—амопроизвольно идущий окислительно-восстановительный процесс в определенных услови€х может создавать электрическую энергию. ѕроцессы превращени€ химической энергии в электрическую можно использовать дл€ создани€ химических источников тока (’»“), простейший из которых гальванический элемент.
√альванический элемент состоит из двух электродов, погруженных в электролиты, которые замыкаютс€ электролитическим ключом.
Ћј——»‘» ј÷»я ЁЋ≈ “–ќƒќ¬
ћеталлический электрод - это металл, погруженный в раствор собственной соли, не €вл€етс€ инертным, а участвует в электродной реакции. Cхематично такой электрод записывают в виде ће|ће n +, где вертикальна€ черта обозначает границу между металлом и раствором. ”равнение Ќернста дл€ металлических электродов имеет вид
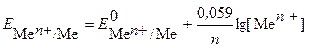 , (4)
, (4)
где  - концентраци€ ионов металла в растворе, моль/л.
- концентраци€ ионов металла в растворе, моль/л.
ќкислительно-восстановительный (редокс-) электрод Ц это инертный металл (токоподвод), погруженный в электролит, содержащий одновременно окисленную и восстановленную формы потенциалопредел€ющих частиц. ¬ качестве инертного металла чаще всего используют платину Pt. —хематично такой электрод можно записать в виде Pt│Me n +, Me m +. Ќа поверхности инертного металла протекает окислительно-восстановительна€ реакци€. Ќапример, дл€ окислительно-восстановительного электрода Pt│Sn4+, Sn2+ такими реакци€ми могут быть: Sn2+ - 2 ē → Sn4+; Sn4+ + 2 ē → Sn2+
”равнение Ќернста дл€ редокс- систем включает концентрацию обоих катионов и имеет вид
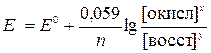 (5)
(5)
где [окисл], [восст] Ц концентрации окисленной и восстановленной форм потенциалопредел€ющих частиц в полуреакции.
√азовые электроды состо€т из инертного металла, который находитс€ в одновременном контакте с газом и раствором, содержащим ионы этого газа. ѕредставител€ми газовых электродов €вл€ютс€ водородный, кислородный, хлорный и другие электроды.
¬одородный электрод состоит из платиновой пластинки, покрытой слоем мелкодисперсной платины ("платиновой черни") и погруженной в раствор кислоты, содержащий ионы водорода. „ерез раствор непрерывно пропускаетс€ поток водорода, водород адсорбируетс€ на поверхности платины, и на границе электрод/раствор устанавливаетс€ равновесие:
H+(раствор) + ē ↔ ½ H2 (г).
ѕри давлении водорода, равном 101,3 кѕа (1 атм), активности (концентрации) ионов водорода 1 моль/л и “ =298 водородный электрод называетс€ стандартным водородным электродом. ѕотенциал такого электрода принимаетс€ за ноль.
”равнение Ќернста дл€ водородного электрода имеет вид
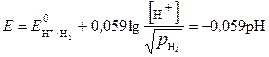 , (6)
, (6)
где 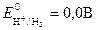 - стандартный электродный потенциал,
- стандартный электродный потенциал,
 - концентраци€ ионов водорода в растворе, моль/л
- концентраци€ ионов водорода в растворе, моль/л
 -парциальное давление водорода над раствором, атм. равно 1 атм.
-парциальное давление водорода над раствором, атм. равно 1 атм.
–ассмотрим работу гальванического элемента на примере элемента ƒаниэл€-якоби. ќн представл€ет собой два сосуда с 1ћ растворами CuSO4 и ZnSO4, в которые погружены соответственно медна€ и цинкова€ пластинки, соединенные проводом. —осуды соединены между собой трубкой, котора€ называетс€ солевым мостиком, заполненной раствором электролита (например, KCl). —олевой мостик €вл€етс€ электролитическим ключом.
|
|
|
Ёлектрод с меньшим значением потенциала зар€жаетс€ отрицательно, €вл€етс€ анодом. Ёлектрод с большим значением потенциала зар€жаетс€ положительно, €вл€етс€ катодом. Ќа аноде протекает процесс окислени€
(отдача электронов), на катоде Ц процесс восстановлени€ (присоединение электронов).
√альванические элементы прин€то записывать в виде схем. јнод со знаком (-) записывают слева, катод со знаком (+) записывают справа. Ќапример, схема медно-цинкового гальванического элемента ƒаниэл€-якоби может быть представлена таким образом:





 ē ē
ē ē
(-) Zn|ZnSO4||CuSO4|Cu (+) или (-) Zn|Zn2+||Cu2+|Cu (+)
ќдна вертикальна€ черта на схеме обозначает границу между металлом и раствором электролита, две черты Ц границу между растворами (солевой мостик).
