јлгоритм обчисленн€ маси речовини
за в≥домою к≥льк≥стю речовини
| ѕосл≥довн≥сть д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | ќбчисл≥ть масу води к≥льк≥стю речовини 1,5 моль. |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: n(Ќ2ќ) = 1,5 моль «найти: m(Ќ2ќ) -? |
3) Ќапиш≥ть формулу звТ€зку м≥ж к≥льк≥стю речовини, мол€рною масою ≥ масою речовини: n = 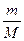
|
n (Ќ2ќ) = 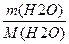
|
| 4) ¬ивед≥ть математичний вираз дл€ маси | m(Ќ2ќ) = n(Ќ2ќ) Ј M(Ќ2ќ) |
| 5) ќбчисл≥ть за формулою масу, враховуючи, що ћ = =ћr | ћ(Ќ2ќ) = 1Ј2 + 16 = 18 г/моль m(Ќ2ќ) = 1,5 моль Ј 18 г/моль = = 27 г |
| 6) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: m(Ќ2ќ) = 27 г |
ћол€рний обТЇм газу
“рохи теор≥њЕ
ћол€рний обТЇм газу Ц це обТЇм 1 моль газу. ÷€ величина залежить в≥д температури й тиску. ѓњ традиц≥йно виражають у л/моль, або м3/моль.
«а нормальних умов (н.у. - 0∞—, 101,3 кѕа) мол€рний обТЇм ≥деального газу становить 22,4 л/моль (число јвогадро). ÷ю константу позначають Vm ≥ називають мол€рним обТЇмом газу.
ћол€рний обТЇм газу Ц це в≥дношенн€ обТЇму газу до в≥дпов≥дноњ к≥лькост≥ речовини:
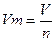 ,
,
де V Ц обТЇм газу; n Ц к≥льк≥сть речовини газу.
« ц≥Їњ формули можна вивести формулу дл€ визначенн€ к≥лькост≥ речовини: 
јлгоритм обчисленн€ обТЇму газу
за в≥домою к≥льк≥стю речовини
| ѕосл≥довн≥сть д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | ќбчисл≥ть обТЇм кисню к≥льк≥стю речовини 3 моль. |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: n(ќ2) = 3 моль «найти: V(ќ2) -? |
3) Ќапиш≥ть формулу звТ€зку м≥ж к≥льк≥стю речовини, мол€рним обТЇмом ≥ обТЇмом газу:
n = 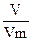
|
n(ќ2) = 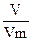
|
| 4) «найд≥ть математичний вираз дл€ обТЇму | V(O2) = n(O2) Ј Vm |
| 5) ќбчисл≥ть за формулою обТЇм, враховуючи, що Vm дл€ вс≥х газ≥в за н.у. складаЇ 22,4 л/моль | V(O2) = 3 моль Ј 22,4 л/моль = = 67,2 л |
| 6) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: V(O2) = 67,2 л |
„исло структурних частинок
“рохи теор≥њЕ
—тала јвогадро (Nј) Ц к≥льк≥сть частинок в 1 моль будь-€коњ речовини. ÷е число стала величина ≥ дор≥внюЇ 6,02Ј1023. —тала јвогадро маЇ розм≥рн≥сть моль-1.
„исло структурних частинок речовини Ц це добуток сталоњ јвогадро на к≥льк≥сть речовини:
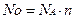
≥льк≥сть речовини визначаЇтьс€ €к в≥дношенн€ числа структурних частинок до сталоњ јвогадро: 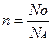
јлгоритм обчисленн€ числа структурних частинок речовини за в≥домою масою
| ѕосл≥довн≥сть д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | —к≥льки молекул м≥ститьс€ в 6,4г с≥рки? |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: m(S) = 6,4г «найти: N0(S) -? |
| 3) Ќапиш≥ть формулу звТ€зку м≥ж к≥льк≥стю речовини, числом структурних частинок та сталою јвогадро та формулу звТ€зку м≥ж к≥льк≥стю речовини, масою та мол€рною масою. | 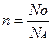 ; ; 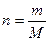
|
| 4) ѕрир≥вн€йте л≥ву та праву частини формул та вивед≥ть число структурних частинок ≥з знайденого сп≥вв≥дношенн€. | 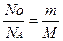 , зв≥дси , зв≥дси 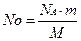
|
| 5) ќбчисл≥ть за формулою число структурних частинок, памТ€таючи, що стала јвогадро становить 6,02Ј1023моль-1. |
N(молекул) = = 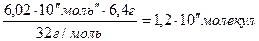
|
| 6) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: N(молекул) = 1,2Ј1023. |
ќб'Їмн≥ в≥дношенн€ газ≥в
|
|
|
“рохи теор≥њЕ
«акон об'Їмних в≥дношень (∆. √ей-Ћюссак, 1802)
ќбТЇми газопод≥бних речовин, що вступають у реакц≥ю, в≥днос€тьс€ м≥ж собою та до обТЇм≥в газопод≥бних продукт≥в реакц≥њ €к невелик≥ ц≥л≥ числа.
