ƒл€ неизолированных систем нужно учитывать не только изменение энтропии, но и изменение энергии. ѕоэтому необхо≠димо рассматривать две тенденции, определ€ющие направление самопроизвольно протекающих процессов:
= стремление системы к достижению минимума энергии;
= стремление системы к максимуму энтропии, то есть к
неупор€доченности.
ѕроцессы, при которых энерги€ в неизолированной системе уменьшаетс€, а энтропи€ возрастает, протекают самопроизвольно. —амопроиз≠вольность других процессов зависит от того, кака€ из этих двух тенденций Ц энергетическа€ или энтропийна€ Ц окажетс€ более эффективной, кака€ из этих противоборствующих тенденций получит перевес над другой. ¬ этом про€вл€етс€ противоречи≠вость материального мира.
ƒл€ однозначной формулировки условий протекани€ само≠произвольных процессов в любых системах введена ещЄ одна термодинамическа€ функци€, котора€ характеризует одновременно и энергетику, и неупор€доченность систем. ¬первые такую термодинамическую функцию ввЄл ƒ. ”. √иббс, и в пам€ть об этом выдающемс€ американском учЄ≠ном еЄ назвали энергией √иббса.
| ƒжозай€ ”иллард √иббс (11.11 1839=28. IV 1903). јмериканский физик и физико-химик, член Ќациональной јЌ —Ўј (с 1879). –одилс€ 11 феврал€ в Ќью-’ейвене. ќкончил …ельский университет (1858; доктор философии, 1863). ¬ 1863=1866 преподавал в …ельском колледже. —овершенствовал образование в —орбонне и оллеж де ‘ранс в ѕариже (1866=1867), в Ѕерлин≠ском (1867) и √ейдельбергском (1868) университетах. — 1871 Ц профессор …ельского университета. ќдин из основоположников тер≠модинамики. | 
|
ѕредложил (1873) графические модели описани€ тер≠модинамических свойств веществ. –азработал (1875=1878) теорию термодинамических потенциалов, изучил (1875) услови€ равнове≠си€ гетерогенных систем. ¬ывел (1875=1878) правило фаз, соглас≠но которому в равновесной гетеро≠генной системе число фаз не мо≠жет превышать числа компонен≠тов системы, увеличенного на два. ѕредложил (1878) графическое изображение состо€ни€ трЄхкомпонентной системы (треугольник √иббса). «аложил основы термо≠динамики поверхностных €влений и электрохимических процессов; ввЄл пон€тие адсорбции. ƒал (1902) атомистическое истолкова≠ние развитым им термодинамиче≠ским методам. ќсуществил иссле≠довани€ в области векторного ана≠лиза и алгебры кратных чисел, электромагнитной и механической теорий света.
√иббс „лен јмериканской академии искусств и наук (с 1880),и многих академий наук и научных обществ.
Ѕиохимические процессы обычно происхо≠д€т при изобарно-изотермических услови€х. ¬ этих услови€х энергетическое состо€ние системы характеризуетс€ энтальпией, а мера еЄ неупор€доченности характеризуетс€ произведением еЄ эн≠тропии и температуры. ‘ункцией, учитывающей обе эти ха≠рактеристики и противоположность в тенденции их изменени€ при самопроизвольных процессах, €вл€етс€ энерги€ √иббса:
G = Ќ -TS.
Ёнерги€ √иббса €вл€етс€ обобщЄнной термодинамической функцией состо€ни€ системы, учитывающей энергетику и неупор€доченность системы, при изобарно-изотермичес≠ких услови€х.
|
|
|
Ёнергию √иббса называют также изобарно-изотермическим потенциалом или свободной энергией.
ѕодобно другим термодинамическим параметрам и функ≠ци€м, характеризующим состо€ние системы, изменение энер≠гии √иббса в результате любого процесса определ€етс€ только конечным и начальным состо€нием системы, независимо от пу≠ти процесса:
DG = Gкон - Gнач
ƒл€ энергетической характеристики веществ при стандартных услови€х используют стандартные энергии √иббса их образовани€ DGо, значени€ которые даны в справочной литературе. ƒл€ простых веществ в термодинамически устой≠чивой форме стандартна€ энерги€ √иббса их образовани€ ус≠ловно принимаетс€ равной нулю.
»зменение энергии √иббса в результате химической реак≠ции при стандартных услови€х вычисл€ют по уравнению:
D G р = ∑vкон D G кон (Yкон) - ∑vисх D G исх (Xисх),
где D G кон(Yкон), D G исх(Xисх) - стандартные энергии √иббса продуктов
реакции Yкон и исходных веществ Xисх.
v кон. и v на ч - соответствующие стехиометрические коэффициенты в уравнении реакции.