ѕри замыкании цепи электроны по внешней цепи пойдут от анода к катоду Ц от цинка к меди. ѕри этом на электродах протекают следующие реакции:
(-) јнод: Zn Ц 2 ē → Zn2+ реакци€ окислени€
(+) атод: Cu2+ + 2 ē →Cu реакци€ восстановлени€
—уммиру€ процессы на катоде и аноде, получаем уравнение окислительно-восстановительной реакции, за счет которой в гальваническом элементе возникает электрический ток:
Zn + Cu2+ = Zn2+ + Cu
“акое уравнение называетс€ уравнением токообразующей реакции.
Ёƒ— гальванического элемента рассчитывают как разность потенциалов катода и анода: ≈ = ≈ кЦ ≈ а.
≈сли концентраци€ ионов в растворе составл€ет 1 моль/л, то Ёƒ— называетс€ стандартной. —тандартна€ Ёƒ— медно-цинкового элемента при 

 -
- 
ѕример 7. –ассчитайте Ёƒ— свинцово-цинкового гальванического элемента при “ = 298 , в котором [Zn2+]=0,1моль/л и [Pb2+]=0,01моль/л. ”кажите знаки полюсов, напишите уравнени€ электродных процессов, составьте схему гальванического элемента. ”кажите направление движени€ электронов при замыкании цепи.
–ешение: Ёƒ— гальванического элемента рассчитывают как разность
равновесных потенциалов катода и анода: ≈ = ≈ к Ц ≈ а.
ѕоскольку концентрации потенциалопредел€ющих ионов отличаютс€ от 1 моль/л, рассчитаем по уравнению Ќернста (ур. 4) значени€ электродных потенциалов цинка и свинца:

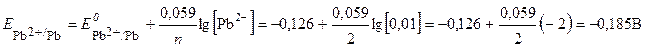 Ёлектрод с меньшим значением потенциала €вл€етс€ анодом (цинковый электрод). Ќа нем протекает реакци€ окислени€:
Ёлектрод с меньшим значением потенциала €вл€етс€ анодом (цинковый электрод). Ќа нем протекает реакци€ окислени€:
(-) ј: Zn Ц 2 ē → Zn2+
Ёлектрод с большим значением потенциала €вл€етс€ катодом (свинцовый электрод), на нем протекает реакци€ восстановлени€:
(+) : Pb2+ + 2 ē → Pb
”равнение токообразующей реакции: Zn + Pb2+ → Zn2+ + Pb.
—хема гальванического элемента: (-) Zn│Zn2+(0,1M)║Pb2+(0,01M)│Pb (+).
–ассчитываем Ёƒ— гальванического элемента:
≈ = ≈ к Ц ≈ а = 
ѕри замыкании цепи, электроны во внешней цепи пойдут от отрицательно зар€женного электрода к положительно зар€женному электроду, то есть - от цинка к свинцу.
ѕример 8. ƒл€ гальванического элемента
Pt│Cr3+ (0,1 моль/л), Cr2+(0,01 моль/л)║Ќ+(рЌ=2)│Ќ2, Pt
рассчитать Ёƒ—, написать уравнени€ электродных процессов, составить уравнение токообразующей реакции, указать знаки полюсов. ќпределить направление движени€ электронов во внешней цепи.
–ешение: данный гальванический элемент составлен из окислительно-
восстановительного и водородного электродов.
ѕотенциал окислительно-восстановительного электрода рассчитываем по уравнению Ќернста: 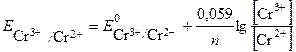
|
|
|
—тандартный потенциал пары —r3+/Cr2+ 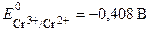 . ѕодставив данные услови€ задачи, рассчитаем потенциал окислительно-восстановительного электрода:
. ѕодставив данные услови€ задачи, рассчитаем потенциал окислительно-восстановительного электрода:
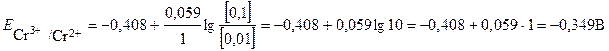 .
.
¬торой электрод данного гальванического элемента €вл€етс€ водородным электродом. ѕотенциал его, согласно уравнению Ќернста:

ќпредел€ем катод и анод. ѕоскольку окислительно-восстановительный электрод имеет меньший потенциал, то в гальваническом элементе он будет играть роль анода (отрицательный полюс), а водородный электрод Ц катода (положительный полюс). ѕосле замыкани€ цепи на первом электроде будет протекать анодный процесс окислени€, на втором Ц катодный процесс восстановлени€:
 (-) ј: Cr2+ - ē → Cr3+ 2
(-) ј: Cr2+ - ē → Cr3+ 2
(+) : 2 Ќ+ + 2 ē → Ќ2
—уммарна€ токообразующа€ реакци€ описываетс€ уравнением
2 Cr2+ + 2 Ќ+ → 2 Cr3+ + Ќ2
Ёлектроны при замыкании внешней цепи будут двигатьс€ от отрицательного полюса к положительному: от хромового окислительно-восстановительного электрода к водородному.
