406. ¬нутри периодов слева направо размеры атомов уменьшаютс€. Ёто происходит вследствие усилени€ взаимодействи€ положительно зар€женного €дра с отрицательно зар€женными электронами по мере заполнени€ ими одного и того же электронного сло€. ѕо группам сверху вниз радиус атома увеличиваетс€ из-за добавлени€ новых электронных слоев. —оответственно наименьший радиус имеет атом гели€, а наибольший - атом франци€. ѕри увеличении радиуса атома усиливаетс€ металличность элемента, уменьшаетс€ его электроотрицательность, сродство к электрону.
407. ќрбитальный радиус - радиус атома измер€емый от центра €дра до положени€ главного максимума плотности внешних электронных облаков. овалентный радиус - радиус атома в молекуле, построенной по типу ковалентной св€зи. ћеталлический радиус - радиус атома в металлической решетке. Ёффективный радиус - практический радиус атома, св€занного с другими атомами. ¬нутри периодов слева направо размеры атомов уменьшаютс€. Ёто происходит вследствие усилени€ взаимодействи€ положительно зар€женного €дра с отрицательно зар€женными электронами по мере заполнени€ ими одного и того же электронного сло€. ѕо группам сверху вниз радиус атома увеличиваетс€ из-за добавлени€ новых электронных слоев. ѕри увеличении радиуса атома усиливаетс€ металличность элемента, уменьшаетс€ его электроотрицательность, сродство к электрону.
408. »зменение ионизационного потенциала обратно пропорционально изменению атомного радиуса. ¬нутри периодов слева направо размеры атомов уменьшаютс€. Ёто происходит вследствие усилени€ взаимодействи€ положительно зар€женного €дра с отрицательно зар€женными электронами по мере заполнени€ ими одного и того же электронного сло€. ѕо группам сверху вниз радиус атома увеличиваетс€ из-за добавлени€ новых электронных слоев. “.е. по периодам ионизационный потенциал увеличиваетс€, а по группам - уменьшаетс€. ѕри уменьшении ионизационного потенциала усиливаетс€ металличность элемента, уменьшаетс€ его электроотрицательность, сродство к электрону.
409. »онизационный потенциал и энерги€ ионизации - это количественные характеристики энергии отрыва электрона от атома. »онизационный потенциал (»ѕ) измер€етс€ в э¬, а энерги€ ионизации (Ё») в кƒж/моль. »ѕ(Li) = 5.19 э¬, следовательно, Ё»(Li) = 5.19*96486 = 500 кƒж/моль. Ё»(ќ) = 1313.0 кƒж/моль, следовательно, »ѕ(ќ) = 1313.0/96.486 = 13.6 э¬. –адиус атома кислорода значительно меньше радиуса атома лити€. —оответственно, взаимодействие внешних электронов с €дром в атоме кислорода выше, а значит выше и »ѕ кислорода. ћожно сделать следующий вывод: чем выше »ѕ, тем сильнее выражены неметаллические свойства элемента.
410. Ќа внешней орбите у атома берилли€ наход€тс€ два электрона, близкие по энергии. —оответственно ионизационные потенциалы дл€ этих электронов близки. ¬ противоположность этому, первый и второй электроны атома лити€ расположены в различных электронных сло€х. », если первый электрон отрываетс€ относительно легко, то второй электрон, наход€щийс€ гораздо ближе к €дру, оторвать значительно сложнее. ќтсюда и разница в ионизационных потенциалах.