»зменение энергии √иббса дл€ биохимических процессов при услови€х, отличных от стандартных, рассчитывают на основе экспериментальных значений DЌ и DS дл€ этих процессов по уравнению:
DG = DЌ - TDS,
где DЌ - характеризует полное изменение энергии системы при р,
“ = const и отражает стремление системы к минимуму энергии;
TDS - характеризует ту часть энергии, которую нельз€ превратить в работу, и отражает стремление системы к максимуму неупор€доченности;
DG - характеризует ту часть энергии, которую можно превратить в работу, и €вл€етс€ термодинамическим критерием возможности самопроиз≠вольного протекани€ любых процессов при р, “ = const.
—оотношение между из≠менени€ми важнейших термо≠ди-намических параметров и функций состо€ний системы, описывающими химические и биохимические процессы при р, “ = const, представлено в таблице и на следующей схеме.

¬торой закон термодинамики систем формули≠руетс€ следующим образом: в системе при посто€нных температуре и давлении са≠мопроизвольно могут совершатьс€ только такие процес≠сы, в результате которых энерги€ √иббса уменьшаетс€ (DGкон < DGнач или DG < 0).
“аким образом, согласно второму закону термоди≠намики самопроизвольно (DGр < 0) протекают экзотермиче≠ские реакции (DЌр < 0) при любой температуре, если они сопро≠вождаютс€ увеличением энтропии (DSp > 0). Ёндотермические реакции (DЌр > 0), сопровождающиес€ уменьшением энтропии (DSp < 0), не могут протекать самопроизвольно ни при какой тем≠пературе, так как при этом DGр > 0.
Ѕиохимические реакции
Ѕиохимические реакции сопровождающиес€ уменьшением энергии √иббса (DGр < 0), называют экзэргоническими реак≠ци€ми; они могут совершатьс€ самопроизвольно. ≈сли в течение экзэргонической реакции энер≠ги€ √иббса только понижаетс€, как показано на рис. 1, то та≠ка€ реакци€ протекает самопроизвольно и не≠обратимо. „ем больше энерги€ √иббса биохимической системы в начальном состо€нии,чем в конечном состо€нии, тем больше разница химического сродства между исходными веществами и продуктами реакции в то есть их реакци≠онна€ способность.

–ис. 1. »зменение энергии √иб≠бса в закрытой системе
|
|
|
в необ≠ратимых экзэргонических реакци≠€х,
совершаемых самопроизвольно (р, “ = const)
ритерий DGp < 0 свиде≠тельствует только о термодина≠мической возможности протека≠ни€ данного процесса и ничего не говорит о его скорости и необходимых услови€х дл€ его начала.
Ќапример, окисление (горение) гра≠фита:
— + O2  —ќ2
—ќ2
—огласно положени€м термодинамики может происхо≠дить при стандартных услови€х самопроизвольно, так как 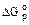 = -393,5 кƒж/моль. однако графит при температуре 298 с кислородом не реагирует, и чтобы реак≠ци€ произошла, необходимо создать определЄнные услови€ (запал подвоз энергии, введение катализатора).
= -393,5 кƒж/моль. однако графит при температуре 298 с кислородом не реагирует, и чтобы реак≠ци€ произошла, необходимо создать определЄнные услови€ (запал подвоз энергии, введение катализатора).
Ѕиохимические реакции, сопровождающиес€ увеличением энер≠гии √иббса (рис. 2), называютс€ эндэргоническими (DGp > 0), и они не могут происходить без посто€нного подвода энергии.
–ис. 2. »зменение энергии √иббса в закрытой системе
в необра≠тимых эндэргонических реакци€х
(р, “ = const)
Ќапример, процесс фотосинтеза в растени€х идЄт только под воздействием солнечной энергии:
6—ќ2 + 6Ќ2ќ  C6H12O6 + 6ќ2= DGp = 2870 кƒж/моль.
C6H12O6 + 6ќ2= DGp = 2870 кƒж/моль.
¬ живых системах эндэргонические реакции происход€т за счЄт их сопр€жени€ с экзэргоническими реакци€ми.