ѕриклад:
–еакц≥€ гор≥нн€ пропану: —3Ќ8 + 5ќ2 = 3—ќ2 + 4Ќ2ќ(газ)
V(—3Ќ8): V(ќ2): V(—ќ2): V(Ќ2ќ)=1:5:3:4.
јлгоритм обчисленн€ об´Їмних в≥дношень газ≥в
за р≥вн€нн€м х≥м≥чноњ реакц≥њ
| ѕосл≥довн≥сть д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | ќбчисл≥ть обТЇм (н.у.) кисню, необх≥дний дл€ реакц≥њ з карбон (≤≤) оксидом обТЇмом 20 л. |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: V(—ќ2) = 20л «найти: V(ќ2) -? |
| 3) —клад≥ть р≥вн€нн€ реакц≥њ | 2—ќ + ќ2 = 2—ќ2 |
| 4) ѕ≥дкресл≥ть формули речовин, про €к≥ йдетьс€ в умов≥ задач≥ | 2—ќ + ќ2 = 2—ќ2 |
| 5) Ќадпиш≥ть над п≥дкресленими формулами числов≥ значенн€ в≥домого обТЇму, п≥д формулами Ц числов≥ значенн€ обТЇм≥в, €к≥ випливають з даного р≥вн€нн€ реакц≥њ та в≥дпов≥дають коеф≥ц≥Їнтам | 20л х л 2—ќ + ќ2 = 2—ќ2 2 1 |
| 6) —клад≥ть в≥дношенн€ та обчисл≥ть обТЇм газу в л≥трах | 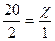 ; ;  (л) (л)
|
| 6) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: V(ќ2) = 10л |
¬≥дносна густина газ≥в
“рохи теор≥њЕ
√устина газу ј за газом ¬ дор≥внюЇ в≥дношенню мол€рноњ маси газу ј до мол€рноњ маси газу ¬. ѓњ позначають D¬(ј). ц€ величина дор≥внюЇ в≥дношенню вим≥р€них в однакових умовах густин газ≥в:
D¬(ј)= 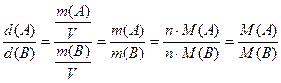
як правило, густини р≥зних газ≥в визначають в≥дносно:
водню Ц ћ(Ќ2) = 2 г/моль,
кисню Ц ћ(ќ2) = 32 г/моль,
пов≥тр€ Ц ћ(пов.) = 29 г/моль.
јлгоритм обчисленн€ в≥дносноњ густини газу
| ѕосл≥довн≥сть д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | ќбчисл≥ть в≥дносну густину сульфур (≤V) оксиду за воднем. |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: SO2 «найти: DЌ2 -? |
| 3) «апиш≥ть розрахункову формулу в≥дносноњ густини за воднем | DЌ2 = 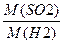 ћ (Ќ2) = 2 г/моль
DЌ2 =
ћ (Ќ2) = 2 г/моль
DЌ2 = 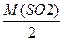
|
| 4) ќбчисл≥ть в≥дносну молекул€рну масу сульфур (≤V) оксиду за х≥м≥чною формулою | ћr(SO2) = 32 + 16Ј2 = 64 ћ (SO2) = 64 г/моль |
| 5) ѕ≥дставте у формулу (д≥€ 3) значенн€ ћ ≥ отримайте в≥дпов≥дь |
DЌ2 = 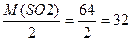
|
| 6) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: DЌ2 = 32 |
«адач≥ за термох≥м≥чними р≥вн€нн€ми
“рохи теор≥њЕ
“епловий ефект реакц≥њ Ц це теплота, €ка вид≥л€Їтьс€ або поглинаЇтьс€ при прот≥канн≥ х≥м≥чноњ реакц≥њ.
“епловий ефект реакц≥њ Q в≥д'Їмний, €кщо теплота поглинаЇтьс€, ≥ додатн≥й, €кщо теплота вид≥л€Їтьс€.
“ермох≥м≥чне р≥вн€нн€ Ц це х≥м≥чне р≥вн€нн€, у €кому вказано тепловий ефект реакц≥њ.
≈нергетика х≥м≥чних реакц≥й
 |

 –еакц≥њ
–еакц≥њ
 |  |
екзотерм≥чн≥ ендотерм≥чн≥ (теплота вид≥л€Їтьс€) (теплота поглинаЇтьс€)
2Ќ2 + ќ2= 2Ќ2ќ + Q 2 ћnќ4 = 2ћnќ4 + ћnќ2 +ќ2↑-Q
јлгоритм обчисленн€ к≥лькост≥ теплоти
за термох≥м≥чним р≥вн€нн€м реакц≥њ
| ѕосл≥довн≥сть д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | ќбчисл≥ть к≥льк≥сть теплоти, що вид≥литьс€ внасл≥док спалюванн€ фосфору масою 6,2 г. “ермох≥м≥чне р≥вн€нн€ реакц≥њ гор≥нн€ фосфору: 4– + 5ќ2 = 2–2ќ5 + 3010 кƒж |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: m(–) = 6,2 г Q1 = 3010 кƒж «найти: Q2 -? |
| 3) «апиш≥ть термох≥м≥чне р≥вн€нн€ | 4– + 5ќ2 = 2–2ќ5 + 3010 кƒж |
| 4) ѕ≥дкресл≥ть формулу речовини з в≥домими вих≥дними даними ≥ числове значенн€ теплового ефекту | 4– + 5ќ2 = 2–2ќ5 + 3010 кƒж |
| 5) Ќад п≥дкресленою формулою речовини вкаж≥ть вих≥дн≥ дан≥, п≥д формулою Ц числов≥ дан≥, законом≥рн≥ дл€ цього р≥вн€нн€, над числовим значенн€м теплового ефекту Ц умовне позначенн€ Q | 6,2 г Q 4– + 5ќ2 = 2–2ќ5 + 3010 кƒж n = 4 моль ћ = 31 г/моль m = 124 г |
| 6) ќбчисл≥ть шукану к≥льк≥сть теплоти в кƒж | 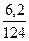 = = 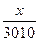 ’ =
’ = 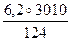 = 150,5 (кƒж) = 150,5 (кƒж)
|
| 7) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: Q2 = 150,5 кƒж |
ћасова частка розчиненоњ речовини у розчин≥
|
|
|
“рохи теор≥њЕ
ћасова частка розчиненоњ речовини Ц це безрозм≥рна величина, що дор≥внюЇ в≥дношенню маси розчиненоњ речовини до загальноњ маси розчину:
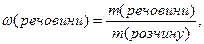
де 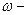 масова частка розчиненоњ речовини; m Ц маси речовини або розчину.
масова частка розчиненоњ речовини; m Ц маси речовини або розчину.
m(розчину) = m(речовини) + m(води),
тобто масову частку можна виразити €к
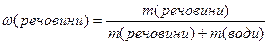 .
.
ћасову частку розчиненоњ речовини ω звичайно виражають у частках в≥д одиниц≥ або у в≥дсотках. Ќаприклад, масова частка розчиненоњ речовини Ц —а—l2 у розчин≥ дор≥внюЇ 0,06, або 6%. ÷е означаЇ, що в розчин≥ альц≥й хлориду масою 100 г м≥ститьс€ хлорид кальц≥ю масою 6 г ≥ вода масою 94 г.
јлгоритм обчисленн€ маси розчиненоњ речовини,
€кщо в≥дома масова частка розчиненоњ речовини й маса розчину
| ѕосл≥довн≥сть д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | ќбчисл≥ть масу натр≥й хлориду, необх≥дну дл€ приготуванн€ розчину масою 200 г з масовою часткою сол≥ 0,05. |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: m(розч.) = 200 г ω (NaCl) = 0,05 «найти: m(NaCl) -? |
3) «апиш≥ть формулу дл€ обчисленн€ маси розчиненоњ речовини, виход€чи ≥з формули: ω (NaCl) = 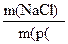
| m(NaCl) = ω (NaCl) Ј m(розч.) |
| 4) ѕ≥дставте в отриманий вираз чисельн≥ дан≥ з умови задач≥ та зд≥йсн≥ть розрахунки | m(NaCl) = 0,05 Ј 200 = 10(г) |
| 7) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: m(NaCl) = 10г |
ћол€рна концентрац≥€
“рохи теор≥њЕ
ћол€рна концентрац≥€ Ц в≥дношенн€ к≥лькост≥ розчиненоњ речовини до обТЇму розчину.
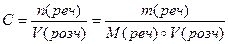
[C] = моль/л
јлгоритм обчисленн€ мол€рноњ концентрац≥њ розчину
| ѕосл≥довн≥сть д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | ќбчисл≥ть мол€рну концентрац≥ю розчину сульфатноњ кислоти масою 225г (ρ = 1,5 г/мл) з масовою часткою кислоти 52%. |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: m(розч.) = 225 г ω (NaCl) = 0,05 ρ(розч) = 1,5 г/мл «найти: —ћ -? |
| 3) «апиш≥ть формулу дл€ обчисленн€ обТЇму розчину за густиною |
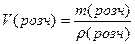
|
| 4) ѕ≥дставте в отриманий вираз чисельн≥ дан≥ з умови задач≥ та зд≥йсн≥ть розрахунки |
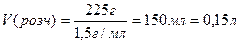
|
| 5) «апиш≥ть формули дл€ обчисленн€ маси речовини та к≥лькост≥ речовини |  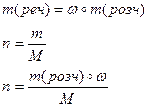
|
| 6) ѕ≥дставте в отриманий вираз чисельн≥ дан≥ з умови задач≥ та зд≥йсн≥ть розрахунки | 
|
| 7) «апиш≥ть формулу дл€ визначенн€ мол€рноњ концентрац≥њ розчину | 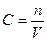
|
| 8) ѕ≥дставте в отриманий вираз чисельн≥ дан≥ з пункт≥в 4 та 6 | 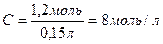
|
| 9) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: —ћ= 8моль/л |
—туп≥нь окисненн€
“рохи теор≥њЕ
—туп≥нь окисненн€ елемента у сполуц≥ Ц це умовний зар€д його атом≥в, обчислений у припущенн≥, що вс≥ зв'€зки у сполуц≥ мають йонний характер.
ѕринцип електронейтральност≥: алгебрањчна сума ступен≥в окисненн€ вс≥х атом≥в у молекул≥ дор≥внюЇ нулю.
≈лементи з≥ сталим ступенем окисненн€ у сполуках
| —туп≥нь окисненн€ | ≈лементи |
| ”с≥ прост≥ речовини Ц метали й неметали | |
| -1 | F |
| +1 | Li, Na, K, Rb, Cs |
| +2 | Be, Mg, Ca, Sr, Ba, Ra, Zn |
| +3 | Al |
√≥дроген ви€вл€Ї у сполуках ступ≥нь окисненн€ +1, р≥дко -1(у сполуках з металами).
ќксиген ви€вл€Ї у сполуках ступ≥нь окисненн€ -2, р≥дко -1 (у пероксидах) та +2 у сполуц≥ з ‘лором (F2ќ).
ќкисно-в≥дновн≥ реакц≥њ
“рохи теор≥њЕ
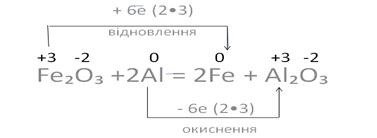
–еакц≥њ, у процес≥ €ких зм≥нюютьс€ ступен≥ окисненн€ елемент≥в, що вход€ть до складу реагуючих речовин, називаютьс€ окисно-в≥дновними.
|
|
|
ѕереб≥г окисно-в≥дновних реакц≥й ≥, отже, зм≥на ступен€ окисненн€ атом≥в, зумовлен≥ переходом електрон≥в в≥д одних атом≥в до ≥нших.


|
|
 |

|
|

ќкисненн€ Цпроцес в≥ддаванн€ електрон≥в.
 S0 Ц 4е → S+4 S0 Ц 4е → S+4
| ¬≥дновленн€Ц процес прийманн€ електрон≥в.
 N+5 + 3е → N+2 N+5 + 3е → N+2
|
| јтоми, йони або молекули, що в≥ддають електрони, називають в≥дновниками.Ќайсильн≥ш≥ в≥дновники: Ќ2, —ќ, Ќ2S, NЌ3, — (вуглець), метали. | јтоми, йони або молекули, що приймають електрони, називають окисниками. Ќайсильн≥ш≥ окисники: ќ2, ќ3, галогени, ЌNќ3, Ќ2Sќ4, ћnќ4. |
—хема взаЇмозвТ€зку зм≥ни ступен≥в окисненн€ з процесами окисненн€ та в≥дновленн€
| ¬≥дновленн€ Ц ступ≥нь окисненн€ зменшуЇтьс€ (+ е) | ||||||||||||
 -4 -4
| -3 | -2 | -1 | +1 | +2 | +3 | +4 | +5 | +6 | +7 | +8 | |
 ќкисненн€ Ц ступ≥нь окисненн€ зб≥льшуЇтьс€ (- е) ќкисненн€ Ц ступ≥нь окисненн€ зб≥льшуЇтьс€ (- е)
|
„исло електрон≥в, €к≥ в≥ддаЇ в≥дновник, завжди дор≥внюЇ числу електрон≥в, €к≥ приЇднуЇ окисник, тобто дотримуЇтьс€ електронний баланс.
–озрахунки за р≥вн€нн€ми х≥м≥чних реакц≥й
“рохи теор≥њЕ
ѕам'€тка: розвТ€зуванн€ розрахункових задач за р≥вн€нн€м реакц≥њ
1. —кладаЇмо р≥вн€нн€ х≥м≥чноњ реакц≥њ.
2. Ќад формулами речовин записуЇмо значенн€ в≥домих ≥ нев≥домих речовин з в≥дпов≥дними одиниц€ми вим≥рюванн€ (т≥льки дл€ чистих речовин).
3. ѕ≥д формулами в≥домих ≥ нев≥домих речовин записуЇмо в≥дпов≥дн≥ значенн€ величин, знайден≥ за р≥вн€нн€м реакц≥њ.
4. —кладаЇмо та розвТ€зуЇмо пропорц≥ю.
5. «аписуЇмо в≥дпов≥дь.
јлгоритм обчисленн€ маси продукту реакц≥њ
за в≥домою к≥льк≥стю речовини
| ѕосл≥довн≥сть д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | ќбчисл≥ть масу води, €ка утворюЇтьс€ при взаЇмод≥њ алюм≥н≥й оксиду к≥льк≥стю речовини 0,5 моль ≥з сульфатною кислотою. |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: n(јl2ќ3) = 0,5 моль «найти: m(Ќ2ќ) -? |
| 3) —клад≥ть р≥вн€нн€ реакц≥њ | јl2ќ3 + 3Ќ2Sќ4=јl2 (Sќ4)3 +3Ќ2ќ |
| 4) ѕ≥дкресл≥ть формули речовин, про €к≥ йдетьс€ в умов≥ задач≥ | јl2ќ3 +3Ќ2Sќ4 =јl2 (Sќ4)3 + 3Ќ2ќ |
| 5) Ќадпиш≥ть над п≥дкресленими формулами вих≥дн≥ дан≥, п≥д формулами Ц дан≥, що законом≥рно вит≥кають з р≥вн€нн€ реакц≥њ | 0,5 моль x моль јl2ќ3 + 3Ќ2Sќ4=јl2 (Sќ4)3 + 3Ќ2ќ 1 моль 3 моль |
| 6) ќбчисл≥ть к≥льк≥сть речовин | 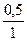 = = 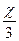 → х = 0,5Ј3 = 1,5 (моль) → х = 0,5Ј3 = 1,5 (моль)
|
| 7) ќбчисл≥ть ћ шуканоњ речовини | ћ(Ќ2ќ) = 1Ј2 + 16 = 18 г/моль |
8) ¬икористовуючи формулу розрахунку к≥лькост≥ речовини 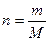 , обчисл≥ть масу речовини
m = ћЈn , обчисл≥ть масу речовини
m = ћЈn
| m(Ќ2ќ) = ћ(Ќ2ќ) Ј n(Ќ2ќ) = = 18 г/моль Ј 1,5 моль = 27 г |
| 9) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: m(Ќ2ќ) = 27 г |
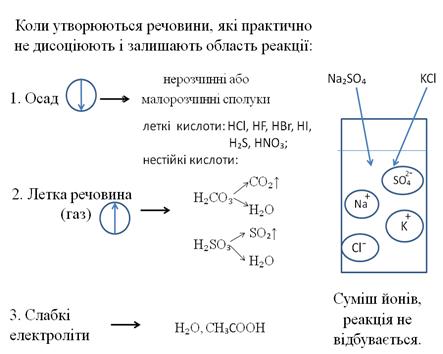
–еакц≥њ йонного обм≥ну
“рохи теор≥њЕ
–еакц≥њ м≥ж йонами, що в≥дбуваютьс€ в розчинах називають реакц≥€ми йонного обм≥ну, а р≥вн€нн€ таких реакц≥й Ц йонними р≥вн€нн€ми.
јлгоритм складанн€ йонних р≥вн€нь
- —класти молекул€рне р≥вн€нн€ реакц≥њ та ур≥вн€ти його:
CuSO4 + 2 NaOH = Cu(OH)2 + Na2SO4
- ¬изначити, користуючись таблицею розчинност≥, €ка з речовин залишаЇ область реакц≥њ (Ї осадом, газом або слабким електрол≥том): CuSO4 + 2 NaOH = Cu(OH)2↓ + Na2SO4
- ористуючись таблицею розчинност≥ записуЇмо дисоц≥ац≥ю ус≥х речовин з урахуванн€м математичних знак≥в х≥м≥чного р≥вн€нн€, кр≥м осаду (газу або слабкого електрол≥ту). “аку форму запису називають повним йонним р≥вн€нн€: Cu2++ SO42- + 2 Nа++2OH- = Cu(OH)2↓ + 2Na++ SO42-
- ѕ≥дкреслюЇмо однаков≥ йони л≥воњ та правоњ частин: Cu2++ SO4 2- + 2 Nа+ +2OH- = Cu(OH)2↓ + 2Na+ + SO4 2-
- «аписуЇмо скорочене йонне р≥вн€нн€ без п≥дкреслених йон≥в: Cu2++ 2OH- = Cu(OH)2↓
|
|
|
”мови переб≥гу реакц≥й йонного обм≥ну до к≥нц€
«адач≥ на надлишок ≥ нестачу
“рохи теор≥њЕ
–еагенти вступають у реакц≥ю в ч≥тко визначених масових сп≥вв≥дношенн€х. ƒосить часто в практичн≥й робот≥ зустр≥чаютьс€ ситуац≥њ, коли один з реагент≥в залишаЇтьс€ в надлишку або, навпаки одного з реагент≥в не вистачаЇ дл€ одержанн€ визначеноњ к≥лькост≥ продукту реакц≥њ. ѕри розвТ€зуванн≥ задач цього типу сл≥д памТ€тати, що ≥з двох вих≥дних речовин розрахунок проводитьс€ лише за т≥Їю речовиною, €ка знаходитьс€ в нестач≥. “обто ц€ речовина п≥д час реакц≥њ витрачаЇтьс€ повн≥стю. ¬изначити, €ка з речовин перебуваЇ в нестач≥, можна склавши в≥дношенн€ маси або обТЇму вих≥дноњ речовини, що дано за умовою задач≥, до в≥дпов≥дноњ маси або обТЇму, визначеного за р≥вн€нн€м реакц≥њ.