Ёƒ— данного элемента 
ѕример 9. акие процессы протекают на электродах в концентрационном гальваническом элементе, имеющем цинковые электроды, если у одного из электродов концентраци€ ионов цинка Zn2+ равна 1 моль/л, а у другого Ц
0,0001 моль/л? акова Ёƒ— этого элемента? Ќапишите схему данного √Ё.
–ешение: онцентрационный гальванический элемент состоит из одинаковых электродов, погруженных в растворы своих солей различной концентрации. ќпределим потенциалы обоих электродов. “ак как концентраци€ ионов цинка
у первого электрода равна 1 моль/л, то потенциал его будет равен стандартному потенциалу цинкового электрода:  .
.
ѕотенциал второго электрода рассчитаем по уравнению Ќернста:
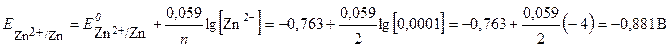 ѕервый электрод €вл€етс€ катодом, на нем после замыкани€ цепи протекает реакци€ восстановлени€ (+) : Zn2+ + 2 ē →Zn
ѕервый электрод €вл€етс€ катодом, на нем после замыкани€ цепи протекает реакци€ восстановлени€ (+) : Zn2+ + 2 ē →Zn
¬торой электрод, имеющий меньший потенциал, будет анодом, на нем протекает реакци€ окислени€: (-) ј: Zn - 2 ē →Zn2+
“окообразующа€ реакци€ в гальваническом элементе будет иметь вид:
Zn + Zn2+→ Zn + Zn2+
–ассчитываем Ёƒ— элемента: ≈ = ≈ к - ≈ а = - 0,763 Ц(-0,881) = 0,122 ¬.
ƒанный гальванический элемент можно отобразить схемой:
(-) Zn│Zn2+(0,0001 моль/л)║Zn2+(1 моль/л)│Zn (+)
«адани€
4. Ќаписать катодный и анодный процессы, уравнение токообразующей реакции и вычислить Ёƒ— гальванического элемента. ”казать пол€рность электродов.
1. Fe| Fe+2 (1моль/л) || Ag+(0,1моль/л) |Ag
2. Cr| Cr+3 (2 моль/л) || Cd+2 (1 моль/л)|Cd
3. Be| Be+2 (0,1моль/л) || Ni+2 (0,01моль/л) |Ni
4. Mn| Mn+2 (0,1 моль/л) || Sn+2 (0,01 моль/л)|Sn
5. Al| Al+3 (2 моль/л) || Cd+2 (0,1 моль/л)|Cd
6. Ni| Ni+2 (0,1 моль/л) || Cu+2 (0,01 моль/л)|Cu
7. Mg| Mg+2 (1 моль/л) || Zn+2 (0,01 моль/л)|Zn
8. Cd| Cd+2 (0,1 моль/л) || Pb+2 (0,01 моль/л)|Pb
9. Fe| Fe+2 (0,01 моль/л) || Fe+2 (1 моль/л)|Fe
10. Co|Co+2 (0,5 моль/л) || Ni+2 (0,5 моль/л)|Ni
11. Zn| Zn+2 (1 моль/л) || Cu+2 (0,01 моль/л)|Cu
12. Pt,H2|H+ (pH=4) || H+ (pH=2)|H2,Pt
13. Al|Al+3 (1 моль/л) ||Al+3 (2 моль/л)|Al
14. Mn|Mn+2 (1 моль/л) || Ni+2 (0,01 моль/л)|Ni
15. Ca|Ca+2 (0,1 моль/л) || Fe+2 (0,01 моль/л)|Fe
16. Pb|Pb+2 (1 моль/л) || Ag+ (0,1 моль/л)|Ag
17. Be|Be+2 (0,001 моль/л) || Fe+2 (0,001 моль/л)|Fe
18. Pt|Cr+3 (0,1 моль/л),Cr+2 (0,1 моль/л) || Fe+3 (0,1 моль/л), Fe+2 (0,1 моль/л)|Pt
