь. —ледовательно, эти орбитали участвуют в образовании химической св€зи вышеприведенных соединений. Ёто можно представить:
 |  |
Li-Li Li-F
 |
F-F
„исло св€зей, которые образует атом по обменному механизму, как правило, равно числу его неспаренных электронов в основном или валентно-возбужденном состо€нии. ¬ методе валентных св€зей исход€т из того, что валентно-возбужденные состо€ни€ атома, отличающиес€ от основного большим числом неспаренных электронов, получают переводом одного или нескольких электронов электронных пар на свободные орбитали другого подуровн€ того же электронного уровн€.
ѕример 2. —колько св€зей по обменному механизму могут образовать атомы серы и кислорода? Ќапишите эмпирические формулы их соединений со фтором.
–ешение. ислород с пор€дковым номером восемь €вл€етс€ элементом 2-го периода. Ёлектронна€ формула 8ќ 1s22s22p6. Ќа валентном уровне атом имеет 4 орбитали и 6 электронов:
2s 2p



 ↑↓ ↑↓ ↑ ↑
↑↓ ↑↓ ↑ ↑
¬ валентном электронном уровне атома кислорода нет вакантных орбиталей. —ледовательно, атом кислорода может образовать по обменному механизму только две св€зи. —оединение со фтором имеет формулу ќF2 и соответственно графическую F-O-F.
—ера с пор€дковым номером шестнадцать Ц элемент 3-го периода. Ёлектронна€ формула 1s22s22p63s23p4.
¬алентный уровень серы состоит из 3-х подуровней и 9 орбиталей.
јтом серы может иметь два валентно-возбужденных состо€ни€.
3s 3p 3d








 ↑↓ ↑↓ ↑ ↑
↑↓ ↑↓ ↑ ↑
3s 3p 3d









 ↑↓ ↑ ↑ ↑ ↑ сера (IV)
↑↓ ↑ ↑ ↑ ↑ сера (IV)
3s 3p 3d








 ↑ ↑ ↑ ↑ ↑ ↑ сера (VI).
↑ ↑ ↑ ↑ ↑ ↑ сера (VI).
ќна может образовывать 2, 4 и 6 св€зей по обменному механизму.
—оединени€ серы со фтором имеют формулы SF2; SF4; SF6.
—в€зи между атомами могут быть кратными (двойными, тройными). ратные св€зи образуютс€ с участием σ- и π- св€зей.
ѕример 3. ѕокажите, какие орбитали и как участвуют в образовании св€зи в молекуле —2. Ќапишите графическую формулу молекулы —2.
–ешение. Ёлектронна€ конфигураци€ атома углерода с пор€дковым номером шесть 6— 1s22s22p2 и соответственно электронно-графическа€ формула имеет вид:
2s 2p



 ↑↓ ↑ ↑
↑↓ ↑ ↑
¬ образовании св€зей у каждого атома углерода могут участвовать два р-электрона (–х, –у): одна св€зывающа€ электронна€ пара образует σ-св€зь (–х-–х), друга€ π (–у-–у)
 p
p
+ +
— —
|

—в€зь может образоватьс€ за счет неподеленной пары электронов одного атома и свободной орбитали другого атома. јтом, представл€ющий дл€ образовани€
 св€зи электронную пару, называетс€ донором (ƒ), а атом, имеющий свободную орбиталь, называетс€ акцептором (ј).
св€зи электронную пару, называетс€ донором (ƒ), а атом, имеющий свободную орбиталь, называетс€ акцептором (ј).
ƒ + ј = ƒ ј
Ќапример, образование иона аммони€:
2s 2p, 1s 




 ↑↓ ↑ ↑ ↑ ¯
↑↓ ↑ ↑ ↑ ¯
N H 
N + 3H Ѓ NH3
|



















 Ќ N +H+ = H N Ќ
Ќ N +H+ = H N Ќ
|
|

ƒ ј
√еометрическую структуру молекул, направленность ковалентных св€зей определ€ет теори€ гибридизации атомных орбиталей центрального атома.
√ибридизаци€ Ц это перераспределение электронной плотности между разными, но близкими по энергии атомными орбитал€ми одного энергетического уровн€ и образование гибридных атомных орбиталей, численно равных количеству исходных и одинаковых по форме и по энергии.
 Ќапример, в гибридизации могут участвовать s- и p-орбитали одного энергетического уровн€, обладающие различной симметрией. ¬ результате образуютс€ две гибридные орбитали. √ибридные атомные орбитали (√јќ) имеют несимметричную форму. ” таких орбиталей электронна€ плотность по одну сторону выше, что и определ€ет перекрывание электронных облаков и делает более устойчивой химическую св€зь.
Ќапример, в гибридизации могут участвовать s- и p-орбитали одного энергетического уровн€, обладающие различной симметрией. ¬ результате образуютс€ две гибридные орбитали. √ибридные атомные орбитали (√јќ) имеют несимметричную форму. ” таких орбиталей электронна€ плотность по одну сторону выше, что и определ€ет перекрывание электронных облаков и делает более устойчивой химическую св€зь.