“аблица
р, “ = const
¬заимосв€зь термодинамических величин

| ||

| ||
’имическое равновесие
|
5. ѕ–»Ќ÷»ѕ ЁЌ≈–√≈“»„≈— ќ√ќ —ќѕ–я∆≈Ќ»я
Ѕ»ќ’»ћ»„≈— »’ –≈ј ÷»…
∆ива€ клетка дл€ своего существовани€ нуждаетс€ в энер≠гии. ѕри этом некоторые (гетеротрофные) клетки получают необходимую энергию в основном за счЄт окислени€ продуктов питани€, а дл€ некоторых (прототрофных клеток источником энергии часто €вл€етс€ солнечный свет. ѕолученна€ энер≠ги€ переводитс€ клетками обоих типов с довольно хорошим ѕƒ (~ 40 %) в химическую энергию за счЄт синтеза в них аденозинтрифосфорной кислоты (ј“‘):
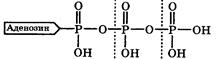
ј“‘ выполн€ет функцию аккумул€тора энергии, так как при его гидролизе (расщеплении водой), обра≠зуютс€ аденозиндифосфорна€ (јƒ‘) и ортофосфорна€ (‘) кислоты и выдел€етс€ энерги€. ѕоэтому ј“‘ называетс€ макроэргическим соединением, а разрывающа€с€ при его гидролизе св€зь ќЦ– - макроэргической.
ћакроэргической св€зью называет≠с€ химическа€ св€зь, при гидролизе которой выдел€етс€ значительна€ энерги€:
ј“‘ + Ќ2ќ Ѓ јƒ‘+ ‘, DGр = -30,5 кƒж/моль;
јƒ‘ + Ќ2ќ Ѓ јћ‘=  = -30,5 кƒж/моль;
= -30,5 кƒж/моль;
ј“‘ + 2Ќ2ќ Ѓ јћ‘ + 2‘,  = -61,0 кƒж/моль.
= -61,0 кƒж/моль.
–азрыв любой химической св€зи (в том числе и макроэр≠гической) требует затраты энергии. ¬ случае же гидро≠лиза ј“‘, кроме разрыва св€зей между фосфатными группами, (DG > 0), происход€т процессы гидрата≠ции (присоединение воды), изомеризации и нейтрализации кислотных продуктов, образующихс€ при гидролизе. ¬ результате этих процессов суммарное изменение энергии √иббса имеет отрицательное значение (∑∆G < 0). —ле≠довательно, макроэргическим €вл€етс€ не разрыв св€зей ќ-–, а энер≠гетический результат реакции гидролиза в целом.
ј“‘ функционирует в клетках как проме≠жуточный продукт, обеспечивающий организм энергией, необ≠ходимой дл€ протекани€ жизненно важных эндэргонических процессов: синтеза метаболитов (химическа€ работа), сокраще≠ни€ мышц (механическа€ работа), переноса вещества через мем≠браны против градиента концентрации (активный транспорт) и передачи информации (в частности, дл€ передачи нервных им≠пульсов).
ƒл€ протекани€ в живых системах эндэргониче≠ских реакций (DGp > 0), необходимо, чтобы они были сопр€жены с экзэргоническими реакци€ми (DGp < 0). “акое сопр€жение воз≠можно, если обе реакции имеют какое-либо общее промежуточ≠ное химическое соединение и на всех стади€х сопр€жЄнных реакций суммар≠ный процесс характеризуетс€ отрицательным значением измене≠ни€ энергии √иббса (Σ∆Gсопр€ж.р-ий 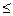 0).
0).
|
|
|
Ќапример, синтез сахарозы из глюкозы и фруктозы €вл€етс€ эндэргонической реакцией и самопроизвольно проис≠ходить не может:
√люкоза + ‘руктоза → —ахароза + Ќ2ќ, 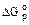 = 20,9 кƒж/моль
= 20,9 кƒж/моль
ќднако сопр€жение этой реакции с экзэргонической реакцией гидролиза ј“‘, сопровождающеес€ образованием общего про≠межуточного соединени€ глюкозо-1-фосфата, приводит к тому, что суммарный процесс имеет ∑DGр < 0:
ј“‘ + √люкоза + ‘руктоза → —ахароза + јƒ‘ + ‘,
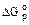 = -29,2 кƒж/моль.
= -29,2 кƒж/моль.
Ќар€ду с ј“‘ в живых организмах имеютс€ более эффек≠тивные макроэргические фосфорилированные соединени€, гид≠ролиз которых сопровождаетс€ выделением большей энергии. “ак, стандартна€ энерги€ √иббса дл€ гидролиза креатинфосфата, 3-фосфоглицерилфосфата и фосфоенолпирувата равна со≠ответственно -43,1, -49,4 и -61,9 кƒж/моль. — помощью этих соединений происходит синтез ј“‘ из јƒ‘, а јƒ‘ из јћ‘.