|
|
|
411. јтомы типичных металлов отличаютс€ от атомов типичных неметаллов значительно большими радиусами, а значит у металлов меньше энерги€ ионизации, электроотрицательность и сродство к электрону. ћеталлы в большинстве - твердые (при н.у.) вещества, проводники, ковкие, пластичные и т.п. ¬ соединени€х металлы имеют только положительные степени окислени€, в то врем€ как неметаллы имеют как отрицательные, так и положительные —ќ. јмфотерные металлы, в отличие от типичных металлов, могут про€вл€ть основные и кислотные свойства как в свободном состо€нии, так и в соединени€х. ћеталлические свойства завис€т от радиуса атома. „ем больше радиус атома, тем сильнее выражены металлические свойства элемента. ¬нутри периодов слева направо размеры атомов уменьшаютс€. Ёто происходит вследствие усилени€ взаимодействи€ положительно зар€женного €дра с отрицательно зар€женными электронами по мере заполнени€ ими одного и того же электронного сло€. ѕо группам сверху вниз радиус атома увеличиваетс€ из-за добавлени€ новых электронных слоев. “.е. с увеличением пор€дкового номера по периодам металличность уменьшаетс€, а по группам увеличиваетс€. Ќеметаллы - хлор, кислород, гелий, кремний, углерод. ћеталлы - франций, натрий, уран, магний. јмфотерные металлы - алюминий, германий, олово, цинк.
412. ћеталлические свойства завис€т от радиуса атома. „ем больше радиус атома, тем сильнее выражены металлические свойства элемента. ¬нутри периодов слева направо размеры атомов уменьшаютс€. Ёто происходит вследствие усилени€ взаимодействи€ положительно зар€женного €дра с отрицательно зар€женными электронами по мере заполнени€ ими одного и того же электронного сло€. ѕо группам сверху вниз радиус атома увеличиваетс€ из-за добавлени€ новых электронных слоев. “.е. по периодам металличность уменьшаетс€, а по группам увеличиваетс€.
413. —родством к электрону называетс€ энерги€, выдел€юща€с€ при присоединении электрона к электронейтральному атому. — ростом пор€дкового номера элемента сродство к электрону по периодам растет, а по группам уменьшаетс€. ћинимальное сродство к электрону имеют щелочные металлы и благородные газы. ћаксимальное сродство к электрону у галогенов.
414. ќтносительна€ электроотрицательность (ќЁќ) - условна€ оценка способности атома данного элемента отт€гивать на себ€ электронную плотность по сравнению с атомами других элементов в соединении. — ростом пор€дкового номера элемента ќЁќ по периодам растет, а по группам уменьшаетс€. ћинимальную ќЁќ имеет франций, а максимальна€ ќЁќ у атома фтора.
415. ћаксимальна€ валентность химических элементов зависит то количества внешних электронов. ѕо периоду число валентных электронов растет - соответственно увеличиваетс€ максимальна€ валентность. ѕо группам значение максимальной валентности не измен€етс€, т.к. не добавл€етс€ валентные электроны.
416. ≈сли иметь в виду только s- и p-элементы, то дл€ их оксидов по группам сверху вниз и по периодам справа налево усиливаютс€ основные свойства и ослабевают кислотные свойства. Ёто происходит вследствие соответствующего изменени€ радиусов атомов центральных элементов в оксидах: чем больше радиус элемента, тем сильнее выражены его металлические свойства, тем более основными свойствами обладает его оксид.
ѕример є1 (по третьему периоду):
Na2O + 2HCl = 2NaCl + H2O; Na2O + KOH = реакци€ не протекает.
|
|
|
Al2O3 + 6HCl = 2AlCl3 + 3H2O; Al2O3 + 6KOH =2K3AlO3 + 3H2O
Cl2O7 + HCl = реакци€ не протекает; Cl2O7 + 2KOH = 2KClO4 + H2O.
ѕример є2 (по третьей группе):
B2O3 + HCl = реакци€ не протекает; B2O3 + 6KOH = 2K3BO3 + 3H2O.
Al2O3 + 6HCl = 2AlCl3 + 3H2O; Al2O3 + 6KOH =2K3AlO3 + 3H2O
In2O3 + 6HCl = 2InCl3 + 3H2O; In2O3 + KOH = реакци€ не протекает.
