√рав≥метричний метод досл≥дженн€ короз≥њ метал≥в
ѕри оц≥нц≥ короз≥йноњ тривкост≥ метал≥в довол≥ розповсюджений грав≥метричний метод. ¬≥н застосовуЇтьс€ в двох вар≥антах: 1) визначенн€ зб≥льшенн€ маси зразка внасл≥док утворенн€ продукт≥в короз≥њ на його поверхн≥ ( ) ≥ 2) визначенн€ втрат маси п≥сл€ видаленн€ продукт≥в короз≥њ (
) ≥ 2) визначенн€ втрат маси п≥сл€ видаленн€ продукт≥в короз≥њ (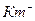 ). ƒругий вар≥ант б≥льш поширений через свою ун≥версальн≥сть [23].
). ƒругий вар≥ант б≥льш поширений через свою ун≥версальн≥сть [23].
Ўвидк≥сть короз≥њ (за першим вар≥антом) можна визначити за формулою:
 (4.1)
(4.1)
де m 1 Ч маса вих≥дного зразка, г; m 2 Ч маса зразка разом ≥з продуктами короз≥њ, г; S Ч площа поверхн≥ зразка, см2; tЧ час ≥спиту, год.; п Ч коеф≥ц≥Їнт, що залежить в≥д складу продукт≥в короз≥њ. ÷€ формула застосовна т≥льки в тих випадках, коли в≥домий х≥м≥чний склад продукт≥в короз≥њ, знайдений за допомогою х≥м≥чного або рентгенограф≥чного анал≥зу. ќстанн€ обставина Ї ≥стотним недол≥ком першого вар≥анта грав≥метричного методу; в≥н застосовний в основному дл€ досл≥дженн€ газовоњ короз≥њ, при €к≥й на поверхн≥ металу утворюЇтьс€ лише нег≥дратований оксид, тому що при висок≥й температур≥ не може утворитис€ шар вологи.
ѕеревага першого вар≥анта пол€гаЇ в тому, що у випадку його застосуванн€ можна простежити за к≥нетикою процесу на тих самих зразках, оск≥льки ведетьс€ спостереженн€ за зб≥льшенн€м њхньоњ маси.
ѕри використанн≥ другого вар≥анта грав≥метричного методу продукти короз≥њ видал€ютьс€ р≥зними розчинами, взаЇмод≥ючими не з основним металом, а лише з продуктами короз≥њ. ѕор€д з цим ставл€ть ≥ так звану Ђсл≥пуї пробу, тобто оброблюють неекспонован≥ зразки, щоб одержати дан≥ про розчиненн€ основного металу.
Ўвидк≥сть короз≥њ за другим вар≥антом грав≥метричного методу розраховують за формулою:
 (4.2)
(4.2)
де m 2 Ч маса зразка п≥сл€ видаленн€ продукт≥в короз≥њ. ” цьому випадку х≥м≥чний склад продукт≥в короз≥њ не маЇ значенн€ дл€ результату розрахунку. «а втратами маси можна оц≥нювати короз≥йну тривк≥сть металу в балах, припускаючи, що метал руйнуЇтьс€ р≥вном≥рно. ƒл€ характеристики короз≥йноњ тривкост≥ ( “) зразка використовують 10 бальну шкалу “ (таблиц€ 4.1) [4].
“аблиц€ 4.1 - ƒес€тибальна шкала короз≥йноњ тривкост≥ метал≥в (за √ќ—“ 13819-68)
| √рупа тривкост≥ | Ўвидк≥сть короз≥њ металу, мм/р≥к | Ѕал |
| ÷≥лком тривк≥ | ћенш 0,001 | |
| ¬ельми тривк≥ | ѕонад 0,001 до 0,005 | |
| ѕонад 0,005 до 0,01 | ||
| “ривк≥ | ѕонад 0,01 до 0,05 | |
| ѕонад 0,05 до 0,1 | ||
| ѕониженотривк≥ | ѕонад 0,1 до 0,5 | |
| ѕонад 0,5 до 1,0 | ||
| ћалотривк≥ | ѕонад 1,0 до 5,0 | |
| ѕонад 5,0 до 10,0 | ||
| Ќетривк≥ | ѕонад 10,0 |
ћетод визначенн€ швидкост≥ короз≥њ за втратами маси маЇ р€д недол≥к≥в: важко повн≥стю видалити вс≥ продукти короз≥њ, не торкаючись основного металу, неможливо оц≥нювати нер≥вном≥рну короз≥ю ≥ м≥жкристал≥тне та транскристал≥тне короз≥йне руйнуванн€.