“аким образом, внутренним источником энергии в живых системах €вл€ютс€ фосфорилированные соединени€, при взаи≠модействии которых с биосубстратами, включа€ воду, выдел€≠етс€ энерги€. ¬ результате сопр€жени€ этих экзэргонических реакций с эндэргоническими реакци€ми обеспечиваетс€ протекание в клетке необходимых эндэргонических процессов.
6.ќ—ќЅ≈ЌЌќ—“» “≈–ћќƒ»Ќјћ» » Ѕ»ќ’»ћ»„≈— »’ ѕ–ќ÷≈——ќ¬ ¬ –ј¬Ќќ¬≈—Ќџ’ —“ј÷»ќЌј–Ќџ’ —ќ—“ќяЌ»я’.ѕќЌя“»я ќ √ќћ≈ќ—“ј«≈
ќтличительна€ особенность протекани€ обратимых биохимических реакций заключаетс€ в стремлении достичь динамического равновеси€, так как это состо€ние возникает и поддерживаетс€ вследствие протекани€ реакций в двух противоположных направ≠лени€х с одинаковыми скорост€ми 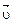 =
= 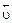 . “акое состо€ние назы≠ваетс€ химическим равновесием. ¬ этом случае изменение энергии √иббса в системе ха≠рактеризуетс€ наличием минимума, который соответствует со≠сто€нию химического равновеси€ (рис. 3, таблица). равновесному состо€нию возможен подход как со стороны исходных веществ (Δ
. “акое состо€ние назы≠ваетс€ химическим равновесием. ¬ этом случае изменение энергии √иббса в системе ха≠рактеризуетс€ наличием минимума, который соответствует со≠сто€нию химического равновеси€ (рис. 3, таблица). равновесному состо€нию возможен подход как со стороны исходных веществ (Δ  < 0), так и со стороны продуктов реакции (∆
< 0), так и со стороны продуктов реакции (∆ 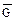 < 0).
< 0).
ѕри протекании обратимых реакций система самопроизвольно приходит к состо€нию хи≠мического равновеси€, из которого она без внешне≠го воздействи€ выйти не может, поскольку это тре≠бует увеличени€ энергии √иббса.
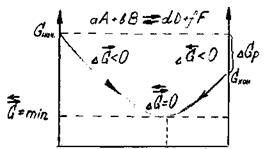
| »сходные вещества | –авновесный состав смеси исходных веществ и продуктов реакции | ѕродукты реакции |
–ис. 3. »зменение энергии √иббса в закрытой системе
в обратимой химической ре≠акции
(р, “ = const)
’имическое и биохимическое равновесное состо€ние сис≠темы характеризуетс€:
= равенством скоростей пр€мой и обратной реакций ( 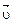 =
= 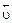 );
);
= энергетической выгодностью ( = min);
= min);
= отсутствием изменений величин параметров и функций состо€ни€ системы: концентрации реагентов, энтальпии, энтропии и энергии √иббса [(Δ 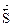 = ќ) (Δ
= ќ) (Δ 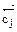 = 0) (Δ
= 0) (Δ  = 0) (ΔG =0)]
= 0) (ΔG =0)]
ѕоскольку в состо€нии химического равновеси€ система достигает минимально возможной энергии √иббса, то реакци€, котора€ приводит в данных услови€х к состо€нию равновеси€, всегда протекает самопроизвольно. Ѕлагодар€ этой особенности обратимых процессов большинство биохимических реакций, протекающих в организме, обратимы.
ƒруга€ особенность биохимических процессов, протекающих в организме, заключаетс€ в их многостадийности, так как ве≠ро€тность обратимого протекани€ отдельной стадии значитель≠но выше, чем процесса в целом (рис. 4). Ёто объ€сн€ет≠с€ тем, что разница между величинами (Gнач и Gкон дл€ каждой отдельной стадии обычно невелика (| ΔGp | < 10 кƒж/моль).
ќб≠ратимость отдельных стадий биохимических процессов позвол€≠ет живому организму легко регулировать синтез тех или иных химических соединений в зависимости от потребности и тем самым поддер≠живать стационарное состо€ние.
|
|
|
—тационарное состо€ние дл€ живого организма характери≠зуетс€ посто€нством его термодинамических величин и неиз≠менностью во времени скоростей поступлени€ и удалени€ ве≠ществ и энергии. Ќесмотр€ на посто€нство термодинамических величин, они не имеют равновесных значений в этом состо€нии. Ѕиологическое развитие организма возможно только в системе, наход€щейс€ в стационарном состо€нии, но далЄком от равнове≠си€. »менно стационарное неравновесное состо€ние живой ма≠терии позвол€ет ей опти≠мизировать свои характе≠ристики и эволюциониро≠вать во времени.