417. ≈сли иметь в виду только s- и p-элементы, то дл€ их гидроксидов по группам сверху вниз и по периодам справа налево усиливаютс€ основные свойства и ослабевают кислотные свойства. Ёто происходит вследствие соответствующего изменени€ радиусов атомов центральных элементов в оксидах: чем больше радиус элемента, тем сильнее выражены его металлические свойства, тем более основными свойствами обладает его гидроксид.
ѕример є1 (по третьему периоду):
NaOH + HCl = NaCl + H2O; NaOH + KOH = реакци€ не протекает
Al(OH)3 + 3HCl = AlCl3 + 3H2O; Al(OH)3 + 3KOH =K3[Al(OH)6]
HClO4 + HCl = реакци€ не протекает; HClO4 + KOH = KClO4 + H2O
ѕример є2 (по третьей группе):
B(OH)3 + HCl = реакци€ не протекает; B(OH)3 + 3KOH = K3BO3 + 3H2O
Al(OH)3 + 3HCl = AlCl3 + 3H2O; Al(OH)3 + 3KOH =K3[Al(OH)6]
In(OH)3 + 3HCl = InCl3 + 3H2O; In(OH)3 + KOH = реакци€ не протекает.
Ё2O3 + 6HCl = 2ЁCl3 + 3H2O.
418. ѕри переходе от одного периода к другому свойства оксидов и гидроксидов d-элементов мен€ютс€ незначительно, либо остаютс€ практически неизменными. “ак, в р€ду оксидов и гидроксидов сканди€ - иттри€ - лютеци€ наблюдаютс€ осн о вные свойства: Ё2ќ3 + ќЌ = реакци€ не протекает. ” соответствующих соединений цинка - кадми€ - ртути свойства измен€ютс€ от амфотерных до осн о вных. ¬ HCl = реакци€ не протекает; Ё2O7 + 2KOH = 2KЁO4 + H2O.
419. ƒиагональным сходством называетс€ сходные свойства элементов наход€щихс€ на одной р€ду оксидов и гидроксидов марганца - технеци€ - рени€ наблюдаютс€ кислотные свойства: Ё2O7 + диагонали в периодической таблице. ѕричиной этого эффекта €вл€етс€ приблизительное равенство радиусов атомов соответствующих элементов. ѕримером диагонального сходства может служить идентичность некоторых свойств лити€ и магни€: металлы, относительно хорошие комплексообразователи, образуют одинаково белые соли со значительной степенью ионности св€зи и т.п.
420. ѕолными электронными аналогами называютс€ элементы, которые в любой степени окислени€ имеют подобную структуру внешнего и предвнешнего электронного уровн€. Ќеполные электронные аналоги имеют подобную структуру внешнего и предвнешнего электронного уровн€ только в максимальной степени окислени€. —ходство: в максимальной степени окислени€ полные и неполные электронные аналоги образуют химические соединени€ подобного стехиометрического состава. Ёти соединени€ имеют схожие химические свойства (кислотность, основность, активность). ¬ промежуточных степен€х окислени€ полные и неполные электронные аналоги образуют сильно отличающиес€ друг от друга соединени€.
’имические элементы называютс€ типическими, если они обладают свойствами, позвол€ющими отнести их только к какому-либо одному классу элементов. »менно отсутствие двойственности свойств, амфотерности отличает типические элементы от нетипических.
421. —в€зь называетс€ ковалентной непол€рной, если она осуществл€етс€ между атомами с одинаковой относительной электроотрицательностью (ќЁќ). ≈сли ќЁќ атомов, образующих св€зь различаетс€ менее, чем на 2.1, то така€ св€зь называетс€ ковалентной пол€рной. ¬ случае, если разница в ќЁќ больше, чем 2.1, то св€зь называетс€ ионной. —в€зь, образованна€ атомами металлов называетс€ металлической.
| јзот (N2) | овалентна€ непол€рна€ |
| ∆елезо (Fe) | ћеталлическа€ |
| ”глекислый газ (CO2) | овалентна€ пол€рна€ |
| ‘торид кали€ (KF) | »онна€ |
| ‘осфин (PH3) | овалентна€ пол€рна€ |
| —ульфат натри€ (Na2SO4) | »онна€ + пол€рна€ |
| ремний (Si) | Ќепол€рна€ |
422. —в€зь называетс€ ковалентной непол€рной, если она осуществл€етс€ между атомами с одинаковой относительной электроотрицательностью (ќЁќ). ≈сли ќЁќ атомов, образующих св€зь различаетс€ менее, чем на 2.1, то така€ св€зь называетс€ ковалентной пол€рной. ¬ случае, если разница в ќЁќ больше, чем 2.1, то св€зь называетс€ ионной. —в€зь, образованна€ атомами металлов называетс€ металлической.
|
|
|
| »одоводород (HI) | овалентна€ пол€рна€ |
| Ќикель (Ni) | ћеталлическа€ |
| ќксид кремни€ (SiO2) | овалентна€ пол€рна€ |
| ќксид магни€ (MgO) | овалентна€ пол€рна€ |
| Ќитрат кали€ (KNO3) | »онна€ + пол€рна€ |
| ‘тор (F2) | овалентна€ непол€рна€ |
423. ’имическа€ св€зь - взаимодействие атомов, обусловливающее образование химически устойчивой двух- или многоатомной системы. “ри основные типа химической св€зи: ковалентна€, ионна€, металлическа€.
| “ип химической св€зи | јтомы каких элементов вступают в св€зь | ѕроцессы в электронных оболочках атомов | ќбразующиес€ частицы и вещества | ѕримеры |
| овалентна€ | Ћюбых | ќбразование электронных пар. Ќезначительна€ деформаци€ | ћолекулы, кристаллы | N2, H2O, FeCl3 |
| »онна€ | Ћюбых | ќбразование электронных пар, сильно прит€нутых к одному из атомов. «начительна€ деформаци€ оболочек | ћолекулы, кристаллы имеющие на некоторых атомах частичные зар€ды (+ и -) | NaF, KCl, CsF |
| ћеталлическа€ | јтомы металлов | ƒелокализаци€ валентных электронов. | ћеталлическа€ решетка, в узлах которой наход€тс€ атомы металлов | Fe, Ni, Cu. |
424. ¬ р€ду HF - HCl - HBr - HI происходит увеличение длины св€зи вследствие увеличени€ радиуса атомов галогенов в р€ду F - Cl - Br - I, поскольку происходит добавление новых электронных слоев.
425. ¬ р€ду H2O- H2S-H2Se-H2Te происходит увеличение длины св€зи вследствие увеличени€ радиуса атомов элементов в 6 группе от кислорода к теллуру вследствие добавлени€ электронных слоев.
426. ¬ р€ду —2Ќ6-—2Ќ4-—2Ќ2 происходит уменьшение длины св€зи, т.к. между атомами углерода происходит увеличение кратности св€зи. “ак, у этана кратность св€зи равна 1, у этилена -2, у ацетилена -3.
427. ¬ р€ду HF - HCl - HBr - HI происходит уменьшение энергии св€зи, поскольку увеличиваетс€ радиус атома галогенов от фтора к йоду. „ем больше радиус атома, тем меньше нужно затратить энергии, чтобы разрушить молекулу.
428. ¬ р€ду —F4 - —Cl4 - —Br4 - —I4 происходит уменьшение энергии св€зи, поскольку увеличиваетс€ радиус атома галогенов от фтора к йоду. „ем больше радиус атома, тем меньше нужно затратить энергии, чтобы разрушить молекулу.
429. ¬ р€ду —2Ќ2 - —2Ќ4 - —2Ќ6 происходит уменьшение энергии св€зи, т.к. между атомами углерода происходит уменьшение кратности св€зи. “ак, у этана кратность св€зи равна 1, у этилена -2, у ацетилена -3. „ем больше кратность св€зи, тем прочнее молекула, тем больше нужно затратить энергии дл€ ее разрушени€.
