√идроксиды €вл€ютс€ характеристическими соединени€ми (в водных растворах*), поскольку свойства гидроксида определ€ют в конечном итоге принадлежность элемента к металлам или неметаллам.
*ќксид азота N2ќ5 с водой образует гидроксид ЌNќ3, который в водном растворе €вл€етс€ сильной кислотой (т.е. азот Ц неметалл, а N2ќ5 Ц кислотный оксид). Ќо в среде жидкого фтороводорода азотна€ кислота ведет себ€ как типичное основание, присоедин€€ протон:
ЌNќ3 + ЌF = Ќ2Nќ3+ + F⎺.
ќбща€ формула гидроксидов Ц Ё − (ќЌ) ’, где х Ц степень окислени€ элемента. ћаксимальное значение х = 8, однако координационные сферы с большим числом гидроксогрупп неустойчивы. ѕовышение устойчивости достигаетс€ путем уменьшени€ координационного числа за счет отщеплени€ одной или нескольких молекул воды. „ем выше степень окислени€ элемента, а следовательно, меньше размер ион-атома, тем большее число молекул воды отщепл€етс€ дл€ обеспечени€ устойчивости координационной сферы. Ёто положение характерно дл€ гидроксидов с кислотными свойствами, т.е. дл€ гидроксидов неметаллов и металлов со степенью окислени€ >+3.
ислотные гидроксиды, номинально отвечающие высшим степен€м окислени€ элементов IIIЦIV групп ѕериодической —истемы.
| ѕериод | √руппа | |||||
| III | IV | V | VI | VII | VIII | |
| ¬(ќЌ)3 | —(ќЌ)4 | N(ќЌ)5 | Ц | Ц | Ц | |
| Ц | Si(ќЌ)4 | –(ќЌ)5 | S(ќЌ)6 | —l(ќЌ)7 | Ц | |
| “i(ќЌ)4 | V(ќЌ)5 | —r(ќЌ)6 | ћn(ќЌ)7 | Ц | ||
| Gе(ќЌ)4 | јs(ќЌ)5 | Sе(ќЌ)6 | ¬r(ќЌ)7 | Ц | ||
| Zr(ќЌ)4 | Nb(ќЌ)5 | ћо(ќЌ)6 | Ц | Ru(ќЌ)8 | ||
| Sn(ќЌ)4 | Sb(ќЌ)5 | “е(ќЌ)6 | I(ќЌ)7 | Ц | ||
| Ќf(ќЌ)4 | “а(ќЌ)5 | W(ќЌ)6 | Rе(ќЌ)7 | ќs(ќЌ)8 |
¬о втором периоде при переходе ¬+3 Ц —+4 Ц N+5 атомный радиус уменьшаетс€.
ѕоэтому дл€ бора характерно сохранение координационного окружени€ из трех гидроксогрупп в ортоборной кислоте (хот€ известны производные и метаборной кислоты Ќ¬ќ2).
ƒл€ углерода повышение координационной устойчивости достигаетс€ за счет удалени€ одной молекулы воды и замены двух лигандов −ќЌ на один лиганд =ќ:

ƒл€ азота более устойчивой оказываетс€ структура, образованна€ удалением двух молекул воды:

”стойчивые формы кислотных гидроксидов 3-го периода подчин€ютс€ той же закономерности: с увеличением номера группы возрастает число отщепленных молекул воды, поскольку в этом направлении размеры атомов уменьшаютс€. ќднако в пределах групп (—+4 Ѓ Si+4, N+5 Ѓ –+5) атомные радиусы возрастают. ѕоэтому дл€ кремни€ и фосфора, помимо мета- форм кислот (Ќ2Siќ3 и Ќ–ќ3), известны и орто- формы (Ќ4Siќ4 и Ќ3–ќ4). — учетом существовани€ ортокремневой и ортофосфорной кислот дл€ элементов 3-го периода наблюдаетс€ обща€ закономерность:
Si(OH)4 ≡ H4SiO4
P(OH)5 Ц H2O Ѓ PO(OH)3 [H3PO4]
S(OH)6 Ц 2H2O Ѓ SO2(OH)2 [H2SO4]
Cl(OH)7 Ц 3H2O Ѓ ClO3(OH) [HClO4]
т.е. с ростом степени окислени€ и уменьшением размера центрального атома число отщепленных молекул воды увеличиваетс€.
ƒл€ гидроксидов более т€желых р-элементов отмечаетс€ тенденци€ к увеличению количества св€занных гидроксоанионов, что также обусловлено возрастанием атомного радиуса элемента при переходе сверху вниз в пределах каждой группы. ѕри этом у элементов 4-го периода (Gе, јs, Sе, ¬r) сохран€етс€ та же тенденци€, что и у их аналогов из 3-го периода (Si, –, S, —l), поскольку атомные радиусы в направлении SiЃGе, –Ѓјs, SЃSе, —lЃ¬r возрастают незначительно. Ёто обусловлено d-сжатием в силу наличи€ у р-элементов 4-го периода завершенной кайносимметричной 3d-оболочки. ¬следствие этого формулы соответствующих высших кислот имеют вид Ќ4Gеќ4, Ќ3јsќ4 (Ц1Ќ2ќ), Ќ2Sеќ4 (Ц2Ќ2ќ) и Ќ¬rќ4 (Ц3Ќ2ќ). Ќо дл€ р-элементов 5-го периода характерны более богатые водой гидратные формы. Ќар€ду с Ќ2“еќ4 известна и ортотеллурова€ кислота Ќ6“еќ6 (“е(ќЌ)6 без потери молекул воды). ј дл€ иода кроме ЌIќ4 известна и Ќ5Iќ6 с потерей всего 1 молекулы воды от гидроксида номинального состава I(ќЌ)7.
|
|
|
“аким образом, наиболее распространенными гидратными формами р-элементов €вл€ютс€ те, формулы которых можно получить путем вычитани€ из предельной формы Ё(ќЌ)n четного числа молекул воды дл€ элементов четных групп и нечетного Ц дл€ элементов нечетных групп. »сключение составл€ют бор, углерод и азот с кайносимметричными 2р-орбитал€ми.
ислотные гидроксиды переходных металлов в высших степен€х окислени€ подчин€ютс€ той же закономерности: с увеличением номера группы число отщепленных молекул воды возрастает. ƒл€ гидроксидов кайносимметричных 3d-элементов наиболее характерны мета -формы: Ќ2“iќ3 (Ц1Ќ2ќ), ЌVќ3 (Ц2Ќ2ќ), Ќ2—rќ4 (Ц2Ќ2ќ) и Ќћnќ4 (Ц3Ќ2ќ). Ёлементы триады железа не про€вл€ют степеней окислени€, отвечающих номеру группы. ” d-элементов 5-го и 6-го периодов помимо мета -форм известны и орто -производные: Ќ4Zrќ4, Ќ3Nbќ4 (Ц1Ќ2ќ). √идроксид рутени€ в степени окислени€ +8 не существует, поскольку в соответствии с общей закономерностью от Ru(ќЌ)8 должны отщепитьс€ 4 молекулы воды, что приводит к составу Ruќ4. ќсобенно нагл€дно эта тенденци€ прослеживаетс€ у d-элементов 6-го периода (у Ќ4Ќfќ4 вода не отщепл€етс€):
“а(ќЌ)5 Ц Ќ2ќ Ѓ Ќ3“аќ4;
W(ќЌ)6 Ц 2Ќ2ќ Ѓ Ќ2Wќ4;
Re(ќЌ)7 Ц 3Ќ2ќ Ѓ ЌRеќ4;
ќs(ќЌ)8 Ц 4Ќ2ќ Ѓ ќsќ4.
ќксиды рутени€ и осми€ в высшей степени окислени€ не могут химически взаимодействовать с водой с образованием устойчивых гидратных форм.
“аким образом, общую формулу гидроксидов правильнее представл€ть не в виде Ё(ќЌ)n, а в виде Ёќm(ќЌ)n, где m = 0÷4, а n = 0÷6 в реально существующих гидроксидах. ѕри m = 0 гидроксиды Ё(ќЌ)n могут обладать как основным, так и кислотным характером, причем с увеличением числа гидроксогрупп в формульной единице (т.е. с увеличением степени окислени€ Ё) основные свойства ослабевают, а кислотные нарастают. Ёто можно объ€снить ростом пол€ризующего действи€ катионообразовател€ в пределах каждого периода в св€зи с уменьшением его радиуса и увеличением степени окислени€, например в р€ду NаќЌ, ћg(ќЌ)2, јl(ќЌ)3, Si(ќЌ)4. ¬ общем случае дл€ гидроксидов типа Ё(ќЌ)n (m = 0) при переходе сверху вниз в пределах каждой группы основный характер гидроксида усиливаетс€, что св€зано с ослаблением пол€ризующего действи€ катионообразовател€ за счет увеличени€ его радиуса.