“ермодинамическа€ осо≠бенность стационарного со≠сто€ни€ открытых систем впервые сформулирована ». –. ѕригожиным (1946)*.
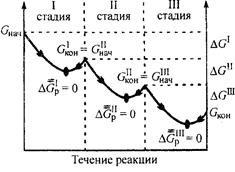
–ис. 4. »зменение энергии √иббса в многостадийном
биохимическом процессе
(р, “ = const)
¬ открытой системе в стационарном состо€нии при≠рост энтропии в единицу времени принимает ми≠нимальное положительное значение (ΔS / Δτ → min).
ѕоскольку энтропи€ €вл€етс€ мерой деградации, или рас≠се€ни€ энергии, принцип ѕригожина приводит к важнейшему заключению: при стационарном состо€нии рассе€ние энергии √иббса открытой системой оказываетс€ минимальным.
“ермодинамические особенности открытых систем, харак≠терные дл€ живого организма, объ€сн€ют его устойчивость, по≠звол€ющую ему в течение многих лет сохран€ть определЄнный уровень работоспособности, а также относительное посто€нство внутренней среды, называемое в биологии гомеостазом.
√омеостаз Ц относительное (!) динамическое посто€нство состава и свойств внутренней среды организма, обуслав≠ливающее устойчивость его физиологических функций.
¬ формировании и поддержании состо€ни€ гомеостаза боль≠шую роль играет обратимость большинства биохимических про≠цессов. Ёти процессы протекают самопроизвольно в на≠правлении достижени€ равновеси€, но, как правило, в орга≠низме они его не достигают, а только привод€т к достижению необходимого соотношени€ между конечными и исходными веществами реакции, протекающей самопроизвольно при данных услови€х. Ёто происходит или за счЄт использовани€ продуктов реакции, протекающей самопроизвольно в других процессах, или за счЄт изменени€ условий в данной системе. “ак, система, приближающа€с€ к химическому равновесию, переноситс€ ор≠ганизмом в другие услови€, при которых к состо€нию химиче≠ского равновеси€ приводит обратна€ реакци€. Ќа≠пример, в лЄгких, где концентраци€ кислорода больша€, гемо≠глобин крови соедин€етс€ с ним, но, не дос≠тигнув состо€ни€ равновеси€ в насыщении кислородом, кровь переноситс€ из лЄгких к ткан€м, и там гемоглобин отдаЄт ќ2, поскольку при переходе от лЄгких к ткан€м в крови из≠мен€ютс€ услови€ дл€ процесса взаимодействи€ гемоглобина с кислородом. ƒругой пример: формирование и рост костной тка≠ни происходит в одних клетках - остеобластах,* а еЄ растворение - в других клетках - остеокластах,** в то же врем€ ра≠бота клеток обоих типов регулируетс€ организмом, что позвол€≠ет ему поддерживать содержание костной ткани на определЄн≠ном уровне.
ќрганизм, таким образом, использует в своей жизнеде€тель≠ности обратимые биохимические процессы и их стремление к состо€нию химического равновеси€, но не допускает наступле≠ни€ устойчивого во времени химического равновеси€, так как это состо€ние приведЄт к гибели организма. ¬ то же врем€ со≠сто€ние гомеостаза поддерживаетс€ за счЄт баланса, то есть необхо≠димого соотношени€ между компонентами с противоположными (антагонистическими) свойствами. “ак, в основе гомеостаза орга≠низма наход€тс€ следующие химические и физико-химические балансы: кислотно-основный, окислительно-восстанови≠тельный, металло-лигандный, гидрофильно-липофильный, водно-электролитный. ¬ литературе пон€ти€ "баланс" и "гомеостаз" часто исполь≠зуютс€ как синонимы.