“ипы гибридизации атомных орбиталей
1. омбинаци€ одной s и одной p-орбиталей приводит к sp-гибридизации. ќбразуютс€ два гибридных облака, ориентированных в пространстве под углом 1800.
Ќапример, образование молекулы ¬е—l2:
 |
4Be
1s22s2 
 *Be 1s22s12p1 (перевод в возбужденное состо€ние). ¬ результате гибридизации 2s и 2p орбиталей образуетс€ линейна€ св€зь и линейна€ молекула
*Be 1s22s12p1 (перевод в возбужденное состо€ние). ¬ результате гибридизации 2s и 2p орбиталей образуетс€ линейна€ св€зь и линейна€ молекула
 Cl Be Cl
Cl Be Cl
1800
Ёлементы II группы, подверга€сь sp-гибридизации, образуют линейные молекулы.
2. омбинаци€ одной s- и двух p-орбиталей приводит к sp2-гибридизации.
Ќапример, образование молекулы ¬Ќ3.
5¬ 1s22s22p1  B* 1s22s12p2. ¬ результате гибридизации одной s- и двух p- орбиталей образуетс€ треугольна€ плоска€ молекула.
B* 1s22s12p2. ¬ результате гибридизации одной s- и двух p- орбиталей образуетс€ треугольна€ плоска€ молекула.
 |

Ёлементы III группы, подверга€сь sp2-гибридизации, образуют треугольные плоские молекулы.
3. омбинаци€ одной s- и трех p- орбиталей приводит к sp3- гибридизации.
 |
Ќапример, образование молекулы —H4:
6— 1s22s22p2  C* 1s22s12p3
C* 1s22s12p3
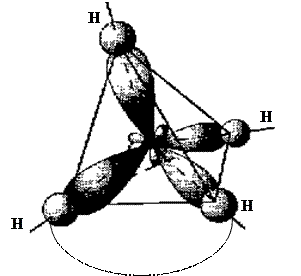 ¬ результате гибридизации одной s- и трех p-орбиталей образуетс€ тетраэдрическа€ молекула.
¬ результате гибридизации одной s- и трех p-орбиталей образуетс€ тетраэдрическа€ молекула.
1090 28¢
Ёлементы IV группы, подверга€сь sp3-гибридизации, образуют тетраэдрические молекулы.
«адачи к разделу 2:
Ђ—троение атома. ’имическа€ св€зьї
2.1 —троение атома
21. —оставьте электронные формулы и укажите значени€ квантовых чисел n и l дл€ внешних электронов в атомах элементов с пор€дковыми номерами 14, 23, 35.
22. ќбъ€сните, какие квантовые числа определ€ют размер, форму и ориентацию в пространстве электронной орбитали. ѕриведите примеры.
23. ќпределите максимальное число электронов, наход€щихс€ на третьем и четвЄртом энергетическом уровн€х. ѕриведите электронные формулы соответствующих атомов.
24. Ќапишите электронные формулы и изобразите электронно-графическую схему распределени€ электронов в атомах с пор€дковыми номерами 46, 55.
25. Ќапишите электронные формулы атомов хрома и меди. ќбъ€сните нахождение одного s-электрона на четвЄртом энергетическом уровне.
26. Ёнергетическое состо€ние внешнего электрона описываетс€ следующими значени€ми квантовых чисел: n = 4, l = 0, ml = 0. јтомы, каких элементов имеют такой электрон? Ќапишите электронные формулы атомов этих элементов.
27. ƒл€ атома возможны два электронных состо€ни€: 1s22s22p63s23p2 и 1s22s22p63s13p3. ак перейти от первого состо€ни€ ко второму? Ќазовите эти состо€ни€ и соответствующий атом, его положение в периодической системе.
28. Ќапишите электронные формулы элементов с пор€дковыми номерами 24 и 53. ѕо электронной формуле определите их положение в периодической системе (период, группу, подгруппу). ѕредставьте их электронно-графические схемы.
29. ”кажите, атомы каких элементов имеют следующее строение внешнего и предвнешнего энергетических уровней: Е3s2 3p3, Е3d10 4s2 4p5, 4d7 5s2.Е4p6. —оставьте электронные формулы.
30. ѕользу€сь правилом √унда, распределите электроны по орбитал€м, соответствующим низшему энергетическому состо€нию, дл€ элементов с пор€дковыми номерами 23, 33, 52.
31. »зобразите электронно-графическую формулу атома элемента с пор€дковым номером 40 и укажите число электронных пар и неспаренных электронов в атоме этого элемента в нормальном состо€нии.
32. ”кажите, положение элементов в периодической системе элементов ƒ.».ћенделеева (период, группу, подгруппу), имеющих следующие электронные формулы: а) 1s22s22p63s23p3, б) 1s22s22p63s23p64s23d2, в) 1s22s22p63s23p64s23d104p5.
33. Ќапишите электронные и электронно-графические формулы атомов элементов: кали€, никел€, магни€. какому типу элементов они относ€тс€ (s-, p-, d-, f- элементам)?
34. Ќапишите электронные и электронно-графические формулы дл€ ионов: Fe3+, S2-, Br¯, Cr3+.
35. — точки зрени€ теории строени€ атома объ€сните изменение свойств элементов в пределах периода и группы.
36. Ќапишите электронные и электронно-графические формулы электронных оболочек ионов: Cu2+, Cl-, Mg2+.
37. ќбъ€сните, почему свойства таких элементов, как азот и висмут, неон и магний резко отличаютс€ друг от друга. —оставьте их электронные формулы.
38. »з двух элементов один образует ион Ё2+, а другой Ц Ё2-. ќба иона имеют одинаковую конфигурацию, котора€ выражаетс€ формулой 1s22s22p63s23p6. ќпределите период, группу, подгруппу и пор€дковый номер каждого элемента.
39. ќпределите, отличаютс€ ли два элемента числом валентных электронов и характером химических свойств по окончани€м их электронных формул: а) Е3d2 ƒата добавлени€: 2017-02-11; ћы поможем в написании ваших работ!; просмотров: 703 | Ќарушение авторских прав Ћучшие изречени€:






