’од опыта и результаты наблюдений опыта.
ƒл€ выполнени€ эксперимента использовались металлы и растворы солей, представленные в табл.1.“ак как исследуемые реакции €вл€ютс€ окислительно-восстановительными и условием их протекани€ €вл€етс€ неравенство 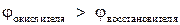 , выписали из табл.ѕ. 2 пособи€ значени€ φ0 дл€ следующих сопр€женных пар:
, выписали из табл.ѕ. 2 пособи€ значени€ φ0 дл€ следующих сопр€женных пар:
 _______
_______
 _______
_______
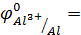 ________
________
 ______
______
φ0 Fe3+/Fe2+ =
¬еличина φ0 металла указывает на меру восстановительной способности
атомов металлов и меру окислительной способности их ионов и, чем больше ее значение, тем €рче выражена окислительна€ способность. »з двух возможных реакций наиболее предпочтительна та, дл€ которой неравенство φо к > φ восст. выполн€етс€ в большей степени. –езультаты теоретического прогноза омечены в табл.1.
“аблица 1.
| ћеталлы | CuSO4 | ZnSO4 | Al2(SO4)3 | FeCl3 (индикатор K3[Fe(CN)6]) |
| Zn | ||||
| Cu | ||||
| Al |
¬ ходе эксперимента проводили только те взаимодействи€, которые возможны в соответствии с установленным условием их протекани€.
јнализа результатов опыта
1. ќбъ€сните по€вление синего окрашивани€ растворов в пробирках с FeCl3 и K3[Fe(CN)6], а также отсутствие признаков реакций Al с растворами солей CuSO4 и ZnSO4 —оставьте уравнение реакции ионов Fe2+ c индикатором K3[Fe(CN)6] и образованием турбулевой сини Fe3[Fe(CN)6]2, а также уравнени€ всех реакций в молекул€рной и краткой ионной формах.
2. акой из исследованных металлов самый активный и какой наименее? –асположите исследуемые металлы по убыванию их активности в водных растворах.
3. ”кажите вли€ние пассивирующих (оксидных) слоев на поверхности активных металлов на характер протекани€ окислительно-восстановительных реакций.
______________________________________________________________________________________
2. ќпределение стандартной Ёƒ— химического гальванического элемента
’од и данные опыта
ƒл€ выполнени€ данного опыта собрали из стандартных электродов 1-го рода медно-цинковый гальванический элемент, исользу€ два химических стакана, емкостью 100мл, 1 ћ растворы солей ZnSO4 и CuSO4, предварительно протравленные электроды из меди и цинка. Ёлектролиты соединили электролитическим мостиком. «начение Ёƒ— определили с помощью цифрового милливольтметра.
јнализа результатов опыта
1. —оставьте электрохимическую схему исследованного гальванического элемента в молекул€рной и ионной формах, уравнени€ анодно-катодных процессов и суммарное уравнение токообразующей реакции. «апишите значение измеренной стандартной Ёƒ—.
________________________________ ________________________________________________________
2. »спользу€ данные табл. ѕ.2, рассчитайте значение стандартной Ёƒ— и сравните с экспериментальным. –ассчитайте изменение свободной энергии √иббса (∆G0), максимально полезную работу (јм) и константу равновеси€ ( –).
|
|
|
______________________________________________________________________________
3. Ќе провод€ эксперимента, рассчитайте значени€ Ёƒ— исследуемого элемента при концентраци€х растворов:
а) 0,001 ћ ZnSO4 и 1 ћ —uSO4; б)1 ћ ZnSO4 и 0,001 M —uSO4.
_______________________________________________________________________________________________
—равните полученные значени€ Ёƒ— с величиной стандартной Ёƒ— и сделайте выводо вли€нии концентраций потенциалопредел€ющих ионов на величины электродных потенциалов и значени€ Ёƒ—.
___________________________________________________________________________
4. ак измен€тс€ процессы, если Zn- и Cu-электроды (оба) поместить в один и тот же раствор серной кислоты (H2SO4)? ак называетс€ такой элемент? ќтвет обосновать приведением электрохимической схемы элемента, уравнений анодно-катодных процессов и токообразующей реакции.
____________________________________________________________________________
5. ќбъ€сните, почему с течением времени в таком элементе значение Ёƒ— уменьшаетс€? ака€ пол€ризаци€ имеет место в элементе ¬ольта? акими способами можно уменьшить пол€ризацию и увеличить численное значение Ёƒ—?
____________________________________________________________________________________
6. —делайте обобщающий вывод, от каких факторов зависит численное значение Ёƒ— химических гальванических элементов.
__________________________________________________________________________________
Ёлектролиз растворов солей на инертных электродах
’од и данные опыта
¬ опыте исследовали реакции, протекающие на графитовых электродах при прохождении посто€нного тока через растворы солей CuSO4, KI, NaCl. ѕри выполнении работы использовали: электролизер (U‑образна€ стекл€нна€ трубка, закрепленна€ в штативе), графитовые электроды, выпр€митель тока ≈‑24ћ или »Ёѕѕ‑2 и 0,5 ћ растворы солей CuSO4, KI, NaCl.
Ёлектролиз сульфата меди. Ќапр€жение 13-20 ¬, в течение 2-3 мин. Ќа катоде выделилась медь, на аноде наблюдали выделение пузырьков газа. ¬ анодное пространство опустили полоску индикаторной бумаги и по цветавой эталонной шкале определили значение рЌ =
Ёлектролиз иодида кали€. ќпыт проводили, использу€ предыдущую методику, наблюда€ выделение пузырьков газа на катоде и окрашивание в светлокоричневый цвет анодного электролита. ѕосле добавлени€ в катодное пространство фенолфталеина, в анодное Ц крахмала, растворы приобрели малиновый и синий цвета соответственно.
Ёлектролиз хлорида натри€. »спользу€ методику предыдущих опытов провели электролиз, наблюда€ выделение пузырьков газа как на аноде, так и
на катоде. ѕосле добавлени€ в катодное пространство фенолфталеина, в анодное иодокрахмального раствора ( J + крахмал) наблюдали малиновую окраску католита и синюю Ц анолита.
јнализа результатов опыта
1. ƒл€ каждого из трех опытов составьте схемы электролизных систем, уравнени€ первичных и вторичных анодно-катодных процессов и суммарные уравнени€ электролиза. ак и почему изменились при этом значени€ рЌ электролитов?
2. ѕочему на катоде не выдел€ютс€ металлические калий и натрий, а медь выдел€етс€? ак это св€зано с их активностью?
|
|
|
3. Ќа примере электролиза раствора NaCl объ€сните вли€ние пол€ризации и перенапр€жени€ на характер анодной реакции. «апишите уравнение качественной реакции на молекул€рный хлор.
3. —делайте обобщающий вывод об особенност€х процессов электролиза на инертных электродах, указав дл€ каждого из трех опытов основные факторы, определ€ющие природу продуктов электролиза.
______________________________________________________________________________.____________________________________________________________________ ___________________________________________________________
__________________________________________________________________ ____________________________________________________________________
4._ ќтветы на контрольные вопросы