«аключение
ќсновные положени€, следующие из законов термодинамики:
= развитие системы происходит под вли€нием двух тенден≠ций =стремлени€ к минимуму энергии и к максимуму энтро≠пии;
= экзэргонические реакции в организме протекают самопро≠извольно, так как Δ G р < 0;
= эндэргонические реакции требуют подвода энергии, так как D G > р0;
= состо€ние равновеси€ в обратимых процессах с позиции термодинамики характеризуетс€ ∆  = 0 и €вл€етс€ энергетически самым выгодным, так как
= 0 и €вл€етс€ энергетически самым выгодным, так как  = min;
= min;
= биологические системы в стационарном состо€нии харак≠теризуютс€ отношением D S / Δτ → min, а в соответствии с законом сохранени€ суммой S + I = const дл€ живых систем должна быть справедлива зако≠номерность Δ I /Δτ → max (в пределах соблюдени€ приведЄнного закона сохранени€). ѕри этом биологические системы далеки от состо€ни€ равновеси€, что позвол€ет организмам оптимизиро≠вать свои характеристики и эволюционировать во времени.
|
|
|
«аконы биохимической термодинамики дают экологу биологу, и врачу ключ к пониманию энергетической стороны биохимических реакций в организме, процессов эмбриогенеза,* регенерации** и старени€ тканей, аналогичных процессов, проте≠кающих в биосфере, а также дают возможность регулировать эти процессы осуществлением профилактических или исправл€ю≠щих (лечебных) меропри€тий.
ќЌ“–ќЋ№Ќџ≈ «јƒјЌ»я
¬ариант 1
1.ѕри сгорании 1 л ацетилена C2H2 выдел€етс€ 58,0 кƒж теплоты. ¬ычислите стандартную энтальпию реакции окислени€ (горени€) ацетилена?
2.ѕри каком соотношении ∆H и T∆S системы наход€тс€ в равновесии?
¬ариант 2
1.¬ычислите тепловой эффект реакции восстановлени€ 1 мол€ оксида железа (III) алюминием.
2.Ќе производ€ вычислений, определите знак изменени€ энтропии дл€ процесса:
а) CO2(к)  CO2(г)
CO2(г)
б) 2NH3(г)  N2(г) + 3H2(г)
N2(г) + 3H2(г)
в) 2H2S(г) + 3O2(г) = 2SO2(г) + 2H2O(ж)
г) 2CH3OH(г) + 3O2(г) = 2CO2(г) + 4H2O(г)
¬ариант 3
1.ѕользу€сь табличными значени€ми стандартных энтальпий образовани€ реагирующих веществ, определите изменение стандартной энтальпии реакции
CaO + CO2 = CaCO3.
2.ѕочему при растворении в воде хлорида кали€ KCl энтропи€ увеличиваетс€, а при растворении газообразного диоксида углерода CO2 Ц уменьшаетс€?
¬ариант 4
- „то €вл€етс€ термодинамическим критерием химического равновеси€ системы:
а) ∆Hо = 0 б) ∆Gо < 0 в) р = const г) ∆Gо = 0
- ќпределите теплоту образовани€ оксида азота (I), исход€ из уравнений реакций:
C + 2N2O  CO2 + 2N2, ∆Hо = -556,9 кƒж;
CO2 + 2N2, ∆Hо = -556,9 кƒж;
C + O2  CO2, ∆Hо = -393,7 кƒж.
CO2, ∆Hо = -393,7 кƒж.
¬ариант 5
- ¬ каком случае возможно самопроизвольное протекание химической реакции при стандартных услови€х, если ∆Hо >> T∆Sо?
а) ∆Hо > 0, ∆Sо < 0 б) ∆Hо > 0, ∆Sо < 0
в) ∆Hо < 0, ∆Sо > 0 г) ∆Hо < 0, ∆Sо < 0
ƒайте подробный ответ.
- „ем отличаютс€ термохимическа€ и термодинамическа€ системы знаков дл€ тепловых эффектов процессов и почему?
¬ариант 6
- »сход€ из теплот образовани€ жидкой воды и вод€ного пара, определите теплоту испарени€ воды.
- –ассчитайте температуру, при которой равноверо€тны оба направлени€ реакции
CuO(к) + C(графит)  Cu(к) + CO(г)
Cu(к) + CO(г)
¬ариант 7
- ¬ычислите стандартную энтальпию образовани€ метилового спирта, исход€ из следующих данных:
H2(г) + 1/2 O2(г) = H2O(ж), ∆H0 = -285,8 кƒж;
C(гр) + O2(г) = CO2(г), ∆H0 = -393,7 кƒж;
CH3OH(ж) + 3/2 O2(г) = CO2(г) + 2 H2O(ж) ∆H0 = -715,0 кƒж.
- ћожет ли протекать реакци€ самопроизвольно при положительном изменении энтальпии и изменении энтропии?
¬ариант 8
- “еплота образовани€ октана C8H18 равна 199,28 кƒж/моль. ќпределите теплоту сгорани€ 1 кг октана.
(ќбразующуюс€ при сгорании воду считать жидкой).
- акую информацию о веществе можно получить, использу€ значение энергии √иббса?
¬ариант 9
- –ассчитайте термодинамические характеристики процесса термического разложени€ карбоната кальци€ —а—ќ3.
- ѕочему в уравнении дл€ вычислени€ ∆G перед энтропийным фактором стоит знак минус?
ѕочему этот фактор выражаетс€ произведением изменени€ энтропии на температуру?
¬ариант 10
1. ѕри нейтрализации гидроксида натри€, какой массы хлороводородом по реакции
NaOH(к) + HCl(г) = NaCl(к) + H2O(ж)
выдел€етс€ 352,9 кƒж теплоты?
2. –ассчитайте температуру, при которой равноверо€тны оба направлени€ реакции
2 SO2(г) + O2(г)  2 SO3(г).
2 SO3(г).
¬ариант 11
- –ассчитайте, €вл€етс€ ли реакци€
Al2(SO4)3(к) = Al2O3(к) + 3SO3(г)
экзо- или эндотермической.
- ќпределите, рассчитав DS, возможность протекани€ реакции
2C(графит) + H2(г) = C2H2(г).
¬ариант 12
- ќпределите знаки ∆Hо, ∆Sо и ∆Gо дл€ реакции
AB(к) + B2(г)  AB3(гр),
AB3(гр),
протекающей при температуре 298 в пр€мом направлении.
Ѕудет ли ∆Gо возрастать или убывать с ростом температуры?
- ƒл€ реакций
Ќ2(г) + ќ (г) = Ќ2ќ(г) (1)
Ќ2(г) + 1/2ќ2(г) = Ќ2ќ(г) (2)
2Ќ(г) + ќ (г) = Ќ2ќ(г) (3)
укажите правильное соотношение стандартных изменений энтальпий:
а) 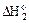 <
<  <
<  б)
б) 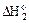 >
>  >
> 
¬ариант 13
- ¬ каком из случаев:
а) ∆H < 0, ∆S > 0; б) ∆H < 0, ∆S < 0 или в) ∆H > 0, ∆S > 0
реакци€ возможна при любых температурах?
ќтвет обоснуйте.
- акой из оксидов Ц CuO или MgO Ц обладает наибольшим химическим сродством к серному ангидриду SO3 ?
ќтвет подтвердите расчЄтом значений ∆Gо реакций.
¬ариант 14
- —колько теплоты выделитс€ при сгорании 20 кг ацетилена C2H2 при стандартных услови€х?
- ≈сли ∆H < 0 и ∆S < 0, то в каком из случаев:
а) ∆H > T ∆S или б) ∆H < T ∆S
реакци€ окислени€ (горение) ацетилена может протекать самопроизвольно?
¬ариант 15
- »сход€ из теплот реакций окислени€ оксида мышь€ка (III):
As2O3 + O2 = As2O5 (∆H0 = Ц271 кƒж),
3As2O3 + 2O3 = 3As2O5 (∆H0 = Ц1096 кƒж),
определите теплоту образовани€ озона из молекул€рного кислорода.
- ¬ каком из случаев
а) ∆H > 0, ∆S > 0, б) ∆H > 0, ∆S < 0 или в) ∆H < 0, ∆S < 0
реакци€ неосуществима при любых температурах?
ќтвет обоснуйте.
“»ѕќ¬џ≈ “≈—“ќ¬џ≈ «јƒјЌ»я
1. “еплосодержание энтальпи€ системы, в которой протекает экзотермическа€ реакци€, в начальном состо€нии Е.
1) больше, чем в конечном
2) меньше, чем в конечном
3) одинаково с конечным
2. ƒл€ эндотермической реакции энтальпи€ в начальном состо€нии Е.
1) больше, чем в конечном
2) така€ же, как конечном
3) меньше, чем в конечном
3. –еакци€ экзотермическа€, если Е.
1) ΔЌ = 0 2) ΔЌ < 0 3) ΔЌ > 0 4)
4. Ёкзотермическа€ реакци€ выражаетс€ термохимическим уравнением Е.
1) ј + ¬ = —, ΔЌ > 0
2) ј + ¬ + Q = —
3) ј + ¬ = —, ΔЌ < 0
4) ј + ¬ = — Ц Q
5. оличество теплоты, поглощаемое при разложении 0,5 моль оксида углерода (IV) по термохимическому уравнению реакции
—ќ2 (г) = — (т) + ќ2 (г) Ц 393,4 кƒж,
равно Е кƒж.
1) 98,35 2) 196,7 3) 786,8 4) 8,96
6. “еплота (энтальпи€) образовани€ сложного вещества равна Е.
1) теплоте сгорани€ этого вещества
2) теплоте, котора€ выдел€етс€ или поглощаетс€ при образовании 1 моль вещества
3) теплоте разложени€ этого вещества
4) теплоте, котора€ выдел€етс€ или поглощаетс€ при образовании 1 моль этого вещества из простых веществ
7. —тандартна€ энтальпи€ образовани€ оксида серы (IV) равна тепловому эффекту реакции Е.
1) —u2S (к) + ќ2 (г) = 2—u (к) + Sќ2 (г)
2) S (к) + 2ќ (г) = Sќ2 (г)
3) Ќ2Sќ3 (ж) = Ќ2ќ (ж) + Sќ2 (г)
4) S (к) + ќ2 (г) = Sќ2 (г)
8. ѕон€тие "теплота образовани€ сложного вещества" относитс€ к Е.
1) 1 г вещества 2) любому количества вещества
3) 1 моль вещества 4) 100 г вещества
9. ѕри стандартных услови€х теплота образовани€ Е равна нулю.
1) ќ3 (г) 2) Sќ2 (г) 3) ќ2 (г) 4) —аќ (т)
10. “епловой эффект реакции
јl2ќ3 + 3Sќ3 = јl2(Sќ4)3
рассчитывают по уравнению Е.
1) ΔЌ0 = ΔЌ0298, јl2(Sќ4)3 + ΔЌ0298, јl2ќ3 + 3ΔЌ0298, Sќ3
2) ΔЌ0 = ΔЌ0298, јl2ќ3 + 3ΔЌ0298, Sќ3 Ц ΔЌ0298, јl2(Sќ4)3
3) ΔЌ0 = ΔЌ0298, јl2(Sќ4)3 Ц ΔЌ0298, јl2ќ3 Ц ΔЌ0298, Sќ3
4) ΔЌ0 = ΔЌ0298, јl2(Sќ4)3 Ц ΔЌ0298, јl2ќ3 Ц 3ΔЌ0298, Sќ3
11. “епловой эффект реакции
4NЌ3 (г) + 3ќ2 (г) = 2N2 (г) + 6Ќ2ќ (г)
(ΔЌ0298, кƒж/моль: Ц46,2 0 0 Ц286)
равен Е кƒж.
1) Ц1531,2 2) Ц239,8 3) Ц1669,8 4) 1900,8
12. »зменение энтропии в химической реакции равно Е.
1) сумме энтропий продуктов реакции
2) разности сумм энтропий исходных веществ и продуктов реакции
3) сумме энтропий исходных веществ
4) разности сумм энтропий продуктов реакции и исходных веществ
13. Ёнтропи€ идеально построенного кристалла при “ → 0 стремитс€ к Е.
1) Ц ∞ 2) 0 3) + ∞ 4) Ц 273
14. Ёнтропи€ увеличиваетс€ в процессе Е.
1) I2 (к) = I2 (г) 2) Sќ3 (г) + Ќ2ќ (ж) = Ќ2Sќ4 (ж)
3) Ќ2ќ (г) = Ќ2ќ (к) 4) N2 (г) + ќ2 (г) = 2Nќ (г)
15. ”величение энтропии происходит в р€ду превращений Е.
1) Ќ2ќ (г) → Ќ2ќ (ж) → Ќ2ќ (т)
2) —ќ2 (г) → —ќ2 (т) → —ќ2 (г)
3) I2 (т) → I2 (ж) → I2 (г)
4) ќ2 (г) → ќ2 (ж) → ќ2 (г)
16. ƒл€ реакций
4Fе (к) + 3ќ2 (г) = 2Fе2ќ3 (к);
Ќ2 (г) + —l2 (г) = 2Ќ—l (г)
энтропи€ соответственно Е.
1) увеличиваетс€; практически не измен€етс€
2) уменьшаетс€; увеличиваетс€
3) увеличиваетс€; увеличиваетс€
4) уменьшаетс€; практически не измен€етс€
17. Ёнерги€ √иббса (изобарно-изотермический потенциал) определ€етс€ соотношением Е.
1) G = Ќ + “S 2) G = S + “Ќ
3) G = Ќ Ц “S 4) G = S Ц “Ќ





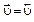
 = 0
= 0