19. Zn|Zn+2(1 моль/л) || Fe+2 (0,01 моль/л)|Fe
20. Al|Al+3 (2 моль/л) || Cu+2 (0,01 моль/л)|Cu
21. Be|Be+2 (1 моль/л) || Cd+2 (0,01 моль/л)|Cd
22. Mn|Mn+2 (0,01 моль/л) || Fe+2 (0,01 моль/л)|Fe
23. Fe|Fe+2 (1 моль/л) || Sn+2 (0,01 моль/л)|Sn
24. Mg|Mg+2 (0,1 моль/л) || Ni+2 (0,01 моль/л)|Ni
25. Ag|Ag+ (0,001 моль/л) || Ag+ (0,1 моль/л)|Ag
26. Fe| Fe+2 (0,005 моль/л) || Pb+2 (0,005 моль/л)|Pb
27. Ca|Ca+2 (1 моль/л) || Mg+2 (0,01 моль/л)|Mg
28. Mn|Mn+2 (0,01 моль/л) || Zn+2 (0,01 моль/л)|Zn
29. Pb|Pb+2 (0,1 моль/л) ||Cu+2 (0,01 моль/л)|Cu
30. Zn|Zn+2 (10-4 моль/л) || Sn+2 (10-2 моль/л)|Sn
|
|
|
31. Cd| Cd+2 (10-4 моль/л) || Cu+2 (10-2 моль/л)|Cu
32. Ni| Ni+2 (0,01 моль/л) || Ag+ (1 моль/л)|Ag
33. Mg| Mg+2 (0,001 моль/л) || Mg+2 (1 моль/л)|Mg
34. Ca| Ca+2 (1 моль/л)|| Cr+2 (0,01 моль/л)|Cr
35. Mn|Mn+2 (0,01 моль/л) || Pb+2 (0,01 моль/л)|Pb
36. Al| Al+3 (2 моль/л) || Cr+2 (0,01 моль/л)|Cr
37. Be| Be+2 (1 моль/л) || Sn+2 (0,01 моль/л)|Sn
38. Ni| Ni+2 (1 моль/л)|| Fe+3(2 моль/л)|Fe
39. Pt, H2|H+ (pH=4) || Ag+ (1 моль/л)|Ag
40. Mg| Mg+2 (10-2 моль/л) || Cu+2 (10-2 моль/л)|Cu
41. Cu| Cu+2 (0,01 моль/л) || Ag+ (0,1 моль/л)|Ag
42. Ca| Ca+2 (0,01 моль/л) || Be+2 (1 моль/л)|Be
43. Zn| Zn+2 (0,01 моль/л) || Ni+2 (1 моль/л)|Ni
44. Al| Al+3 (2 моль/л) || Fe+2 (0,01 моль/л)|Fe
45. Ca| Ca+2 (1 моль/л) || Al+3 (0,5 моль/л)|Al
46. Fe| Fe+3 (2 моль/л) || Cu+2 (0,01 моль/л)|Cu
47. Zn| Zn+2 (1 моль/л) || Pb+2 (0,01 моль/л)|Pb
48. Sn| Sn+2 (0,01 моль/л) || Ag+(0,1 моль/л)|Ag
49. Zn| Zn+2 (10-2 моль/л) || H+(pH=2)|H2,Pt
50. Pt| Sn+4 (0,1 моль/л),Sn+2 (0,1 моль/л) || Hg+2 (1 моль/л)|Hg
51. Al| Al+3 (2 моль/л) || Cd+2 (0,01 моль/л)|Cd
52. Pt|Cr+3(10-2 моль/л),Cr+2(0,1 моль/л) || Sn+4 (10-3 моль/л),Sn+2(0,1 моль/л)|Pt
53. Ni| Ni+2 (0,01 моль/л) || Pb+2 (0,01 моль/л)|Pb
54. Mg| Mg+2 (10-4 моль/л) || H+(pH=2)|H2,Pt
55. Cd| Cd+2 (10-2 моль/л) || Cd+2 (1 моль/л)|Cd
56. Be| Be+2 (10-4 моль/л) || Zn+2 (10-2 моль/л)|Zn
57. Mn| Mn+2 (1 моль/л) || Cu+2 (0,1 моль/л)|Cu
58. Ca| Ca+2 (0,01 моль/л) || Pb+2 (0,01 моль/л)|Pb
59. Sn| Sn+2 (0,01 моль/л) || Cu+2 (1 моль/л)|Cu
60. Cd| Cd+2 (10-1 моль/л) || Sn+2 (10-1 моль/л)|Sn
61.Pt|Fe+3(10-2 моль/л),Fe+2(0,1 моль/л) || Sn+4(10-2 моль/л),Sn+2(0,1 моль/л)|Pt
62.Pt|Co+3(10-2 моль/л), Co+2 (0,1 моль/л) ||Sn+4 (0,1 моль/л),Sn+2 (10-2 моль/л)|Pt
63. Pt|Fe+3(0,1 моль/л), Fe+2(10-2 моль/л) ||Au+ (10-2 моль/л), Au+3 (0,1 моль/л)|Pt






