–асчет навески Ц g
g = CN Х ћЁ Х V (л) = 1 Х 74 Х 0,1 = 7,4 г
ћ = 35 + 39 = 74 г
ћЁ = ћ Х fЁ = 74 Х 1 = 74 г/ћоль
2. ѕриготовление запасного стандартного раствора Mg2+: ѕриготовьте 100 мл 1,0 ћ раствора хлорида магни€ MgCl2. Ќавеску (20,3303±0,0010) г MgCl2Х7H2O (проверьте расчетами правильность навески), предварительно высушенную до посто€нной массы при 1100 —, взвесьте на ј¬ и растворите в мерной колбе в дистиллированной воде.
–асчет навески Ц g
g = CN Х ћЁ Х V (л) = 1 Х 213 Х 0,1 = 21,3 г
ћ = 94 + 119 = 213 г
ћЁ = ћ Х fЁ = 213 Х 1 = 213 г/ћоль
3. ѕриготовление рабочих стандартных растворов хлорида магни€ с концентрацией Mg2+ 1,0Х10-1 Ц 1,0Х10-6 ћ: ѕриготовьте серию стандартных растворов 1,0Х10-1 Ц 1,0Х10-6 ћ Mg2+ по 50 мл каждый. –астворы готов€тс€ из запасного стандартного раствора (см. п. 2) методом последовательного разбавлени€. аждый последующий раствор готов€т разбавлением предыдущего в мерных колбах на 50 мл согласно алгоритму, предложенному в таблице 4.
“аблица 4
| Ќаименование компонента | оличество раствора, мл | |||||
| Ќомер эталонного раствора (пробы) | ||||||
| —тандартный раствор с концентрацией Mg 2+: а) 1,0ћ | 5,0 | |||||
| б) 1,0Х10-1ћ | 5,0 | |||||
| в) 1,0Х10-2ћ | 5,0 | |||||
| г) 1,0Х10-3ћ | 5,0 | |||||
| д) 1,0Х10-4ћ | 5,0 | |||||
| е) 1,0Х10-5ћ | 5,0 | |||||
| 1,0 ћ Cl | ѕо 5 мл в каждую мерную колбу | |||||
| Ќ2ќ дист | ƒовести в мерных колбах до метки | |||||
| онцентраци€ Mg 2+, моль/л | 1,0Х10-1 | 1,0Х10-2 | 1,0Х10-3 | 1,0Х10-4 | 1,0Х10-5 | 1,0Х10-6 |
| рMg = -lg[Mg 2+] | 1,00 | 2,00 | 3,00 | 4,00 | 5,00 | 6,00 |
4. алибровка (настройка) магний селективного электрода по стандартным растворам (смотрите паспорт электрода). ѕолученные стандартные растворы (см. п.3) налейте в чистые стаканчики и, погрузив в раствор магний-селективный индикаторный электрод и хлорид-серебр€ный электрод сравнени€, измерьте Ёƒ— €чейки (≈, м¬). »змерени€ проводите, переход€ от раствора с меньшей концентрацией к раствору с более высокой концентрацией (в этом случае электроды можно не мыть, а лишь вытирать фильтровальной бумагой). ѕравила работы на потенциометре смотрите в паспорте прибора. ѕолученные данные занесите в табл. 5, обработайте их математически ћЌ .
“аблица 5
| є п/п | рMg | ≈, м¬ измеренна€ | ≈, м¬ рассчитанна€ | ≈ рассчит.= = a + в рMg |
| 1,0 2,0 3,0 4,0 5,0 6,0 | -361,4 -394,7 -416,2 -431,1 -445,3 -447,3 | -373,41429 -390,44858 -407,48287 -424,51716 -441,55145 -458,58574 | r = 0,96077 а = -356,38000 в = -17,03429 n = 6 |
«аполните табл. 5 до конца и постройте (на миллиметровой бумаге) калибровочный график ≈ = ƒ (рMg). —равните экспериментально полученное значение крутизны электродной функции электрода (в) с паспортными данными электрода и оцените его качество.
|
|
|
5. јнализ воды: а) 1. ¬озьмите в мерную колбу на 50 мл аликвоту воды 5 мл, добавьте 5 мл 1ћ Cl, перемешайте и доведите до метки дистиллированной водой. ѕерелейте полученный раствор в чистый стаканчик и измерьте Ёƒ—, как это делали при калибровке электрода. ѕо калибровочному графику определите рMg.
