5.1. Ќаписать в ионно-молекул€рной форме уравнени€ реакций, привод€щих к образованию малорастворимых осадков или газов:
а) Pb(NO3)2 + KI; б) NiCl2 + H2S; в) K2CO3 + HCl; г) CuSO4 + NaOH;
д) CaCO3 + HCl; е) Na2SO3 + H2SO4; ж) AlBr3 + AgNO3.
5.2. Ќаписать в ионно-молекул€рной форме уравнени€ реакций, привод€щих к образованию малодиссоциированных соединений: а) Na2S + H2SO4; б) FeS + HCl; в) HCOOK + HNO3; г) NH4Cl + Ca(OH)2; д) NaOCl + HNO3.
5.3. Ќаписать в ионно-молекул€рной форме уравнени€ реакций нейтрализации: а) HCl + Ba(OH)2; б) HF + KOH; в) Fe(OH)3 + HNO3; г) CH3COOH + NH4OH; д) HNO2 + NH4OH; е) H2S + NH4OH.
”казать, какие из этих реакций протекают обратимо, а какие Ц необратимо.
5.4. —оставить в молекул€рной форме уравнени€ реакций, которые выражаютс€ следующими ионно-молекул€рными уравнени€ми:
NO2Ц + H+ = HNO2;
Cu2+ + 2OHЦ = Cu(OH)2↓;
Pb2+ + 2I Ц = PbI2↓.
5.5. Ќаписать в ионно-молекул€рной форме уравнени€ реакций взаимодействи€ между водными растворами следующих веществ: а) NaH—O3 и HCl; б) FeCl3 и KOH; в) Pb(CH3COO)2 и Na2S; г) KHS и H2SO4; д) Zn(NO3)2 и KOH (избыток); е) Ca(OH)2 и CO2; ж) Ca(OH)2 и CO2 (избыток).
ƒл€ каждого случа€ указать причину смещени€ равновеси€ в сторону пр€мой реакции.
5.6. ¬ каком направлении будет смещено равновесие реакции
AgI (к.) + NaCl (водн.)  AgCl (к.) + NaI (водн.):
AgCl (к.) + NaI (водн.):
а) в направлении пр€мой реакции; б) в направлении обратной реакции?
5.7. ¬ каком направлении будет смещено в водном растворе равновесие реакции
CH3COONa+CH2ClCOOH  CH3COOH+CH2ClCOONa:
CH3COOH+CH2ClCOONa:
а) в направлении пр€мой реакции; б) в направлении обратной реакции?
¬ј–»јЌ“џ ƒќћјЎЌ»’ «јƒјЌ»…
| ¬ариант | Ќомера задач | |||||||
| 1.9 | 1.2(а) | 3.6 | 2.1 | 2.16 | 4.2(а,в) | 4.19 | 5.6 | |
| 1.1 | 1.3 | 3.1 | 2.2 | 2.17 | 4.3(а,б) | 4.7 | 5.1(в,г) | |
| 1.4 | 1.7 | 3.2 | 2.3 | 2.18 | 4.3(б,в) | 4.17 | 5.5(а,ж) | |
| 1.6 | 1.1 | 3.3 | 2.4 | 2.19 | 4.1(а,б) | 4.11 | 5.4 | |
| 1.8 | 1.2(б) | 3.4 | 2.5 | 2.20 | 4.5 | 4.18 | 5.3(а,б) | |
| 1.9 | 1.3 | 3.5 | 2.6 | 2.25 | 4.6 | 4.9(а,г) | 5.3(в,г) | |
| 1.7 | 1.4 | 3.15 | 2.7 | 2.17 | 4.1(а,в) | 4.12 | 5.3(д,е) | |
| 1.5 | 1.10 | 3.7 | 2.8 | 2.20 | 4.8 | 4.16(а,в) | 5.5(е,ж) | |
| 1.1 | 1.8 | 3.8 | 2.9 | 2.24 | 4.2(а,б) | 4.22 | 5.7 | |
| 1.10 | 1.2(в) | 3.9 | 2.10 | 2.14 | 4.13 | 4.16(б,в) | 5.5(д,е) | |
| 1.11 | 1.4 | 3.10 | 2.1 | 2.26 | 4.4(а,б) | 4.15 | 5.2(г,д) | |
| 1.2(г) | 1.7 | 3.11 | 2.12 | 2.27 | 4.10 | 4.18 | 5.5(г,д) | |
| 1.3 | 1.9 | 3.14 | 2.13 | 2.10 | 4.4(б,в) | 4.20 | 5.1(а,б) | |
| 1.1 | 1.11 | 3.12 | 2.14 | 2.30 | 4.14 | 4.21 | 5.5(б,в) | |
| 1.5 | 1.10 | 3.13 | 2.15 | 2.29 | 4.9(б,в) | 4.13 | 5.1(е,ж) |
ѕ–»Ћќ∆≈Ќ»≈
“аблица 1
онстанты диссоциации некоторых слабых
электролитов в водных растворах при 25о—
| Ёлектролит | онстанта диссоциации | ||
| јзотиста€ кислота | HNO2 | 2,6∙10-5 | |
| јммони€ гидроксид | NH4OH | 4∙10-4 | |
| Ѕорна€ кислота | H3BO3 | 5,8∙10-4 | |
| Ѕромноватиста€ кислота | HOBr | 2,1∙10-9 | |
| ¬одорода пероксид | H2O2 | 2,6∙10-12 | |
| ремниева€ кислота | H2SiO3 | K1 K2 | 2,2∙10-10 1,6∙10-12 |
| ћуравьина€ кислота | HCOOH | 1,8∙10-4 | |
| —ерниста€ кислота | H2SO3 | K1 K2 | 1,6∙10-2 6,3∙10-8 |
| —ероводород | H2S | K1 K2 | 6∙10-8 1∙10-14 |
| ”гольна€ кислота | H2CO3 | K1 K2 | 4,5∙10-7 4,7∙1011 |
| ”ксусна€ кислота | CH3COOH | 1,8∙10-5 | |
| ‘осфорна€ кислота | H3PO4 | K1 K2 K3 | 7,5∙10-3 6,3∙10-8 1,3∙10-12 |
| ÷иановодород | HCN | 7,9∙10-10 | |
| ўавелева€ кислота | H2C2O4 | K1 K2 | 5,4∙10-2 5,4∙10-5 |
|
|
|
“аблица 2
ѕроизведение растворимости некоторых
малорастворимых электролитов при 25о—
| Ёлектролит | ѕ– | Ёлектролит | ѕ– | Ёлектролит | ѕ– |
| AgBr | 5,3Ј10-13 | CaSO4 | 6,1Ј10-5 | MnS | 2,5Ј10-10 |
| Ag2CO3 | 8,2Ј10-12 | Ca3(PO4)2 | 1,0Ј10-29 | Ni(OH)2 | 6,3Ј10-18 |
| AgCl | 1,8Ј10-10 | CdS | 1,6Ј10-28 | PbBr2 | 9,1Ј10-6 |
| Ag2CrO4 | 1,1Ј10-12 | CoCO3 | 1,5Ј10-10 | PbCO3 | 7,5Ј10-14 |
| AgI | 8,3Ј10-17 | Co(OH)2 | 2Ј10-16 | PbCl2 | 1,56Ј10-5 |
| Ag2S | 5,3Ј10-50 | CrPO4 | 2,4Ј10-23 | PbF2 | 2,7Ј10-8 |
| Ag2SO4 | 1,6Ј10-5 | CuCO3 | 2,5Ј10-10 | PbI2 | 1,1Ј10-9 |
| Ag3PO4 | 1,3Ј10-20 | Cu(OH)2 | 1,6Ј10-19 | PbS | 2,5Ј10-27 |
| Al(OH)3 | 5Ј10-33 | CuS | 6,3Ј10-36 | PbSO4 | 1,6Ј10-8 |
| AlPO4 | 5,7Ј10-19 | Fe(OH)2 | 8Ј10-16 | Pb3(PO4)2 | 7,9Ј10-43 |
| BaCO3 | 5,1Ј10-9 | Fe(OH)3 | 6,3Ј10-38 | Sb2S3 | 1,6Ј10-93 |
| BaCrO4 | 1,2Ј10-10 | FePO4 | 1,3Ј10-22 | SrCO3 | 1,1Ј10-10 |
| BaSO4 | 11Ј10-10 | FeS | 5Ј10-18 | SrCrO4 | 3,6Ј10-5 |
| Ba3(PO4)2 | 6,0Ј10-39 | HgS | 1,6Ј10-52 | SrF2 | 2,5Ј10-9 |
| BeCO3 | 1Ј10-3 | MgCO3 | 2,1Ј10-5 | SrSO4 | 3,2Ј10-7 |
| CaCO3 | 4,8Ј10-9 | Mg(OH)2 | 6Ј10-10 | ZnCO3 | 1,4Ј10-14 |
| CaF2 | 4,0Ј10-11 | Mg3(PO4)2 | 1Ј10-13 | Zn(OH)2 | 1Ј10-17 |
| CaHPO4 | 2,7Ј10-7 | MnCO3 | 1,8Ј10-11 | α-ZnS | 1,6Ј10-24 |
| Ca(H2PO4)2 | 1Ј10-3 | Mn(OH)2 | 1,9Ј10-13 | Zn3(PO4)2 | 9,1Ј10-33 |
“аблица 3
| »онна€ сила раствора I | «ар€д иона z | »онна€ сила раствора I | «ар€д иона z | |||||
| ±1 | ±2 | ±3 | ±1 | ±2 | ±3 | |||
| 0,001 | 0,98 | 0,78 | 0,73 | 0,1 | 0,81 | 0,44 | 0,16 | |
| 0,002 | 0,97 | 0,74 | 0,66 | 0,2 | 0,80 | 0,41 | 0,14 | |
| 0,005 | 0,95 | 0,66 | 0,55 | 0,3 | 0,81 | 0,42 | 0,14 | |
| 0,01 | 0,92 | 0,60 | 0,47 | 0,4 | 0,82 | 0,45 | 0,17 | |
| 0,02 | 0,90 | 0,53 | 0,37 | 0,5 | 0,84 | 0,50 | 0,21 | |
| 0,05 | 0,84 | 0,50 | 0,21 |
—ѕ»—ќ –≈ ќћ≈Ќƒ”≈ћќ… Ћ»“≈–ј“”–џ
1. јхметов Ќ.—. ќбща€ и неорганическа€ хими€. - ћ.: ¬ысша€ школа, 2002.-743 с.
2. √линка Ќ.Ћ., ≈рмаков ј.». ќбща€ хими€. - ћ.: »нтеграл-пресс, 2004 - 728 с.
3. арапетъ€нц ћ.’., ƒракин —.». ќбща€ и неорганическа€ хими€. - ћ.: ’ими€, 2000 Ц 532 с..
4. √линка Ќ.Ћ. «адачи и упражнени€ по общей химии. 2004, ћ. Ђ»нтеграл-ѕрессї, с. 240.
«адание є6
по теме: Ђ√»ƒ–ќЋ»« —ќЋ≈…ї
¬опросы и задачи дл€ самосто€тельного решени€
1. Ќаписать ионно-молекул€рные уравнени€ реакции гидролиза с указанием pH при растворении в воде следующих солей: сульфата цинка, нитрата кали€, хлорида цези€, сульфата хрома (III).
|
|
|
2. Ќаписать в ионно-молекул€рной форме уравнени€ гидролиза следующих солей: K2S, K2CO3, Li2S, K3PO4, K2SO3.
3. Ќаписать в молекул€рной форме уравнение гидролиза гидросолей и определить pH среды их водных растворов: NaHSO3, NaHS, KHCO3, Na2HPO4, NaH2PO4.
4. Ќаписать в молекул€рно-ионной форме уравнение гидролиза солей и указать реакцию их водных растворов: ZnCI2, Cu(NO3)2, FeSO4, AICI3, CrCI3.
5. Ќаписать молекул€рно-ионные формы уравнений гидролиза (отдельно дл€ катиона и аниона) и указать реакцию водных растворов солей: (NH4)2S, (NH4)2SO3, (NH4)3PO4, (NH4)2HPO4.
6. Ќаписать молекул€рно-ионные формы уравнений гидролиза и указать реакцию водных растворов солей:
а) NH4HS; б) NH4HSO3; в)NH4H2PO4.
7. Ќаписать молекул€рно-ионные формы уравнений гидролиза и указать реакцию водных растворов солей:
а) AI(CH3COO)3; б) Fe(HCOO)3; в) Cu(CH3COO)2.
8. Ќаписать в молекул€рной и молекул€рно-ионной форме уравнени€ реакций и объ€снить механизм их протекани€:
 а) Fe2(SO4)3 + Na2CO3 + H2O......
а) Fe2(SO4)3 + Na2CO3 + H2O......
б  ) AICI3 + (NH4)2S + H2O......
) AICI3 + (NH4)2S + H2O......
 в) Cr2(SO4)3 + (NH4)2S + H2O......
в) Cr2(SO4)3 + (NH4)2S + H2O......
9. Ќаписать в молекул€рной и молекул€рно-ионной форме уравнени€ реакций и объ€снить механизм их протекани€:
 а) AICI3 + CH3COONa + H2O AI(OH) (CH3COO)2 +......
а) AICI3 + CH3COONa + H2O AI(OH) (CH3COO)2 +......
 б) CuSO4 + Na2CO3 + H2O [Cu(OH)]2CO3 +......
б) CuSO4 + Na2CO3 + H2O [Cu(OH)]2CO3 +......
 в) Na2SiO3 + NH4CI + H2O......
в) Na2SiO3 + NH4CI + H2O......
10. ”казать, не производ€ вычислений, в каком из растворов двух солей равной концентрации pH больше или меньше (см. табл.):
а) NaCIO4 и NaCIO;
б) K2S и K2Se;
в) Na2CO3 и NaHCO3.
11. ”казать, не производ€ вычислений, в каком из растворов двух солей равной концентрации pH больше или меньше (см. табл.).
а) CH3COONa и HCOONa;
б) Na2CO3 и NaSO3 ;
в) HCOONa и HCOONH4.
12. ќхарактеризовать поведение в растворе следующих солей и указать реакцию их водных растворов: HCOOK, NH4Br, K2HPO4, Cu(NO3)2.
13. –аствор NaH2PO4 имеет слабо кислую, а раствор Na3PO4 имеет сильнощЄлочную реакцию. ќбъ€снить эти факты, подтвердить уравнени€ми.
14. ѕри сливании водных растворов Cr(NO3)3 и Na2S образуетс€ осадок Cr(OH)3 и выдел€етс€ газ. —оставить молекул€рное и ионно-молекул€рное уравнение реакций.
15. акие из перечисленных солей подвергаютс€ гидролизу:
а) NaHCO3; б) NaCN; в) KNO3.
Ќаписать молекул€рные и ионно-молекул€рные уравнени€ гидролиза.
16. ¬ычислить константу гидролиза фторида кали€, определить степень гидролиза этой соли в 0,01 ћ растворе и pH раствора.
17. ¬ычислить pH 0,02 н. раствора соды Na2CO3, учитыва€ только первую ступень гидролиза.
18. —равнить степень гидролиза соли и pH среды в 0,1 ћ и 0,001 ћ растворах цинанида кали€.
19. ѕри 600— ионное произведение воды H2O = 10-12. —чита€, что константа диссоциации хлорноватистой кислоты не измен€етс€ с температурой, определить pH 0,001Ќ раствора KOCI при 25 и 600—.
20. pH 0,1ћ раствора натриевой соли органической одноосновной кислоты равен 10.
¬ычислить диссоциации этой кислоты.
21. »сход€ из значений диссоциации соответствующих кислот и оснований, указать реакцию водных растворов следующих солей: NH4CN, NH4F, (NH4)2S.
22. ѕочему раствор NaHCO3 имеет слабощелочную среду, а NaHSO3 слабокислую реакцию? ќтвет обосновать.
23. ¬ычислить константу гидролиза карбоната натри€, степень гидролиза соли в 0,1 ћ растворе и pH среды.
24. ¬ычислить константу гидролиза ортофосфата кали€. аков pH в 2,4 ћ растворе Na3PO4? ќпределить степень гидролиза.
25. ¬ычислить константу гидролиза ортофосфата кали€. аков pH в 3н. растворе Na3PO4. акова степень гидролиза?
26. ¬ычислить константу гидролиза сульфита натри€, степень гидролиза и pH 0,6 ћ раствора.
27. ¬ычислите pH 0,2 ћ раствора NaHCOO, если KHCOOH =1,7∙10-4.
28. ќцените pH раствора, полученного растворением 0,001г NH4CI в 10л воды. KNH4OH = 1,8∙10-5.
29. ¬ чем состоит отличие реакций гидролиза AI(CH3COO)3 и AI2S3 ?
30. ќбъ€сните, почему при введении в раствор FeCI3 раствора соды в осадок выпадает не карбонат железа, а его гидроксид. Ќапишите уравнение процессов.
|
|
|
31. —оставьте уравнение реакций, протекающих в водных растворах:
 а) AICI3 + H2O
а) AICI3 + H2O
 б) AICI3 + (NH4)2S + H2O
б) AICI3 + (NH4)2S + H2O
 в) AICI3 + (NH4)2CO3 + H2O
в) AICI3 + (NH4)2CO3 + H2O
32. ѕредполагают, что гидролиз буры протекает в 2 стадии:

 B4O72- + 3H2O 2H3BO3 + 2BO2-
B4O72- + 3H2O 2H3BO3 + 2BO2-

 BO2- + 2H2O H3BO3 + OH-
BO2- + 2H2O H3BO3 + OH-
ак рассчитать pH раствора буры известной концентрации?
33. ¬ычислите pH раствора, в 5л которого содержитс€ 20 г NH4CI, если KNH4OH = 1,8∙ 10-5.
34. ” какого раствора pH больше: SnCI2 или SnCI4 (при одинаковых концентраци€х)?
35. ќтличаетс€ ли гидролиз AICI3 от AIF3? ќтвет обосновать уравнени€ми.
36. ѕриведите возможные способы смещени€ равновеси€ реакций гидролиза вправо и влево.
37. ѕриготовлены растворы солей Na2CO3, FeCI3 и CuSO4. Ќапишите по-стадийно уравнени€ гидролиза. ак осуществить последнюю стадию гидролиза?
38. –асположите соединени€ Na2CO3, NaHCO3 и NaOH в пор€дке увеличени€ pH их растворов одинаковой концентрации.
39. Ќапишите сокращенным молекул€рно-ионным способом уравнени€ реакций гидролиза следующих солей:
1) FeCI3; 2) Fe2(SO4)3; 3) Fe(OH)CI2.
40. Ќапишите сокращЄнное молекул€рно-ионное уравнение реакций гидролиза следующих солей:
1) NaNO3; 2) NH4NO2; 3) NH4NO3.
41. Ќапишите сокращЄнное молекул€рно-ионное уравнение реакций гидролиза следующих солей:
1) Zn(NO3)2; 2) Cu(NO3)2; 3)Ca(NO2)2.
42. Ќапишите сокращЄнное молекул€рно-ионное уравнение реакций гидролиза следующих солей:
1) Cu(CH3COO)2; 2) CuSO4; 3) Cu(OH)NO3.
”кажите реакцию среды.
43. ”кажите способы смещени€ равновеси€ реакций гидролиза вправо:

 CO32- + H2O HCO3- + OH-
CO32- + H2O HCO3- + OH-

 HCO3- + H2O H2CO3 + OH-
HCO3- + H2O H2CO3 + OH-
44. акие соли железа гидролизуютс€ сильнее: FeCI2 или FeCI3 и почему? ќтвет обосновать.
45. ¬ычислите pH 0,1 ћ раствора NH4CI (KNH4OH = 1,8 ∙10-5).
46. –аствор, содержащий в 1л 3,81 г тетрабората натри€ Na2B4O7 ∙ 10H2O (бура), имеет pH = 9,18. Ќапишите уравнение реакции гидролиза и вычислите константу первой стадии гидролиза, предполага€, что она обусловливает щЄлочную среду раствора.
47. ¬ычислите константу диссоциации BeOH+
BeOH+ = Be2+ + OH-
исход€ из того, что pH 2 ∙ 10-2ћ раствора BeCI2 составл€ет 4,2.
48. —оли, образованные многоосновными кислотами и многокислотными основани€ми, гидролизуютс€ по ступен€м. ƒокажите, что полна€ константа гидролиза равна произведению константы гидролиза всех ступеней гидролиза.
49. ¬ычислите pH 0,2 ћ раствора NaHCOO, если KЌ—ќќЌ = 1,8 ∙ 10-4.
50.Ќапишите сокращенное молекул€рно-ионное уравнение реакций гидролиза следующих солей (если возможно):
NaCIO4, NH4CIO4, Na2S.
51. ѕри каких услови€х можно ожидать (теоретически) нейтральную реакцию среды раствора соли?
52. –еакци€ среды водного раствора MgCI2 нейтральна. ќ чЄм это говорит?
53. Ќапишите сокращенное молекул€рно-ионное уравнение реакций гидролиза следующих солей:
NaCH3COO, NH4CH3COO, Fe(OH)SO4.
54. ¬одные растворы HCI и FeCI3 показывают кислую среду. Ёто объ€сн€етс€ протеканием следующих процессов:
HCI + H2O = H2O+ +CI-
FeCI3 + 2H2O H3O+ + CI- + FeOHCI2
”кажите признаки сходства и различи€ этих процессов. Ќазовите процессы.
55. –азбавленные растворы LiJ и CsF нейтральны. ѕо мере повышени€ концентрации раствор LiJ начинает показывать кислую реакцию, а раствор CsF - щЄлочную. ак это объ€снить?
56. ѕредскажите реакцию среды (кисла€ или щЄлочна€) водных растворов Na3PO4, Na2HPO4 и NaH2PO4.
57. Ќапишите сокращенное молекул€рно-ионное уравнение реакций гидролиза следующих солей:
Fе(NO3)3, Fe(OH)2NO3, Cu(NO3)2.
58. ќпределите pH 0,1 ћ раствора ортофосфата кали€.
|
|
|
59. ” какого раствора рЌ больше: FeCl2 или FeCl3 (при одинаковых концентраци€х)?
60. ќпределите рЌ 0,1 ћ раствора дегидрофосфата кали€.
¬арианты контрольных заданий
| ¬ариант | Ќомер задачи | |||
ѕримеры решени€ задач
ѕример 1. ¬ычислить степень гидролиза цианида кали€ при концентрации 0,1 и 0,001 г-экв/л, если константа диссоциации HCN=7,2∙10-10.
–ешение:
1) «апишем уравнение диссоциации HCN:

 HCN H+ + CN-
HCN H+ + CN-
KHCN = 
2) «апишем уравнение гидролиза KCN в 3-х формах:
а) молекул€рной

 KCN + H2O HCN + KOH;
KCN + H2O HCN + KOH;
 б) ионно-молекул€рной
б) ионно-молекул€рной


 K+ + CN- + H2O HCN + K+ + OH- ;
K+ + CN- + H2O HCN + K+ + OH- ;
в) в краткой ионно-молекул€рной форме

 CN- + H2O HCN + OH-.
CN- + H2O HCN + OH-.
ѕодставл€ем данные значени€ в формулу определени€ степени гидролиза:
h =  =
=  = 0.0118,
= 0.0118,
% = 0.011∙8100 = 1.18%; h = 1,18.
ƒл€ с= 0,001  имеем h =
имеем h =  =
= 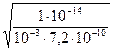 = 0.118 или
= 0.118 или
1,18%.
ѕример 2. ¬ычислить гидролиза, h и pH 0,1 моль/л раствора CH3COONa.
–ешение:
«апишем уравнение гидролиза CH3COONа в 3-х формах:
а) молекул€рной

 CH3COONa + H2O CH3COOH + NaOH;
CH3COONa + H2O CH3COOH + NaOH;
б) ионно-молекул€рной

 CH3COO- + Na+ + H2O CH3COOH + Na+ + OH- ;
CH3COO- + Na+ + H2O CH3COOH + Na+ + OH- ;
в) краткой ионно-молекул€рной

 CH3COO- + H2O CH3COOH + OH-.
CH3COO- + H2O CH3COOH + OH-.
Kг =  ,
,
KCH3COOH = 1,74 ∙10-5,
KH2O = 10-14,
Kг =  = 5,7 ∙10-10;
= 5,7 ∙10-10;
степень гидролиза h определ€ем по формуле
h =  =
=  = 7,4∙10-5 или h = 0,0076 %
= 7,4∙10-5 или h = 0,0076 %
[OH-]- = c ∙ h
[OH-] = 10-1 ∙ 7,6 ∙ 10-6 моль;
ионное произведение воды
[H+] ∙ [OH-] = KH2O,
отсюда [H+] =  ; pH = - Ig [H+],
; pH = - Ig [H+],
pH = - Ig  ≈ 6,88.
≈ 6,88.
ѕример 3. ¬ычислить, чему равна константа гидролиза, h и pH 0,1 моль/л раствора фосфата натри€ Na3PO4.
–ешение:
«апишем уравнение гидролиза по 3-м ступен€м в 3-х формах:
I ступень:
а) молекул€рна€

 Na3PO4 + H2O Na2HPO4 + NaOH;
Na3PO4 + H2O Na2HPO4 + NaOH;
б) ионно-молекул€рна€




 3Na+ + PO43- + H2O 2Na+ + (HPO4)2- + Na+ + OH-;
3Na+ + PO43- + H2O 2Na+ + (HPO4)2- + Na+ + OH-;
в) кратка€ ионно-молекул€рна€

 PO43- + H2O HPO42- + OH- .
PO43- + H2O HPO42- + OH- .
II ступень:
а) молекул€рна€

 Na2HPO4 + H2O NaH2PO4 + NaOH;
Na2HPO4 + H2O NaH2PO4 + NaOH;
б) ионно-молекул€рна€




 2 Na+ + HPO42- + H2O Na+ + H2PO-4 + Na+ + OH-;
2 Na+ + HPO42- + H2O Na+ + H2PO-4 + Na+ + OH-;
в) кратка€ ионно-молекул€рна€

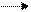 HPO42- + H2O H2PO4- + OH- .
HPO42- + H2O H2PO4- + OH- .
III ступень:
а) молекул€рна€

 NaH2PO4 + H2O H3PO4 + Na+ + OH-;
NaH2PO4 + H2O H3PO4 + Na+ + OH-;
б) ионно-молекул€рна€

 Na+ + H2PO4- + H2O H3PO4 + NaOH;
Na+ + H2PO4- + H2O H3PO4 + NaOH;
в) кратка€ ионно-молекул€рна€

 H2PO4 + H2O H3PO4 + OH-.
H2PO4 + H2O H3PO4 + OH-.
’от€ ионы HPO42- и H2PO4- способны гидролизоватьс€, однако степени гидролиза ионов HPO42- и H2PO4- малы.
¬торой и третьей стадией гидролиза можно пренебречь.
“огда гидролиза, h и pH раствора определ€ем дл€ уравнени€

 PO43- + H2O HPO4 + OH-
PO43- + H2O HPO4 + OH-
»з табл. 2 имеем:
KH3PO4 = 7,6 ∙ 10-3,
KH2PO4- = 6,2 ∙ 10-8,
KHPO42- = 44 ∙ 10-13,
г =  =
=  =
=  = 0,023.
= 0,023.
ƒл€ определени€ h воспользуемс€ формулой
h =  =
=  = 0.377, или в процентах 37,7%.
= 0.377, или в процентах 37,7%.
ќтсюда
[OH-] = ch = 0,1∙ 0,377 = 3,77 ∙ 10-2,
pOH ≈ 1,42,
pH = 10 Ц 1,42 = 12,6.
ѕример 4. ак будет измен€тьс€ pH при растворении в воде солей
1. CuCl2, 2. Na2SO3, 3. K2SO4 ?
–ешение:
1) √идролиз соли CuCI2 проходит ступенчато (в основном по I ступени) по катиону Cu2+.
ѕерва€ ступень гидролиза в 3-х формах:
а) молекул€рна€

 CuCI2 + H2O Cu(OH)CI + HCI;
CuCI2 + H2O Cu(OH)CI + HCI;
б) ионно-молекул€рна€




 Cu2+ + 2CI- + H2O (CuOH)+ + CI- + H+ + CI-;
Cu2+ + 2CI- + H2O (CuOH)+ + CI- + H+ + CI-;
в) кратка€ ионно-молекул€рна€
 Cu2+ + H2O (CuOH)+ + H+.
Cu2+ + H2O (CuOH)+ + H+.
¬тора€ ступень гидролиза практически не протекает:
а) молекул€рна€

 Cu(OH)CI + H2O (CuOH)2↓ + HCI;
Cu(OH)CI + H2O (CuOH)2↓ + HCI;
б) ионно-молекул€рна€



 (CuOH)+ + CI- + H2O (CuOH)2↓ + H+ + CI-;
(CuOH)+ + CI- + H2O (CuOH)2↓ + H+ + CI-;
в) кратка€ ионно-молекул€рна€

 (CuOH)+ + H2O (CuOH)2↓ + H+.
(CuOH)+ + H2O (CuOH)2↓ + H+.
2) √идролиз соли Na2SO3 гидролиз протекает по аниону.
I ступень гидролиза:
а) молекул€рна€

 Na2SO3 + H2O NaHSO3 + NaOH;
Na2SO3 + H2O NaHSO3 + NaOH;
б) ионно-молекул€рна€

 2Na+ + SO32- + H2O Na+ + HSO3- + Na+ + OH- ;
2Na+ + SO32- + H2O Na+ + HSO3- + Na+ + OH- ;
в) кратка€ ионно-молекул€рна€

 SO32- + H2O HSO3- + OH-.
SO32- + H2O HSO3- + OH-.
II ступень гидролиза:
а) молекул€рна€

 NaHSO3 + H2O H2SO3 + NaOH;
NaHSO3 + H2O H2SO3 + NaOH;
б) ионно-молекул€рна€
|
|
|

 Na+ + HSO3-+ H2O H2SO3 + Na+ + OH-;
Na+ + HSO3-+ H2O H2SO3 + Na+ + OH-;
в) кратка€ ионно-молекул€рна€

 HSO3-+ H2O H2SO3 + OH-.
HSO3-+ H2O H2SO3 + OH-.
3) —оль K2SO4 образована сильным основанием ( ќЌ) и сильной кислотой (Na2SO4).
√идролизу не подвергаетс€, pH = 7.
ѕример 5. «акончить уравнение реакции с учЄтом возможности необратимого гидролиза образуемых солей.
 AI2(SO4)3 + Na2S + H2O?
AI2(SO4)3 + Na2S + H2O?
–ешение:
√идролиз в данном случае идЄт до конца, так как образуютс€ осадок AI(OH)3 и газ H2S.
”равнение гидролиза будет выгл€деть следующим образом:
 AI2(SO4)3 + 3Na2S + 6H2O 3Na2SO4 + 2AI(OH)3↓ + 3H2S↑
AI2(SO4)3 + 3Na2S + 6H2O 3Na2SO4 + 2AI(OH)3↓ + 3H2S↑
онстанты диссоциации некоторых слабых электролитов
в водных растворах при 250—
| Ёлектролит | |||
| јзотиста€ кислота | HNO2 | 2,6 ∙10-5 | |
| јммони€ гидроксид | NH4OH | 4 ∙10-4 | |
| Ѕорна€ кислота | H3BO3 | K1 | 5,8 ∙10-4 |
| Ѕромноватиста€ кислота | HOBr | 2,1 ∙ 10-9 | |
| ¬одорода пероксид | H2O2 | K1 | 2,6 ∙10-12 |
| ремниева€ кислота | H2SiO3 | K1 K2 | 2,2 ∙10-10 1,6 ∙10-12 |
| ћуравьина€ кислота | HCOOH | 1,8 ∙10-4 | |
| —ерниста€ кислота | H2SO3 | K1 K2 | 1,6 ∙ 10-2 6,3 ∙10-8 |
| —ероводород | H2S | K1 K2 | 6 ∙10-8 1 ∙10-14 |
| ”гольна€ кислота | H2CO3 | K1 K2 | 4,5 ∙10-7 4,7 ∙10-11 |
| ”ксусна€ кислота | CH3COOH | 1,8 ∙10-5 | |
| ‘осфорна€ кислота | H3PO4 | K1 K2 K3 | 7,5 ∙ 10-3 6,3 ∙10-8 1,3 ∙10-12 |
| ÷иановодород | HCN | 7,9 ∙10-10 | |
| ўавелева€ кислота | H2C2O4 | K1 K2 | 5,4 ∙10-2 5,4 ∙10-5 |
—писок рекомендуемой литературы
1. арапеть€нц ћ.’., ƒракин —.».. ќбща€ и неорганическа€ хими€.- ћ.: ’ими€, 2001.- 632 с.
2. «айцев ќ.—. «адачи и вопросы по химии.- ћ.: ’ими€, 1985.- 301 с.
3. √линка Ќ.Ћ. «адачи и упражнени€ по общей химии.- Ћ.: ’ими€, 2004.- 270 с.
4. √ольбрайх «.≈. —борник задач и упражнений по химии.- ћ.: ¬ысша€ школа, 2004.- 224 с.
«адание є 7
по теме: ЂќкислительноЦосстановительные реакции. Ёлектрохими€ї
«адани€
1. ѕодберите коэффициенты в следующих реакци€х ионно-электронным методом. ”кажите окислитель и восстановитель, определите направление протекани€ реакции и вычислите э.д.с.
2. ¬ычислите э.д.с. гальванического элемента, составленного из стандартных электродов. ќпределите направление тока во внешней цепи указанного гальванического элемента Me|Me+n||Me+n|Me
Ќa каком электроде будет идти растворение металла?
3. акой металл будет подвергатьс€ коррозии, если заданна€ пара металлов, наход€щихс€ в контакте, попадет в кислую среду? —оставьте схему образующегос€ при этом гальванического элемента.
4. акой металл €вл€етс€ анодным (катодным) по отношению к покрываемому металлу? —оставьте уравнени€ реакций, протекающих при атмосферной коррозии (во влажном воздухе).
5. ќпределите электродный потенциал электрода, в котором металлическа€ пластинка погружена в раствор собственной соли с заданной концентрацией катионов.
6. —оставьте схемы электролиза водного раствора заданного вещества
а) с инертным анодом.
б) с использованием активного анода
«адани€ с первого по шестой Ђбї выполн€ютс€ на основании таблицы
| є вари-антов | Ќомера заданий | |||||||
| 1 реак-ции | 6а | 6б | ѕриме-ча-ние | |||||
| 1, 4, 89 | Cu|Cu+2||Fe+2|Fe | Na/Ba | Fe покрыт Ni Fe покрыт Mn | Pb|Pb+2 0,0012 н | NaOH | Zn | ||
| 2, 6, 87 | Fe|Fe+2||Zn+2|Zn | Ca/Ni | Zn покрыт AI Zn покрыт Cd | Zn|Zn+2 2 ћ | BaCI2 | Ag | ||
| 3, 8, 88 | Mg|Mg+2||Fe+2|Fe | Sr/Ca | Mg покрыт Ca Mg покрыт Zn | Ni|Ni+2 2 ћ | HNO3 | AI | ||
| 5, 12,34 | Mg|Mg+2||Zn+2|Zn | Cu/Fe | Mn покрыт Mg Mn покрыт Fe | Zn|Zn+2 2 н | KNO3 | Ni | ||
| 7, 16,79 | Zn|Zn+2||Sn+2|Sn | Zn/Sn | Pb покрыт Cu Pb покрыт Cо | Zn|Zn+2 0,001 ћ | NaBr | Fe | ||
| 9, 19,37 | Zn|Zn+2||AI+3|AI | Fe/Ag | AI покрыт Sr AI покрыт Cu | Cd|Cd+2 0,1 ћ | Ba(NO3)2 | Cu | ||
| 10, 21,44 | Ag|Ag+||Mg+2|Mg | Na/Ag | Cr покрыт Mn Cr покрыт Sn | Ag|Ag+ 1 н | Zn(NO3)2 | Co | ||
| 11, 32,50 | AI| AI+3||Mg+2|Mg | AI/Ag | Cd покрыт Mg Cd покрыт Pb | Fe|Fe+2 0,002 н | AICI3 | Pb | ||
| 13, 36,42 | Pb|Pb+2||Ni+2|Ni | Ni/Pb | Ni покрыт Zn Ni покрыт Cu | Cu|Cu+2 0.1 н | Cu(NO3)2 | Mn | ||
| 14, 28,56 | Mn|Mn+2||Ni+2|Ni | Na/Zn | Fe покрыт Zn Fe покрыт Cu | Ag|Ag+ 2 н | Pb(NO3)2 | Mg | ||
| 15, 39,43 | Pb|Pb+2||Ag+|Ag | Mn/Ni | Zn покрыт Ag Zn покрыт Mg | Cd|Cd+2 0,01 ћ | AI(NO3)3 | Sn | ||
| 17, 48,51 | AI| AI+3||Sn+2|Sn | Pb/Fe | Pb покрыт Bi Pb покрыт Ni | Cu|Cu+2 0,2 ћ | AgNO3 | AI | ||
| 18, 53,60 | Sn|Sn+2||Cu+2|Cu | Ca/AI | AI покрыт Mg AI покрыт Pb | Ag|Ag+ 0,02 ћ | MnCI | Zn | ||
| 20, 64,63 | Zn|Zn+2||Ni+2|Ni | Mn/Sn | Ni покрыт Cr Ni покрыт Ag | Cu|Cu+2 2 ћ | Sn(NO3)2 | Ag | ||
| 22, 67,70 | Cu|Cu+2||AI+3|AI | Zn/Ni | Mg покрыт Ca Mg покрыт Cu | Sn|Sn+2 0,0045 ћ | Ni(SO4)2 | Fe | ||
| 23, 68,78 | Zn|Zn+2||Pb+2|Pb | AI/Cu | Sb покрыт Cr Sb покрыт Ag | Zn|Zn+2 0,01 ћ | H2SO4 | Pb | ||
| 30, 65,66 | Zn|Zn+2||Ag+|Ag | Na/Zn | AI покрыт Ca AI покрыт Zn | Sn|Sn+2 2 ћ | Na2SO4 | Mg | ||
| 31, 73,62 | Ag|Ag+||Sn+2|Sn | Zn/Ag | Fe покрыт Ni Feпокрыт Mn | Ni|Ni+2 0,014 н | ZnSO4 | Mn | ||
| 35, 61,72 | Mn|Mn+2||Pb+2|Pb | Na/Fe | Zn покрыт Cd Zn покрыт AI | Ag|Ag+ 2 ћ | KOH | Fe | ||
| 38, 80,69 | Ni|Ni+2||AI+3|AI | Mn/Ni | Mg покрыт Ca Mg покрыт Zn | Cu|Cu+2 0,05 ћ | CuCI2 | Cu | ||
| 40, 82,71 | Ni|Ni+2||Ag+|Ag | Zn/Pb | Mn покрыт Fe Mn покрыт Mg | Cd|Cd+2 3 ћ | HCI | Ni | ||
| 45, 81,41 | Ni|Ni+2||Cu+2|Cu | Mn/Pb | Pb покрыт Cr Pb покрыт Cu | Fe|Fe+2 0,15 ћ | Na2CO3 | Co | ||
| 46, 84,77 | AI|AI+3||Fe+2|Fe | Ag/K | AI покрыт Cu AI покрыт Ca | Ni|Ni+2 0,002 н | NiCI2 | Pb | ||
| 47, 83,58 | Pb|Pb+2||Cu+2|Cu | Ni/K | Cr покрыт Sn Cr покрыт Mn | Zn|Zn+2 0,21 ћ | Ca(NO3)2 | Ag | ||
| 24,49,74 | Zn|Zn+2||Pb+2|Pb | Fe/Mg | Cd покрыт Cd Cd покрыт Mg | Pb|Pb+2 0,1 н | MgBr2 | Zn | ||
| 52, 25,85 | Mn|Mn+2||Fe+2|Fe | Zn/Pb | Ni покрыт Cu Ni покрыт Zn | Mn|Mn+2 0,005 ћ | AgNO3 | AI | ||
| 54,29,86 | Mg|Mg+2||Pb+2|Pb | Sn/Mn | Fe покрыт Ag Fe покрыт Zn | Mg|Mg+2 2 ћ | NiBr2 | Ni | ||
| 26,33,75 | Pb|Pb+2||Cu+2|Cu | Pb/Na | Zn покрыт Cu Zn покрыт Mg | AI|AI+3 0.003 н | FeCI2 | Co | ||
| 55, 59,76 | Cr|Cr+3||Cu+2|Cu | Ag/Cu | Fe покрыт Ni Fe покрыт Mn | Pb|Pb+2 0,012 н | PbCI2 | Ag | ||
| 57, 27,90 | Cr|Cr+3||Mg+2|Mg | Mg/Cu | Zn покрыт AI Zn покрыт Cd | Zn|Zn+2 0,4 ћ | MgI2 | AI |