«разки, призначен≥ дл€ грав≥метричних вим≥р≥в, можуть мати р≥зноман≥тну форму, але дл€ одержанн€ швидких ≥ точних результат≥в необх≥дно, щоб площа була максимальною при м≥н≥мальн≥й мас≥, що зв'€зано ≥з можливост€ми анал≥тичних терез≥в. ¬трата маси за величиною повинна бути на один пор€док вище можливост≥ терез≥в, тобто зразки повинн≥ бути €к можна б≥льш тонкими. ѕоверхн€ зразк≥в повинна мати високий ≥ однаковий клас шорсткост≥, тому що зразки з нер≥вною поверхнею, мають реальну площу, що набагато перевищуЇ геометричну. ÷е приводить до похибок у розрахунках.
|
|
|
ќб'Їмний (волюмометричний) метод досл≥дженн€ швидкост≥ короз≥њ метал≥в
ѕри переход≥ металу в продукти короз≥њ вид≥л€Їтьс€ екв≥валентна к≥льк≥сть водню (процес з водневою депол€ризац≥Їю) або поглинаЇтьс€ кисень, розчинений в електрол≥т≥ (процес кисневоњ депол€ризац≥њ). «а обс€гом вид≥леного водню (або поглиненого кисню), вим≥рюваного за допомогою встановленого над зразком евд≥ометра, можна обчислити втрати маси металу. ” випадку короз≥њ, що прот≥каЇ з≥ зм≥шаною депол€ризац≥Їю, схема досл≥ду б≥льш складна [23].
ƒл€ розрахунк≥в використовують так≥ формули та р≥вн€нн€:
 ;
;  (4.3)
(4.3)
з урахуванн€м тиску насиченоњ вод€ноњ пари, мм.рт.ст. (таблиц€ 4.2);
 (4.4)
(4.4)
 (4.5)
(4.5)
 (4.6)
(4.6)
де A - атомна маса, ј = 55,85 Ц дл€ вуглецевоњ стал≥;
n Ц зар€д ≥ону, що утворюЇтьс€: дл€ Fe2+ n = 2;
ρће Ц густина металу, г/см3 (ρFe= 7,85 г/см3)
–Ќ2ќ Ц тиск насиченоњ вод€ноњ пари, мм. рт. ст.(таблиц€ 4.2)
“аблиц€ 4.2 Ц “иск насиченоњ вод€ноњ пари
| “, | |||||||||||
| –Ќ2ќ, мм. рт. ст. | 9,21 | 12,79 | 13,63 | 14,53 | 15,48 | 16,48 | 17,53 | 18,65 | 19,83 | 21,09 | 22,38 |
ороз≥€ метал≥в у кислотах з неокислювальним ан≥оном (Ќ—l, розведена Ќ2Sќ4) прот≥каЇ переважно з водневою депол€ризац≥Їю (Ќ+).
“ому катодний процес (при рЌ<7) в≥дбуваЇтьс€ здеб≥льшого за схемою [4]:
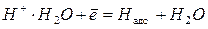 , рЌ = 0;
, рЌ = 0;  = 0 (4.7)
= 0 (4.7)
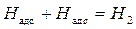 рЌ = 1;
рЌ = 1; 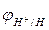 =-0,059 ¬ (4.8)
=-0,059 ¬ (4.8)
—упутн≥м процесом Ї процес водневоњ оклюз≥њ (наводнюванн€).
јле можливе ≥ часткове прот≥канн€ процесу кисневоњ депол€ризац≥њ (аерований розчин електрол≥ту рЌ<7):
 рЌ = 0; j = 1,23 ¬ (4.9)
рЌ = 0; j = 1,23 ¬ (4.9)
„астку кисневоњ депол€ризац≥њ визначають за формулою:
 (4.10)
(4.10)
јнодний процес при рЌ<7, наближено, вигл€даЇ так:
Fe Ѓ Fe2+ + 2ē  = -0,44 ¬ (4.11)
= -0,44 ¬ (4.11)
¬ д≥йсност≥ в≥н багатостад≥йний, €к ≥ катодний процес.
ѕор≥вн€йте наведен≥ парц≥альн≥ короз≥йн≥ процеси з тими що в≥дбуваютьс€ у нейтральному та лужному середовищах (рЌ ³ 7), де термодинам≥чно б≥льш в≥рог≥дн≥ катодн≥ процеси з кисневою депол€ризац≥Їю ( ) [4]:
) [4]:
O2 + 2H2O + 4 ē = 4ќЌ - рЌ = 7; j = 0,815 ¬ (4.12)
рЌ = 14; j = 0,401 ¬
јле й при рЌ ³ 7 можлив≥ процеси з Ќ+-депол€ризац≥Їю (що характерно дл€ метал≥в з великим негативним значенн€м потенц≥алу, наприклад,
дл€ Mg j0 =-2,37 ¬ у розчин≥ NaCl):
H2O + ē = Hадс + ќЌ - (4.13)
—хематичний анодний процес (при рЌ ³ 7) з утворенн€м ≥рж≥ (FeOOH):
Fe Ѓ Fe2+ + 2ē (4.14)
Fe2+ +2ќЌ- Ѓ Fe(ќЌ)2 (4.15)
4Fe(ќЌ)2 + ќ2 + 2Ќ2ќ Ѓ 4Fe(OH)3 (4.16)
Fe(ќЌ)3 Ѓ FeOOH + H2O (4.17)
≈кспериментальна частина
ѕор€док виконанн€ роботи:
1. ѕопередн€ п≥дготовка зразк≥в - знежиренн€ в бензин≥, ацетон≥, еф≥р≥, спирт≥.