6. –асчеты: –ассчитайте —ћ Mg2+ в воде (моль/л), зна€, что рMg = -lg[Mg2+]. ѕересчитайте —ћ на массовую концентрацию магни€, мг/л, по формуле:
— Mg2+, мг/л = —ћ ХћХ1000 = 24,3050 Х 10 -4,98524Х 1000 = 0,251435225,
— пересчетом на разбавление: 2,51
где —ћ Ц мол€рна€ концентраци€ магни€, рассчитанна€ через рMg; ћ Ц мол€рна€ масса магни€, г/моль, найденна€ по справочнику Ћурье; 1000 Ц коэффициент пересчета с г на мг.
–асчет абсолютной и относительной погрешностей определени€
јбсолютна€ погрешность (ошибка):
Dабс. = ’практ. Ц ’ист = 2,51 Ц 24,31 = -21,8.
ќтносительна€ погрешность (ошибка):
Dотн. = çDабс.ç *100% / ’ист = 21,8*100 / 24,31 = 89,7.
Ѕ) —пособом добавок. 1. ¬озьмите в мерную колбу на 50 мл аликвоту воды 5 мл, добавьте 5 мл 1,0 ћ Cl, перемешайте и доведите до метки дистиллированной водой. 40 мл полученного раствора налейте в чистый стакан вместимостью 100 мл и измерьте Ёƒ— (≈1), как это делали при калибровке электрода.
2. «атем добавьте в него 1 мл стандартного раствора хлорида магни€ с концентрацией MgSO4 0,05 ћ и измерьте Ёƒ— (≈2).
“аблица 6
| є п/п | ќбъем анализируемого раствора, Vх, мл | онцентраци€ Mg2+ в добавл€емом растворе, —ст., моль/л | ќбъем добавки, Vст., мл | Ёƒ— (≈1) | Ёƒ— (≈i) | онцентраци€ Mg2+ в анализируемом растворе, —х, моль/л |
| 1,0Х10-2 | -443,8 | -443,8 | ||||
| -439,0 | 0,0002634 | |||||
| -436,2 | 0,000265 | |||||
| -433,9 | 0,0002441 | |||||
| -432,6 | 0,000209 | |||||
| -431,2 | 0,0002444 |
3. ѕовторите п. 2 четыре раза, добавл€€ в стакан с анализируемой смесью каждый раз дополнительно по 1 мл стандартного раствора. ¬с€кий раз измер€йте Ёƒ— (≈i). ƒанные измерений занесите в табл. 6.
4. –асчеты: –ассчитайте концентрацию ионов магни€ в растворе по формулам:
Ј с учетом разбавлени€ 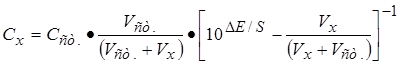 ; (смотри таблица 6)
; (смотри таблица 6)
Ј без учета разбавлени€  :
:
—х1 = 0
—х2 = 0,000275
—х3 = 0,0001375
—х4 = 0,00009
—х5 = 0,00007
—х6 = 0,0000125
√де —х Ц концентраци€ ионов магни€ в анализируемом растворе, моль/л; —ст. Ц концентраци€ ионов магни€ добавл€емого стандартного раствора, моль/л; ∆≈ Ц разность потенциалов ≈i Ц ≈1, м¬; S Ц крутизна электродной функции, м¬.
—равните два способа анализа по воспроизводимости и правильности. ак это сделать?