јлгоритм обчисленн€ за р≥вн€нн€м х≥м≥чноњ реакц≥њ,
€кщо одна з вих≥дних речовин уз€та в надлишку
| ѕор€док д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | ќбчисл≥ть масу осаду, €кий утворитьс€ внасл≥док взаЇмод≥њ розчин≥в, €к≥ м≥ст€ть купрум (≤≤) сульфат масою 8г та натр≥й г≥дроксид масою 10г. |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: m(CuSO4) = 8г m(NaOH) = 10г «найти: m(Cu(OH)2) -? |
| 3) «апиш≥ть р≥вн€нн€ реакц≥њ | CuSO4+2 NaOH = Cu(OH)2 + Na2SO4 |
| 4) ѕ≥дкресл≥ть формули речовин, про €к≥ йдетьс€ в умов≥ задач≥ | CuSO4 + 2 NaOH = Cu(OH)2 + Na2SO4 |
| 5) Ќадпиш≥ть над п≥дкресленими формулами вих≥дн≥ дан≥, п≥д формулами Ц дан≥, що законом≥рно вит≥кають з р≥вн€нн€ реакц≥њ | 8г 10г х г CuSO4 + 2 NaOH = Cu(OH)2↓ + Na2SO4 n=1моль n=2моль n=1моль M=160г/моль M=40г/моль M=98г/моль m = 160 г m = 80 г m = 98 г |
| 6) «найд≥ть, €ка ≥з двох даних речовин вз€та в нестач≥ | 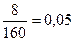 ; ; 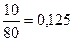 ; 0,05 < 0,125
¬ нестач≥ CuSO4, тому розрахунок проводимо саме за ц≥Їю речовиною. ; 0,05 < 0,125
¬ нестач≥ CuSO4, тому розрахунок проводимо саме за ц≥Їю речовиною.
|
| 7) –озрахунок зд≥йснюЇмо за речовиною, €ка повн≥стю використовуЇтьс€, тобто за т≥Їю, €ка в нестач≥ | 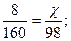 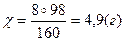
|
| 8) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: m(Cu(OH)2) = 4,9 г |
«адач≥ на дом≥шки
“рохи теор≥њЕ
” природ≥ чистих речовин не буваЇ. ожна з природних речовин Ї сум≥шшю к≥лькох р≥зних речовин. –ечовини, кр≥м основноњ, €к≥ в даному випадку не Ї ц≥нними та не використовуютьс€ на виробництв≥, називають дом≥шками. ”часть в х≥м≥чн≥й реакц≥њ приймаЇ лише чиста речовина, дом≥шки участ≥ в реакц≥њ не приймають, тому њх масу сл≥д в≥дн≥мати в≥д загальноњ маси речовини.
ƒл€ техн≥чних речовин (речовини з дом≥шками) можна виразити масову частку дом≥шок, або масову частку чистоњ речовини у частках в≥д одиниц≥, або у в≥дсотках.
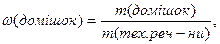 або
або 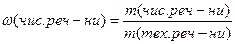
јлгоритм обчисленн€ маси продукту реакц≥њ
за в≥домою масою вих≥дноњ речовини, €ка м≥стить дом≥шки
| ѕосл≥довн≥сть д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | яку масу кальц≥й оксиду можна отримати з вапн€ку масою 400 кг з масовою часткою дом≥шок 0,2? |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: m(вапн€ку) = 400 кг ω(дом≥шок) = 0,2 «найти: m(CaO) -? |
| 3) ќбчисл≥ть масу речовини за формулами | ω(чист.реч) = 1 - ω(дом≥шок) ω(—а—ќ3) = 1 Ц 0,2 = 0,8 m(ч.реч)= ω(чист.реч) Ј m(вапн€ку) m(—а—ќ3) = 0,8 Ј 400 = 320 (кг) |
| 4) —клад≥ть р≥вн€нн€ реакц≥њ | —а—ќ3 → CaO + CO2 |
| 5) ѕ≥дкресл≥ть формули речовин, про €к≥ йдетьс€ в умов≥ задач≥ | —а—ќ3 → CaO + CO2 |
| 6) Ќадпиш≥ть над п≥дкресленими формулами вих≥дн≥ дан≥ задач≥, п≥д формулами Ц дан≥, €к≥ законом≥рн≥ дл€ р≥вн€нн€ реакц≥њ | 320 кг х кг —а—ќ3 → CaO + CO2 n = 1 моль n = 1 моль ћ = 100 кг/моль ћ = 56 кг/моль m = 100 кг m = 56 кг |
| 7) —клад≥ть пропорц≥ю та обчисл≥ть масу продукту реакц≥њ | 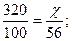 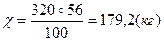
|
| 8) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: m(CaO) = 179,2 кг |
«адач≥ на практичний вих≥д
“рохи теор≥њЕ
ѕрактично при проведенн≥ будь-€кого х≥м≥чного процесу в≥дбуваЇтьс€ втрата де€коњ к≥лькост≥ реагуючих речовин. “ому дл€ одержанн€ бажаноњ маси (чи обТЇму)продукту реакц≥њ необх≥дно враховувати виробнич≥ втрати, тобто розраховувати практичний вих≥д реакц≥њ ≥, навпаки, за практичним виходом розраховувати маси (обТЇми, к≥льк≥сть речовини) реагент≥в. ћаси (обТЇми) продукт≥в реакц≥њ, розрахован≥ за р≥вн€нн€м реакц≥њ, називаютьс€ теоретичним виходом. “еоретичний вих≥д приймають за 100%, тому що одержати продукт≥в реакц≥њ б≥льше, н≥ж ц€ розрахункова величина неможливо. ћаси (обТЇми) продукт≥в реакц≥њ, обчислен≥ з урахуванн€м втрат, називаютьс€ практичним виходом. ѕрактичний вих≥д завжди менший за теоретичний. ¬их≥д продукту розраховують за формулою:
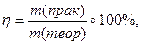 або
або 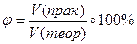
|
|
|
јлгоритм обчисленн€ виходу продукту реакц≥њ
пор≥вн€но з теоретично можливим
| ѕор€док д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | ≤з негашеного вапна масою 112 г одержали гашене вапно масою 120 г. ќбчисл≥ть вих≥д продукту в≥д теоретично можливого. |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: m(—аќ) = 112г mпрак(—а(ќЌ)2) = 120г «найти: η -? |
| 3) «апиш≥ть р≥вн€нн€ реакц≥њ | —аќ + Ќ2ќ = —а(ќЌ)2 |
| 4) ѕ≥дкресл≥ть формули речовин, про €к≥ йдетьс€ в умов≥ задач≥ | —аќ + Ќ2ќ = —а(ќЌ)2 |
| 5) ”каж≥ть над п≥дкресленими формулами вих≥дн≥ к≥лькост≥ речовин, п≥д формулами Ц дан≥, законом≥рн≥ дл€ р≥вн€нн€ реакц≥њ. ”вага! ѕо р≥вн€нню реакц≥њ знаход€ть масу теоретичну! | 112г х г —аќ + Ќ2ќ = —а(ќЌ)2 n = 1 моль n = 1 моль ћ = 56 г/моль ћ = 74 г/моль m = 56 г m = 74 г |
| 6) ќбчисл≥ть теоретичну масу гашеного вапна | 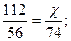 
|
7) ќбчисл≥ть вих≥д продукту в≥д теоретично можливого за формулою:
η = 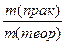 Ј 100% Ј 100%
|
η = 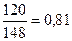 , або 81% , або 81%
|
| 8) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: η(—а(ќЌ)2) = 81% |
«адач≥ на виведенн€ формули орган≥чноњ речовини
јлгоритм обчисленн€ молекул€рноњ формули речовини
за њњ в≥дносною густиною ≥ масою продукт≥в гор≥нн€
| ѕосл≥довн≥сть д≥й | ѕриклади виконаних д≥й |
| 1) ѕрочитайте текст задач≥ | ѕ≥д час спалюванн€ речовини масою 4,6 г утворивс€ карбон (≤V) оксид масою 8,8 г ≥ вода масою 5,4 г. √устина пар≥в ц≥Їњ речовини за пов≥тр€м становить 1,59. ¬ивед≥ть молекул€рну формулу речовини. |
| 2) «апиш≥ть скорочено умову задач≥ | ƒано: m(реч) = 4,6г m(—ќ2) = 8,8г m(Ќ2ќ) = 5,4г Dпов = 1,59 «найти: ‘ормула -? |
3) ќбчисл≥ть мол€рну масу речовини за формулою: D х = 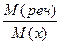
| Dпов = 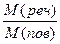 ; ћ(пов) = 29 г/моль
ћ(реч) = ћ(пов) Ј Dпов = 29 Ј 1,59 =
= 46 г/моль ; ћ(пов) = 29 г/моль
ћ(реч) = ћ(пов) Ј Dпов = 29 Ј 1,59 =
= 46 г/моль
|
| 4) «найд≥ть масу арбону в нев≥дом≥й речовин≥ | —ќ2 — ћ(—ќ2) = 44г/моль
44г м≥стить 12г
8,8г -------- ’г
х = 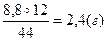
|
| 5) «найд≥ть масу √≥дрогену в нев≥дом≥й речовин≥ | Ќ2ќ Ќ ћ(Ќ2ќ) = 18 г/моль
18г м≥стить 2г х = 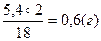 5,4г ------- ’г
5,4г ------- ’г
|
| 6) ¬изначте, чи входить до складу речовини ќксиген | m(ќ) = m(реч) Ц m(—) Ц m(Ќ) m(ќ) = 4,6 Ц 2,4 Ц 0,6 = 1,6 (г) |
| 7) «найд≥ть ≥ндекси дл€ арбону, √≥дрогену та ќксигену | —: Ќ: ќ = = 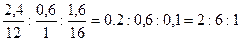
|
| 8) «апиш≥ть найпрост≥шу та ≥стинну формули | Ќайпрост≥ша формула: —2Ќ6ќ ѕерев≥римо, чи Ї вона ≥стинною. ƒл€ цього визначимо ћ знайденоњ речовини. ћ(—2Ќ6ќ) = 12Ј2 + 1Ј6 + 16 = 46 г/моль n = 46/46 = 1 (перев≥рка з числом у 1 д≥њ) |
| 9) «апиш≥ть в≥дпов≥дь | ¬≥дпов≥дь: —2Ќ6ќ |
ƒодатки
1. ¬алентност≥ де€ких х≥м≥чних елемент≥в
| ≈лементи з≥ сталою валентн≥стю |
| ќдновалентн≥: Ќ, , Na, јg, F |
| ƒвовалентн≥: ќ, ¬а, —а, ћg, Zn |
| “ривалентн≥: јl, ¬ |
| ≈лементи з≥ зм≥нною валентн≥стю |
| Fe Ц ≤≤, ≤≤≤ |
| —u Ц ≤, ≤≤ |
| јs, – Ц ≤≤≤, V |
| –b, Sn, — Ц ≤≤, ≤V |
| S Ц ≤≤, ≤V, V≤ |
| —l, ¬r, ≤ Ц ≤, ≤≤≤, V, V≤≤ |
2. ƒе€к≥ позначенн€
+δ, -δ Ц зар€д на атом≥ (в молекул≥, сполуц≥); записують над символом елемента.
н.у. Ц нормальн≥ умови: тиск 101,3 кѕа, або 760 мм рт. ст., температура 0∞—, або 273 .
е- - електрон.
р+ - протон.
n0 - нейтрон.
 t (у р≥вн€нн≥ реакц≥њ) Ц реакц≥€ в≥дбуваЇтьс€ при нагр≥ванн≥.
t (у р≥вн€нн≥ реакц≥њ) Ц реакц≥€ в≥дбуваЇтьс€ при нагр≥ванн≥.
 – (у р≥вн€нн≥ реакц≥њ) Ц реакц≥€ в≥дбуваЇтьс€ за п≥двищеного тиску.
– (у р≥вн€нн≥ реакц≥њ) Ц реакц≥€ в≥дбуваЇтьс€ за п≥двищеного тиску.
 k (у р≥вн€нн≥ реакц≥њ) Ц реакц≥€ в≥дбуваЇтьс€ за участ≥ к≥тал≥затора.
k (у р≥вн€нн≥ реакц≥њ) Ц реакц≥€ в≥дбуваЇтьс€ за участ≥ к≥тал≥затора.
↑ (у р≥вн€нн≥ реакц≥њ п≥сл€ формули речовини) Ц вид≥ленн€ газу (речовини в газуватому стан≥).
↓ (у р≥вн€нн≥ реакц≥њ п≥сл€ формули речовини) Ц утворенн€ осаду речовини в розчин≥.
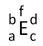 ≈ Ц узагальнений символ елемента:
≈ Ц узагальнений символ елемента:
a Ц пор€дковий номер (протонне число) елемента;
b Ц нуклонне число;
c Ц ≥ндекс (к≥льк≥сть атом≥в елемента);
d Ц зар€д простого йона (спочатку записують значенн€, пот≥м знак);
f Ц ступ≥нь окисненн€ елемента (спочатку записують знак, пот≥м значенн€.
≈* - атом елемента ≈ у збудженому стан≥.