√идроксиды Ёќm(ќЌ)n (m ≠ 0) обладают исключительно кислотными свойствами. Ёто объ€сн€етс€ тем, что атомы кислорода, ковалентно св€занные с элементом, способствуют пол€ризации св€зи ќ−Ќ, в силу чего и облегчаетс€ диссоциаци€ по кислотному типу с отщеплением протона. ќчевидно, с ростом m (числа ковалентно св€занных с элементом кислородных лигандов) сила кислот должна возрастать. ќт величины n сила кислоты практически не зависит, поскольку дл€ многоосновных кислот концентраци€ протонов в растворе определ€етс€ в основном первой константой диссоциации:
|
|
|
Ёќm(ќЌ)n = Ќ+ + [Ёќm(ќЌ)n−1ќ]⎺
ѕравило ѕолинга:
ислоты типа Ё(ќЌ)n (m = 0) принадлежат к очень слабым электролитам (Ќ3¬ќ3, Ќќ—l, Ќ6“еќ6).
ислоты типа Ёќ(ќЌ)n (m = 1) Ц слабые электролиты (Ќ—lќ2, ЌNќ2, Ќ2—ќ3).
ислоты типа Ёќ2(ќЌ)n (m = 2) сильные (ЌNќ3, Ќ2Sќ4)
ислоты типа Ёќ3(ќЌ)n (m = 3) очень сильные (Ќ—lќ4)
Ётот подход позвол€ет объ€снить изменение кислотно-основного характера гидроксидов в зависимости от степени окислени€ элементов. “ак, азотиста€ кислота ЌNќ2 или Nќ(ќЌ), слабее азотной ЌNќ3 или Nќ2(ќЌ), серниста€ Ќ2Sќ3 или Sќ(ќЌ)2 слабее серной Ќ2Sќ4 или Sќ2(ќЌ)2.
¬ р€ду Mn(ќЌ)2, Mn(ќЌ)3, Mn(ќЌ)4, Ќ3Mnќ4 (Mnќ(ќЌ)3), Ќ2Mnќ4 (Mnќ2(ќЌ)2), ЌMnќ4 (Mnќ(ќЌ)3) дл€ гидроксидов Ё(ќЌ)n с увеличением n ослабевает основный характер, а дл€ Mnќ3(ќЌ)), с возрастанием m усиливаютс€ кислотные свойства. ѕри этом гидроксиды Mn(ќЌ)3 и Mn(ќЌ)4 про€вл€ют амфотерность. ’от€ оба гидроксида принадлежат к очень слабым электролитам, у первого €рче выражены основные, а у второго Ц кислотные свойства.
— общей точки зрени€ кислотно-основные свойства гидроксидов, как и кислотно-основные свойства других соединений, могут быть выделены лишь при химическом взаимодействии. Ёти свойства €вл€ютс€ про€влением единого процесса кислотно-основного взаимодействи€. ислотный характер соединени€ становитс€ очевидным лишь при взаимодействии с соединени€ми, обладающими основными функци€ми, и наоборот. ѕоэтому амфотерность в известном смысле можно рассматривать как универсальное свойство гидроксидов. Ќапример, ћn(ќЌ)2 и —u(ќЌ)2 обладают преимущественно основным характером. ќднако в жестких услови€х Ц при кип€чении с концентрированными растворами щелочей, а тем более при сплавлении со щелочами Ц образуютс€ соединени€, отвечающие кислотной функции этих гидроксидов.
— другой стороны, ЌNќ3 в большинстве случаев про€вл€ет кислотные свойства. ќднако дл€ ЌNќ3 можно обнаружить и основную функцию, например при взаимодействии с Ќ—lќ4 и Ќ2Sќ4:
ЌNќ3 + Ќ—lќ4 = Ќ3ќ+ + Nќ2+ + 2—lќ4⎺
ЌNќ3 + Ќ2Sќ4 = Ќ3ќ+ + Nќ2+ + 2ЌSќ4⎺
ѕри этом образуютс€ перхлорат и гидросульфат нитроила.
“аким образом, в зависимости от степени про€влени€ кислотно-основных свойств характер диссоциации гидроксидов в водной среде, а также и особенности взаимодействи€ оксидов с водой закономерно измен€ютс€.
| ’арактер диссоциации гидроксидов | ||
| ќсновные гидроксиды | јмфотерные гидроксиды | ислотные гидроксиды |
| —лабоосновные гидроксиды диссоциируют с образованием гидроксокатионов и гидроксидионов: Fе(ќЌ)2 = Fе(ќЌ)+ + ќЌ⎺ | јмфотерные оксиды с преобладанием кислотных свойств образуют с водой гидроксоанионы в щелочной среде: —еќ2 + 2Ќ2ќ + 2ќЌ⎺ = [—е(ќЌ)6]2− | —лабокислотные оксиды образуют при взаимодействии с водой соответствующие кислоты, которые диссоциируют преимущественно до кислых анионов: Sќ2 + Ќ2ќ ⇄ ЌSќ3⎺ + Ќ+ |
| √идроксиды типичных металлов с сильноосновным характером в водных растворах образуют гидратированные простые катионы (аквакомплексы): NаќЌ + аq = Nа+Јаq + ќЌ⎺ | јмфотерные оксиды с преобладанием основных свойств образуют с водой гидроксокатионы в кислой среде: Zn(ќЌ)2 + Ќ+ = Zn(ќЌ) + + Ќ2ќ | ќксиды типичных неметаллов с водой дают сильные кислоты, диссоциирующие с образованием оксоанионов: Sќ3 + Ќ2ќ Ѓ Sќ42− + 2Ќ+ |
| ≈сли кислотные и основные свойства амфотерного гидроксида выражены примерно в одинаковой степени, то возможны взаимодействи€ по обеим схемам: Gа(ќЌ)2+ + Ќ2ќ Gа(ќЌ)3 Ѓ [Gа(ќЌ)6]3− |
ќ—Ќќ¬јЌ»я M+x(OH)X ⎺
ќснованием называетс€ соединение, образующее при диссоциации в водном растворе из отрицательных ионов только гидроксид-ионы (определение с точки зрени€ “ЁЋƒ)
|
|
|
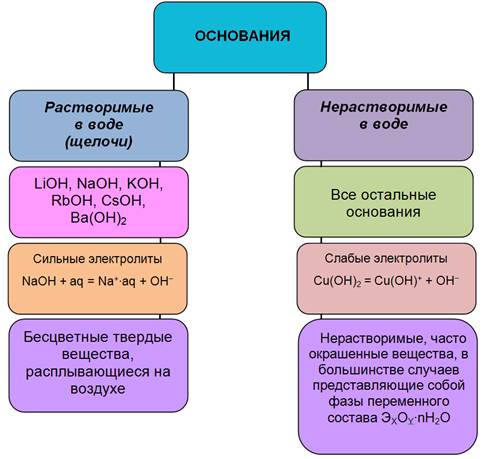

ѕолучение оснований
1. ѕолучение щелочей
1.1. 2Na + 2H2O Ѓ 2NaOH + H2;
1.2. BaO + H2O Ѓ Ba(OH)2;
1.3. NaH + H2O Ѓ NaOH + H2;
1.4. 2NaCl Ц(электролиз)Ѓ 2NaOH + H2 + Cl2;
1.5. K2SO4 + Ba(OH)2 Ѓ BaSO4 + 2KOH; Na2CO3 + Ca(OH)2 Ѓ CaCO3 + 2NaOH.
2. ѕолучение нерастворимых оснований и гидрата аммиака
2.1. –еакци€ обмена
CuCl2 + Ba(OH)2 Ѓ BaCl2 + Cu(OH)2;
ZnCl2 + 2NH3ЈH2ќ Ѓ 2NH4Cl + Zn(OH)2;
¬ избытке щелочи амфотерный гидроксид раствор€етс€. Ћучше брать водный раствор аммиака, с избытком которого амфотерный гидроксид не реагирует.
Ќапример: AlCl3 + 3KOH Ѓ Al(OH)3 + 3KCl;
в избытке щелочи Al(OH)3 растворитс€ AlCl3 + 6KOH Ѓ K3[Al(OH)6] + 3KCl;
AlCl3 + 3NH3ЈH2ќ Ѓ Al(OH)3 + 3NH4Cl Ц в этом случае образуетс€ осадок гидроксида алюмини€, который не раствор€етс€ в избытке водного раствора аммиака.
2.2. √идрат аммиака получают растворением аммиака в воде: NH3 + H2O Ѓ NH3ЈH2ќ;
или по реакции обмена в водном растворе:
(NH4)2SO4 + Ba(OH)2 Ѓ BaSO4 + 2NH3ЈH2ќ.
2.3. –еакции замещени€ Mg + 2H2O Ѓ Mg(OH)2 + H2.
—войства оснований
—войства щелочей
1.1. »зменение окраски индикаторов:
лакмус Ц синий; фенолфталеин Ц малиновый; метилоранж Ц желтый
1.2. ¬заимодействие с кислотными оксидами:
2KOH + CO2 Ѓ K2CO3 + H2O;
KOH + CO2 Ѓ KЌCO3;
Ba(OH)2 + SO3 Ѓ BaSO4 + H2O.
1.3. ¬заимодействие с кислотами (реакци€ нейтрализации):
KOH + HCl Ѓ KCl + H2O;
2NaOH + H2SO4 Ѓ Na2SO4 + 2H2O;
NaOH + H2SO4 Ѓ NaЌSO4 + H2O;
Ba(OH)2 + H2SO4 Ѓ BaSO4 + H2O;
1.4. –еакции обмена с сол€ми:
2KOH + 2CuSO4 Ѓ K2SO4 + [Cu(OH)]2SO4;
2KOH + CuSO4 Ѓ K2SO4 + Cu(OH)2;
NaOH + FeCl3 Ѓ Fe(OH)Cl2 + NaCl;
2NaOH + FeCl3 Ѓ Fe(OH)2Cl + 2NaCl;
3NaOH + FeCl3 Ѓ Fe(OH)3 + 3NaCl;
NaHCO3 + NaOH Ѓ Na2CO3 + H2O.
1.5. ¬заимодействие с неметаллами (Cl, S, Si, P)
6KOH + 3Cl2 Ц(t∞)Ѓ 5KCl + KClO3 + 3H2O;
2KOH + Cl2 Ѓ KCl + KClO + H2O;
Si (аморфн.) + 4NaOH (конц.) Ѓ Na4SiO4 + 2H2;
–4 + 8NaOH (конц.) + 4Ќ2O = 4Na2(PHO3) + 6Ќ2≠ (кип.);
–4 + 3NaOH (конц.) + 3Ќ2O (хол.) Ц(t)Ѓ 3Na(PH2O2) + PH3;
2–4 + 3¬а(ќЌ)2 (конц.) + 6Ќ2O = 3¬а(–Ќ2O2)2 + 2PH3≠ (70∞—).
1.6. ¬заимодействие с амфотерными металлами (Al, Zn)
2Al + 2KOH + 6H2O Ѓ 2K[Al(OH)4] + 2H2
1.7. ¬заимодействие с амфотерными оксидами и гидроксидами
јl2O3 + 2NaOH (конц., гор.) + 3Ќ2O Ѓ 2Na[Al(OH)4];
јl2O3 + 2NaOH Ѓ 2NaAlO2 + Ќ2O (900Ц1100∞—);
јl(ќЌ)3 + NaOH (конц.) Ѓ Na[Al(OH)4].
1.8. “ермолиз. »з гидроксидов щелочных металлов термолизу подвергаетс€ только гидроксид лити€. ¬ случае щелочноземельных металлов все гидроксиды термически неустойчивы.
2LiќЌ Ѓ Li2O + Ќ2O (800Ц1000∞—, в атмосфере Ќ2).
¬а(ќЌ)2 Ѓ ¬аќ + Ќ2O (780Ц800∞—).
2. —войства нерастворимых оснований
2.1. ¬заимодействие с кислотами (реакци€ нейтрализации):
Cu(OH)2 + HCl Ѓ Cu(OH)Cl + H2O;
Cu(OH)2 + 2HCl Ѓ CuCl2 + 2H2O;
Cu(OH)2 + 2NaHSO4 Ѓ Na2SO4 + CuSO4 + 2H2O;
2.2. “ермолиз.
—u(ќЌ)2 (суспензи€) Ѓ —uќ + Ќ2ќ (40Ц80∞—);
—u(ќЌ)2 Ѓ —uќ + Ќ2ќ (200∞—);
Fe2O3∙nЌ2O Ѓ 2FeO(OH) + (nЦ1)Ќ2O (200Ц250∞—),
Fe2O3∙nЌ2ќ Ѓ Fe2O3 + nЌ2O (500Ц700∞—).
2.3. ќкислительно-восстановительное взаимодействие.
4Fe(OH)2 (суспензи€) + O2 (воздух) Ѓ 4FeO(OH)¯ + 2H2O (кип.);
4Cu(OH)2 + N2H4ЈH2O Ѓ 2Cu2O¯ + N2≠ + 7Ќ2ќ (кип.).
2.4. омплексообразование
—u(ќЌ)2 + 4(NЌ3ЈЌ2ќ) [конц.] Ѓ [Cu(NH3)4](ќЌ)2 + 4Ќ2ќ;
2Cu(OH)2 + NaEO + 2NaOH (конц.) + H2O Ѓ 2Na[Cu(OH)4] + NaE (E = Cl, Br);
јl(ќЌ)3 + NaOH (конц.) Ѓ Na[Al(OH)4].
»—Ћќ“џ HX+1R−x
— точки зрени€ теории электролитической диссоциации кислота Ц соединение, образующее при диссоциации в водном растворе из положительных ионов только протоны Ќ+(согласно “ЁЋƒ).
|
|
|



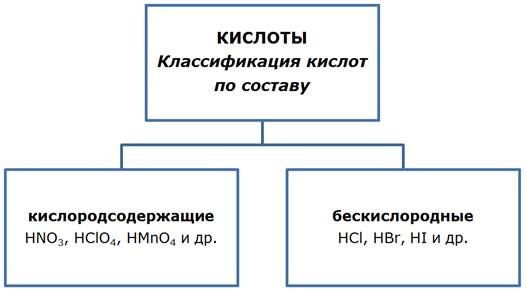


ѕолучение кислот
1.1. ¬заимодействие простых веществ (получение газов, растворение в воде которых ведет к получению водных растворов бескислородных кислот)
H2 + Cl2 Ѓ 2HCl;
H2 + S Ѓ H2S.
1.2. ¬заимодействие кислотных оксидов с водой.
SO2 + H2O ⇄ H2SO3;
SO3 + H2O Ѓ H2SO4.
1.3. –еакции обмена:
CuSO4 + H2S Ѓ CuS¯ + H2SO4;
FeS + H2SO4 Ѓ H2S≠ + FeSO4;
AgNO3 + HCl Ѓ AgCl¯ + HNO3.
1.4. ќкислительно-восстановительное взаимодействие.
H2O + SO2 + H2O Ѓ H2SO4 + H2O;
3P + 5HNO3 + 2H2O Ѓ 3H3PO4 + 5NO.
—войства кислот
2.1. ќбщие свойства
2.1.1. »зменение окраски индикатора
лакмус Ц красный; фенолфталеин Ц бесцветный; метилоранж Ц красный
2.1.2. ¬заимодействие с металлами
а) кислоты-неокислители реагируют с металлами, сто€щими в р€ду напр€жений до водорода, образу€ соль и металл
Zn + 2HCl Ѓ ZnCl2 + H2.
б) кислоты-окислители в реакци€х с металлами образуют соль, воду и продукт восстановлени€ центрального атома кислоты
Cu + 2H2SO4 (конц.) Ѓ CuSO4 + SO2 + 2H2O
2.1.3. –еакции нейтрализации
HCl + KOH Ѓ KCl + H2O;
2HCl + CuO Ѓ CuCl2 + H2O;
2HNO3 + Zn(OH)2 Ѓ Zn(NO3)2 + 2H2O.
2.1.4. –еакции обмена:
2HCl + CaCO3 Ѓ CaCl2 + CO2 + H2O;
2H2SO4 + Na2[Zn(OH)4] Ѓ Na2SO4 + ZnSO4 + 4H2O.
2.2. —пецифические свойства кислот
2.2.1. ќкислительно-восстановительное взаимодействие.
16HCl + 2KMnO4 Ѓ 5Cl2 + 2KCl + 2MnCl2 + 8H2O;
H2S + Br2 Ѓ S + 2HBr;
H2SO3 + Cl2 + H2O Ѓ H2SO4 + 2HCl.
2.2.2. “ермолиз.
H2SO4 (безводн.) Ѓ Ќ2O + SO3 (450∞—).
2ЌI ⇄ Ќ2 + I2 (выше 200º—)
“≈ќ–»» »—Ћќ“ » ќ—Ќќ¬јЌ»…
ислоты и основани€. “ермины Ђкислотыї и Ђосновани€ї вполне сформировались в 17 в. »х содержание неоднократно пересматривалось и дополн€лось. Ётот процесс происходил и происходит в острых столкновени€х представителей разных взгл€дов на природу кислот и оснований.