2. ¬им≥р площ≥ зразка (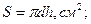

 ), зважуванн€ на анал≥тичних терезах.
), зважуванн€ на анал≥тичних терезах.
3. «разки п≥сл€ знежирюванн€ пом≥щають у евд≥ометри з розчинами сол€ноњ та с≥рчаноњ кислот р≥зноњ концентрац≥њ (без ≥нг≥б≥тору та з ≥нг≥б≥тором) Ц за завданн€м викладача.
|
|
|
4. ѕрот€гом 2 годин реЇструють вид≥ленн€ водню Ќ2 внасл≥док катодного процесу з водневою депол€ризац≥Їю (через кожн≥ 10-15 хв.).
5. ќчистка зразк≥в в≥д продукт≥в короз≥њ, за допомогою ластику та проточноњ води, знежирюванн€, зважуванн€.
6. ќтриман≥ результати розрахунк≥в за формулами 4.2-4.6, записують у таблицю 4.3.
“аблиц€ 4.3 Ц –езультати спостережень та розрахунк≥в
| єпп | ис-лота | “, o— | “рива-л≥сть досл≥-ду, хв. | m 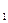 , г , г
| m  , г , г
|  , см , см 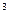
| Km-, 
|  , см , см 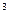 /см /см 
| Kѕ, мм/р≥к | н ic, A/см2 | m ic, A/см2 | 
| Kт, бал |
| Ќ—l: 1н. (+≤н) H2SO4: 1н. (+≤н) | Е Е |
 7. «а даними таблиц≥ 4.3 будують граф≥ки к≥нетики кислотноњ короз≥њ:
7. «а даними таблиц≥ 4.3 будують граф≥ки к≥нетики кислотноњ короз≥њ:


–исунок 4.1 - ≥нетика кислотноњ короз≥њ V0 / S = f(t)
8. –озраховують волюмометричний (н ic) та грав≥метричний показник швидкост≥ короз≥њ (m ic) у струмових одиниц€х (у ј/см2) за такими формулами:
н ic  (4.15)
(4.15)
m ic  (4.16)
(4.16)
де F Ц пост≥йна ‘араде€, F = 26,8 ј×год./г-екв.
n - зар€д ≥ону, що утворивс€ внасл≥док короз≥њ, n = 2
9. ¬изначають частку кисневоњ депол€ризац≥њ за формулою 4.10
ѕри р≥вност≥ н ic = m ic, частка ќ2 - депол€ризац≥њ дор≥внюЇ: 
¬ цьому випадку короз≥€ в≥дбуваЇтьс€ т≥льки з водневою депол€ризац≥Їю.
4.4 ¬исновки: експериментально визначено, що короз≥йна тривк≥сть стал≥ 20 у розчинах (...) становить (...), а при на€вност≥ ≥нг≥б≥тору в (...) короз≥йна тривк≥сть (...), при цьому частка кисневоњ депол€ризац≥њ (...). ¬изначено короз≥йну тривк≥сть за 10-бальною шкалою (...).
4.5 “естов≥ запитанн€ дл€ контролю —–
1. ѕо€сн≥ть сутн≥сть грав≥метричного методу досл≥дженн€ короз≥йноњ тривкост≥ метал≥в.
2. ѕо€сн≥ть сутн≥сть волюмометричного методу досл≥дженн€ короз≥йноњ тривкост≥ метал≥в.
3. яку шкалу використовують дл€ визначенн€ короз≥йноњ тривкост≥ у балах?
4. « €ким видом депол€ризац≥њ переважно прот≥каЇ короз≥€ метал≥в у кислотах з неокислювальним ан≥оном? як≥ електродн≥ процеси мають м≥сце?
5. яким небезпечним €вищем супроводжуЇтьс€ короз≥€ з водневою депол€ризац≥Їю? ўо таке наводнюванн€?
6. « €ким видом депол€ризац≥њ переважно прот≥каЇ короз≥€ метал≥в у нейтральних та лужних середовищах (рЌ ³ 7)? ¬каж≥ть електродн≥ процеси.
7. „и характерн≥ процеси з Ќ+-депол€ризац≥Їю при рЌ ³ 7 (у €ких випадках)?
8. ¬каж≥ть парц≥альн≥ процеси короз≥њ стал≥ в розчин≥ HCl.
9. ¬каж≥ть парц≥альн≥ процеси короз≥њ стал≥ в морський вод≥.
10. ¬каж≥ть парц≥альн≥ процеси короз≥њ стал≥, цинкового сплаву в лужному розчин≥.
Ћабораторна робота є5
¬изначенн€ к≥нетичних та термодинам≥чних параметр≥в короз≥њ стал≥ на потенц≥остат≥ ѕ-5827ћ. ≤нг≥б≥тори короз≥њ та комплексна оц≥нка њх захисноњ д≥њ
5.1 ћета роботи: визначити к≥нетичн≥ та термодинам≥чн≥ параметри короз≥њ стал≥ та оц≥нити ефективн≥сть ≥нг≥б≥тор≥в короз≥њ ≥з застосуванн€м електрох≥м≥чного методу.