430. ¬алентным углом называетс€ угол между лини€ми св€зи (лини€ св€зи - лини€, проход€ща€ через центры €дер атомов) 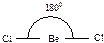 ,
, 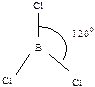 ,
, 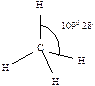 .
.
431. “еори€ валентных св€зей исходит из положени€, что кажда€ пара атомов в молекуле удерживаетс€ вместе при помощи одной или нескольких общих электронных пар. “.е. химическа€ св€зь локализована между двум€ атомами: она двуцентрова€ и двухэлектронна€.
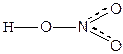 јтом хлора имеет один неспаренный электрон, соответственно между двум€ атомами хлора и между хлором и водородом может образоватьс€ только одна св€зь за счет образовани€ одной обобществленной электронной пары: Cl 3s2 3p5, H 1s1 => —l-Cl, H-Cl. ¬алентные возможности атома азота ограничены трем€ неспаренными электронами, поэтому с атомами кислорода в молекуле азотной кислоты азот св€зан как бы полуторными св€з€ми. —техиометрическа€ валентность азота равна 5, степень окислени€ = +5, электронна€ валентность равна 4, кратность св€зей 1.5.
јтом хлора имеет один неспаренный электрон, соответственно между двум€ атомами хлора и между хлором и водородом может образоватьс€ только одна св€зь за счет образовани€ одной обобществленной электронной пары: Cl 3s2 3p5, H 1s1 => —l-Cl, H-Cl. ¬алентные возможности атома азота ограничены трем€ неспаренными электронами, поэтому с атомами кислорода в молекуле азотной кислоты азот св€зан как бы полуторными св€з€ми. —техиометрическа€ валентность азота равна 5, степень окислени€ = +5, электронна€ валентность равна 4, кратность св€зей 1.5.
432. »зменение валентности атома в большинстве случаев происходит за счет возбуждени€, при котором распариваетс€ электронна€ пара, образу€ два неспаренных электрона. “.е. валентность увеличиваетс€ на 2. —оответственно у р-элементов переменной валентности она различаетс€ на 2. јтом фтора имеет один неспаренный электрон на р-орбитали и не имеет вакантных орбиталей дл€ возбуждени€, т.е. его валентность всегда равна 1. јтом Cl имеет подобную оболочку, однако у него есть возможность переходить в возбужденное состо€ние за счет перехода электронов на вакантные d-орбитали. —оответственно, Cl может иметь валентность 1,3,5,7. F 2s22p5, Cl 3s2 3p53d0. ClF, ClF3, ClF5, ClF7.
|
|
|
433. ¬алентное состо€ние электронов, орбиталей и атома в целом означает возможность образовани€ атомом химической св€зи. јтом фтора имеет один неспаренный электрон на р-орбитали и не имеет вакантных орбиталей дл€ возбуждени€, т.е. его валентность всегда равна 1. F 2s22p5. јтом хлора имеет подобную оболочку, однако у него есть возможность переходить в возбужденное состо€ние за счет перехода электронов на вакантные d-орбитали. —оответственно, хлор может иметь валентность 1,3,5,7. Cl 3s2 3p53d0. јтом кислорода имеет два неспаренных электрона на р-орбитали и подобно фтору не имеет вакантных орбиталей дл€ возбуждени€, т.е. его валентность всегда равна 2. ќ 2s22p4. —ера, как и хлор, находитс€ в третьем периоде, в котором есть вакантна€ d-орбиталь. —оответственно сера может быть 2-,4-,6-тивалентной. S 3s2 3p43d0. ƒл€ атома берилли€, валентные электроны которого наход€тс€ на 2s орбитали вакантной €вл€етс€ 2р-орбиталь, на которую может происходить возбуждение электронов. ¬алентность всегда берилли€ равна 2. ¬е 2s22p0. Ѕор имеет на один электрон больше, чем бериллий, т.е. его валентность всегда равна 3 (поскольку 2s- и 2р-орбитали близки по энергии, возбуждение берилли€ протекает очень легко и валентность 1 дл€ него не характерна). ¬ 2s22p1. ”глерод имеет на один электрон больше, чем бор; его валентность равна 4 (поскольку 2s- и 2р-орбитали близки по энергии, возбуждение углерода, как и берилли€, протекает очень легко и валентность 2 дл€ — не характерна). — 2s22p2.
434. Ќаход€сь в основном состо€нии элементы обычно имеют немного неспаренных электронов, однако многие элементы способны переходить в возбужденное состо€ние, что приводит к по€влению дополнительных неспаренных электронов, что увеличивает валентные возможности элементов. ќсновное состо€ние атома характеризуетс€ минимумом энергии, в то врем€ как возбужденное состо€ние характеризуетс€ избыточной энергией. огда атом находитс€ в возбужденном состо€нии его электроны могут распариватьс€ и переходить на вакантные орбитали. PCl3, PCl5. ¬алентна€ конфигураци€ атома фосфора в основном состо€нии: 3s23p3, т.е. валентность фосфора в основном состо€нии равна 3. —ледовательно в молекуле PCl3 фосфор находитс€ в основном, а в PCl5 - возбужденном состо€нии. H2S, SO3. ¬алентна€ конфигураци€ атома серы в основном состо€нии: 3s23p4, т.е. валентность серы в основном состо€нии равна 2. —ледовательно в молекуле H2S сера находитс€ в основном, а в SO3 - возбужденном состо€нии.
435. √лавной причиной образовани€ химической св€зи между атомами €вл€етс€ стремление к минимуму энергии: при образовании св€зи электронные облака перестраиваютс€ так, что нова€ система энергетически более выигрышна, чем предыдуща€, представл€юща€ собой два отдельных атома (молекулы). ќ перекрывании электронных облаков при образовании молекулы водорода говорит тот факт, что линейный размер молекулы меньше суммы диаметров двух атомов водорода.
436. ќбразование одиночной св€зи (σ-св€зь) происходит вдоль линии св€зывани€, при этом происходит наибольшее перекрывание, поэтому σ-св€зь наиболее прочна€. ¬тора€ и треть€ св€зь (π-св€зь) образуютс€ над линией св€зи, при этом сильного перекрывани€ электронных облаков не происходит, поэтому энерги€ σ-св€зь + π-св€зь не равна удвоенной σ-св€зи.
| ќдинарна€ св€зь | 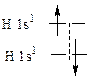
|
| ƒвойна€ св€зь | 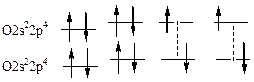
|
| “ройна€ св€зь | 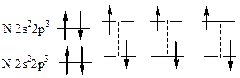
|
437. а) только σ-св€зь: —Ќ4, NaCl, KF
б) одна σ-св€зь + одна π-св€зь: ќ2, —ќ2
в) одна σ-св€зь + две π-св€зи: N2, CO
438. SF6 - шесть σ-св€зей (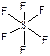 ), SO3 - три σ-св€зи + три π-св€зи (
), SO3 - три σ-св€зи + три π-св€зи (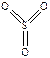 ), POCl3, - четыре σ-св€зи + одна π-св€зь (
), POCl3, - четыре σ-св€зи + одна π-св€зь (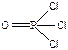 ), C2H2, - три σ-св€зи + две π-св€зи (
), C2H2, - три σ-св€зи + две π-св€зи (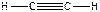 ), C2H4, - п€ть σ-св€зей + одна π-св€зь (
), C2H4, - п€ть σ-св€зей + одна π-св€зь (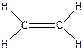 ), COCl2, - три σ-св€зи + одна π-св€зь (
), COCl2, - три σ-св€зи + одна π-св€зь (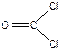 ), SO2Cl2. - четыре σ-св€зи + две π-св€зи (
), SO2Cl2. - четыре σ-св€зи + две π-св€зи (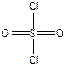 ).
).
439. ќбразование ковалентной св€зи по донорно-акцепторному механизму происходит следующим образом: один атом €вл€етс€ донором электронной пары, а второй Ц имеет вакантную орбиталь и €вл€етс€ акцептором. Ќапример: Ќٱ +:NH3 = [NH4]+. ” водорода имеетс€ вакантна€ орбиталь, а у азота Ц электронна€ пара. ѕри образовании донорно-акцепторной св€зи стехиометрическа€ валентность азота увеличилась на 1, а степень окислени€ и электронна€ валентность не изменились. —техиометрическа€ валентность и степень окислени€ водорода увеличились на 1, а электронна€ валентность не изменилась.
|
|
|
440. —2Ќ6 - между атомами углерода образована одинарна€ св€зь, в то врем€ как в молекуле —2Ќ4 между атомами углерода образована двойна€ св€зь, в результате чего в —2Ќ4 энерги€ св€зи между —-— выше. ¬ бензоле - —6Ќ6 - между атомами углерода формально двойна€ св€зь, однако она делокализована по всему углеродному кольцу, из-за чего энерги€ св€зи —-— ниже, чем в этилене.
441.
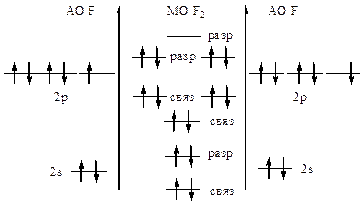
| ћолекула диамагнитна.
KK(σ2sсв)2(σ2sр)2(σ2рхсв)2(πсв2ру,2pz)4(πp2ру,2pz)4
ратность св€зи = 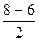 =1, т.е. F-F
ѕри отрыве электрона энерги€ св€зи увеличитс€, т.к. внешний электрон находитс€ на разрыхл€ющей ћќ. =1, т.е. F-F
ѕри отрыве электрона энерги€ св€зи увеличитс€, т.к. внешний электрон находитс€ на разрыхл€ющей ћќ.
|
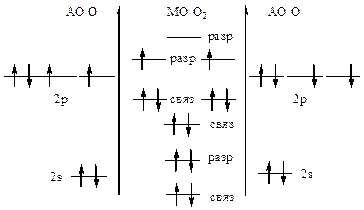
| ћолекула парамагнитна.
KK(σ2sсв)2(σ2sр)2(σ2рхсв)2(πсв2ру,2pz)4(πp2ру,2pz)2
ратность св€зи = 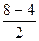 =2, т.е. ќ=ќ
ѕри отрыве электрона энерги€ св€зи увеличитс€, т.к. внешний электрон находитс€ на разрыхл€ющей ћќ. =2, т.е. ќ=ќ
ѕри отрыве электрона энерги€ св€зи увеличитс€, т.к. внешний электрон находитс€ на разрыхл€ющей ћќ.
|
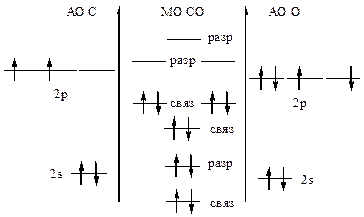
| ћолекула диамагнитна.
KK(σ2sсв)2(σ2sр)2(σ2рхсв)2(πсв2ру,2pz)4
ратность св€зи = 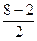 =3, т.е. —≡ќ
ѕри отрыве электрона энерги€ св€зи уменьшитс€, т.к. внешний электрон находитс€ на св€зывающей ћќ. =3, т.е. —≡ќ
ѕри отрыве электрона энерги€ св€зи уменьшитс€, т.к. внешний электрон находитс€ на св€зывающей ћќ.
|
—
443.
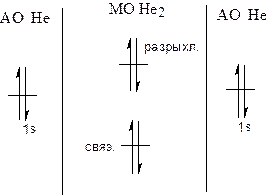
| —огласно теории ћо, молекула может существовать, если кратность св€зей (р) больше 0. ј) р(Ќе2) = (2-2)/2 = 0 Ц молекула не существует Ѕ) ќтрыв одного электрона осуществл€етс€ с разрыхл€ющей ћќ: р = (2-1)/2 = 0.5 Ц молекула существует. ¬) He2- - не существует, т.к. 5 электронов не могут разместитьс€ на двух ћќ. √) ЌеF не может существовать, т.к. энерги€ јќ Ќе и F сильно отличаютс€. ƒ) ЌеЌ. – = (2-10/2 = 0.5 ћолекула может существовать. |
—
445. »онизационный потенциал характеризует энергию, необходимую дл€ отрыва электрона от атома. —огласно теории ћќ электроны легче оторвать от разрыхл€ющих молекул€рных орбиталей. ¬нешние электроны молекул водорода, углерода и азота наход€тс€ на св€зывающих ћќ, поэтому их легче оторвать от атомов, чем от молекул.
446. »онизационный потенциал характеризует энергию, необходимую дл€ отрыва электрона от атома. —огласно теории ћќ электроны легче оторвать от разрыхл€ющих молекул€рных орбиталей. ¬нешние электроны молекул кислорода и азота наход€тс€ на разрыхл€ющих ћќ, поэтому их легче оторвать от молекул, чем от атомов.
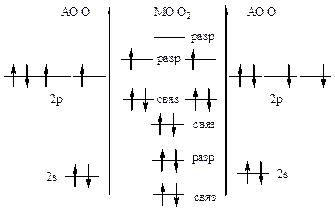
| 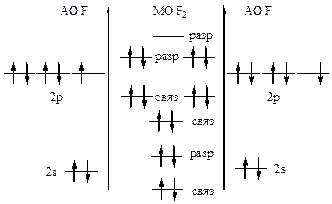
|
447. »онизационный потенциал характеризует энергию, необходимую дл€ отрыва электрона от атома. —огласно теории ћќ электроны легче оторвать от разрыхл€ющих молекул€рных орбиталей молекулы, чем со св€зывающих орбиталей. Ёнерги€, затрачиваема€ на отрыв электронов от атомов имеет промежуточное значение между энергией электронов св€зывающих и разрыхл€ющих ћќ. ¬нешние электроны молекулы —ќ наход€тс€ на св€зывающих ћќ, поэтому их легче оторвать от молекулы, чем от отдельных атомов — и ќ.
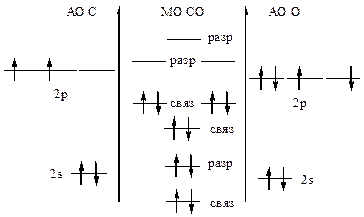
|
448. —огласно теории ћќ, чем больше кратность св€зи (р) молекулы, тем молекула устойчивее (больше ≈св и меньше длина св€зи)
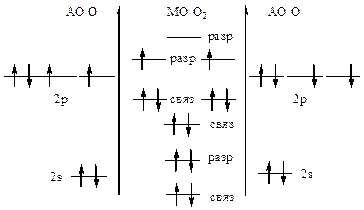
| ƒобавление электрона к ќ2 происходит на разрыхл€ющую ћќ: устойчивость уменьшаетс€. ”даление электрона с разрыхл€ющей ћќ, наоборот, делает молекулу устойчивее:
р(ќ2) = 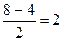 р(ќ2+) = р(ќ2+) = 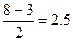 р(ќ2-) =
р(ќ2-) = 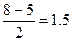 р(ќ22-) = р(ќ22-) = 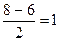
|
449. а) Ц невозбужденна€, т.к. на p2р-св€зывающих орбитал€х азота наход€тс€ спаренные электроны, на разрыхл€ющих p2р-орбитал€х азота неспаренных электронов нет.
б)-возбужденна€
450.






